

X-ray tomographic microscopy tightens affinity of the early Cambrian Oymurania to the brachiopod stem group
ARTEM KOUCHINSKY and STEFAN BENGTSON
The geologically rapid biotic evolution in the early Cambrian is marked by the first appearance of major groups of animals in the fossil record (e.g., Budd and Jensen 2000; Kouchinsky et al. 2012). Along with the earliest crown-group representatives of the phylum Brachiopoda, more basal branches of the phylogenetic tree belonging to the stem-group Brachiopoda, such as tannuolinids and mikwitziids, diversified and became widespread during the early Cambrian (e.g., Williams and Holmer 2002; Balthasar et al. 2009; Skovsted et al. 2014). Synchrotron-radiation X-ray tomographic microscopy (SRXTM) of Oymurania gravestocki reveals the microstructure of its calcium-phosphatic shell differentiated into two layers and intersecting systems of canals. The outer layer shows the acrotretoid columnar microstructure and the inner layer consists of continuous prismatic columns. Phosphatized setae preserved within tangential canals, as well as perpendicular canals of Micrina–Setatella type demonstrate homology with the tannuolinid Micrina and the mickwitziid Setatella. A unique and novel combination of microstructural features in Oymurania confirms its evolutionary position within the brachiopod stem group.
Material and methods
The material investigated under the SRXTM consists of two shell fragments of Oymurania gravestocki, SMNH X5299 and X5302, previously described by Kouchinsky et al. (2015: figs. 10 and 12A, respectively) from the Bergeroniellus micmacciformis–Erbiella Zone, lower Botoman Stage (Cambrian Stage 3). They derive from sample AKT, collected by AK from Bed 21, Member 3, of the Perekhod Formation, Achchagyi-Kyyry-Taas section, middle Lena River, Siberia. The specimens were extracted from the sample with buffered 10% diluted acetic acid, and examined under a scanning electron microscope (SEM), Hitachi S4300 at NRM. The early mature part of SMNH X5299 and presumably later mature part of SMNH X5302 were further investigated with SRXTM at the TOMCAT beamline of the Swiss Light Source, Paul Scherrer Institute, Villigen, Switzerland. SEM image of an additional Oymurania gravestocki shell, SMNH X5294 (see also Kouchinsky et al. 2015: figs. 12C) from the same sample is figured herein.
Institutional abbreviations.—SMNH, Swedish Museum of Natural History, Stockholm, Sweden.
Other abbreviations.—SRXTM, synchrotron-radiation X-ray tomographic microscopy.
Results
Inner and outer shell layers can be distinguished in medium-sized Oymurania gravestocki shells (Kouchinsky et al. 2015: fig. 10; Fig. 1A1, A4; SOM 1: video 1, 2: Supplementary Online Material available at http://app.pan.pl/SOM/app62-Kouchinsky_Bengtson_SOM.pdf). The outer shell layer consists of spacious laminae oblique to the shell surface and connected by numerous rod-shaped elements representing the acrotretoid columnar microstructure (Fig. 1A1, A2, A4). The acrotretoid columns are pore-bearing and continuous through several lamellae (Kouchinsky et al. 2015; Fig. 1A4), thus belonging to the second type, according to Butler et al. (in press).
The inner shell layer is compact, laminated parallel to the inner shell surface and penetrated by a polygonal framework (Fig. 1A1, A4). The framework subdivides the inner shell layer into tightly packed continuous prismatic columns, 10–12 µm in cross-section, oriented perpendicularly to the inner shell surface and to the compact lamination. The polygonal bases of the columns adjacent to the acrotretoid columnar microstructure of the outer layer are exposed in broken sections (Fig. 1B), whereas the inner shell surface is covered with a polygonal relief (Fig. 1A4; Kouchinsky et al. 2015: fig. 11).
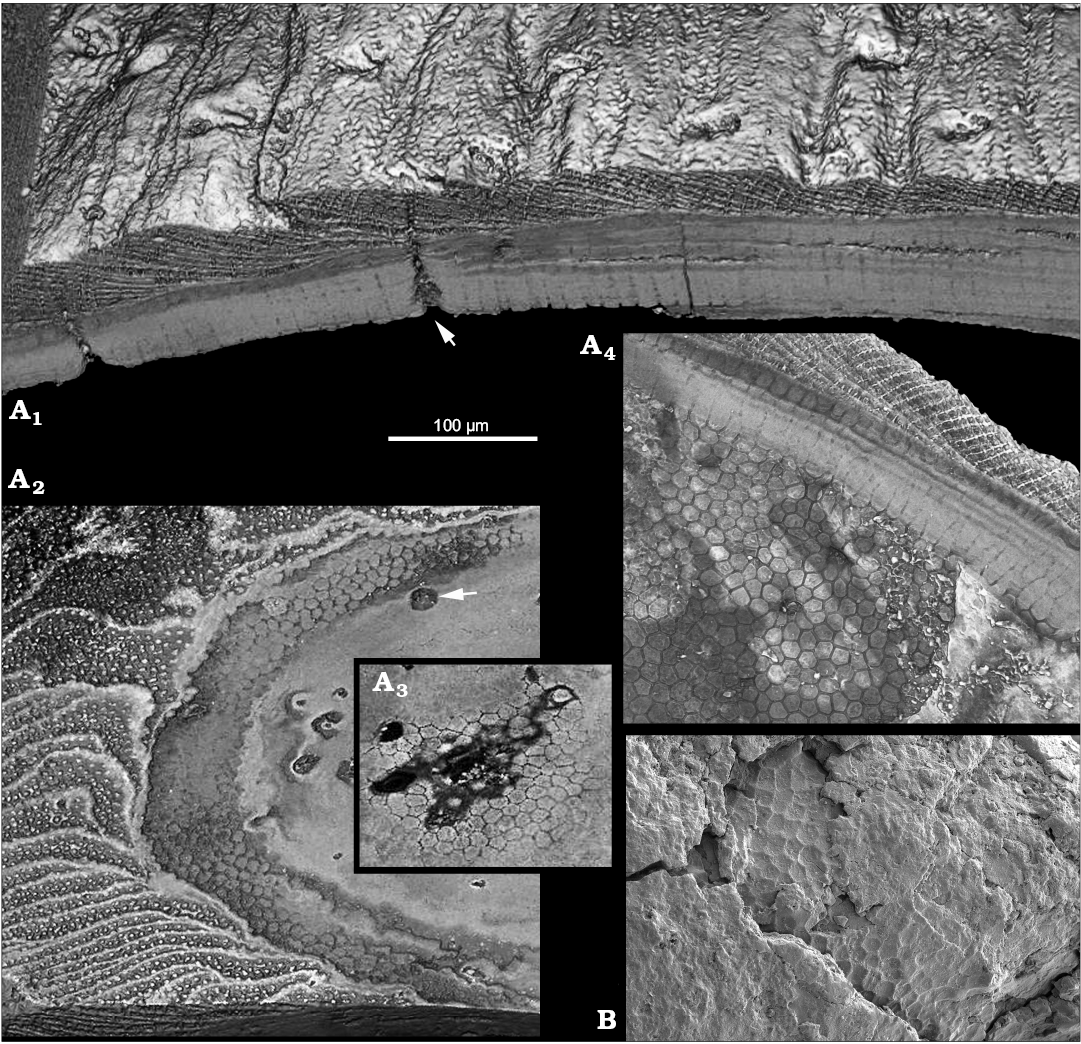
Fig. 1. Stem-group brachiopod Oymurania gravestocki Ushatinskaya in Kouchinsky et al., 2015 from the lower Cambrian Perekhod Formation, Achchagyi-Kyyry-Taas section, Siberia. A. SMNH X5299, SRXTM volumetric renderings through the proximal part of medium-sized shell; A1, virtual section through the shell in radial direction showing the inner compact layer consisting of perpendicularly oriented prismatic columns and the outer spacious layer featuring the acrotretoid columnar microstructure; note perpendicular Micrina–Setatella type canals (arrowed) penetrating the shell; A2, virtual tangential section through the lamellar acrotretoid microstructure (left in the image) and prismatic inner part (right in the image) of the shell; note cross-sections through the Micrina–Setatella type canals (arrowed); A3, fragment of a deeper virtual tangential section through the inner layer showing cross-sections of the prisms; A4, polygonal ends of the prismatic columns on the inner shell surface. B. SMNH X5294, SEM image showing polygonal texture in broken section near the outer surface of apical part of the shell (see Kouchinsky et al. 2015: fig. 12C1).
Continuous prismatic columns cannot be distinguished in the distal parts of larger shells (Fig. 2B; SOM 1: video 3, 4), where the wall consists of the acrotretoid columnar microstructure. The latter is represented by a series of spacious laminae connected by numerous rod-shaped acrotretoid columns alternating with more compact areas with less distinct columns. Lamination tends to follow a corrugated relief of the outer shell surface shaped by growth increments.
The shell is penetrated by perpendicular Micrina–Setatella type canals (Figs. 1A1, A2 arrowed, 2A, C). Canal openings scattered on the inner shell surface (Kouchinsky et al. 2015: figs. 10E, 11B) have single apertures. Laminae adjacent to the canals are deflected towards the outer shell surface (Fig. 2C arrowed). Successively deflected laminae appear in the SRXTM 3D renderings as stacked funnel-like structures through the wall (Fig. 2A, SOM 2: figs. 1–4). One or two openings of the perpendicular canals are grouped in the same depression or indentation on the shell exterior, distally with respect to each single opening of another type of canal, the tangential canal (see below and Kouchinsky et al. 2015: fig. 12A3, A5, A6, A8, B1).
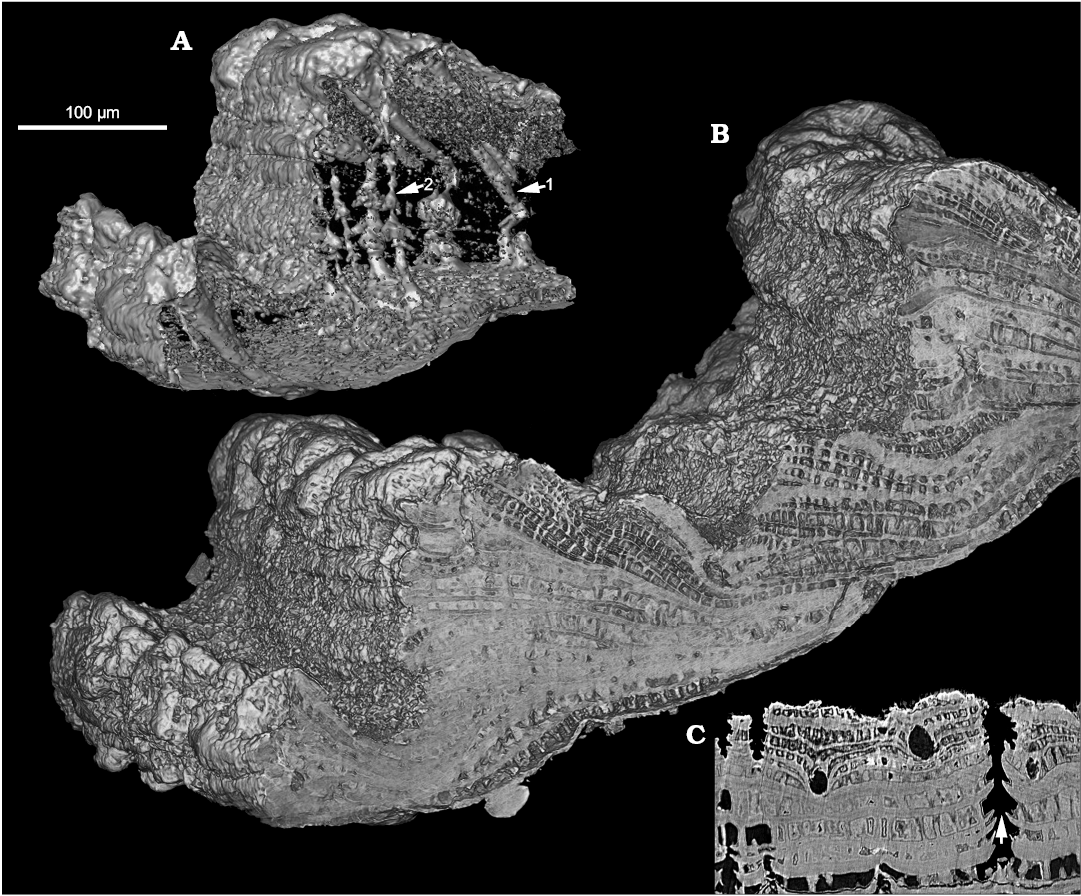
Fig. 2. Stem-group brachiopod Oymurania gravestocki Ushatinskaya in Kouchinsky et al., 2015 (SMNH X5302) from the lower Cambrian Perekhod Formation, Achchagyi-Kyyry-Taas section, Siberia. SRXTM volumetric renderings (A, B) and slice (C) through the distal part of an older shell. A. Intersecting systems of canals inside the wall fragment (B); note the tangential (setigerous) canals with closed inner terminations (arrow 1) and the perpendicular Micrina–Setatella type canals (arrow 2). C. Virtual slice through the same specimen showing rounded cross-sections of tangential (setigerous) canals and vertically oriented Micrina–Setatella type canals with laminae adjacent to the canals deflected towards the outer shell surface (arrowed).
Tangential canals open at the edges of growth increments and run in the radial direction through the acrotretoid microstructure, oblique to the surface of growth increments (Kouchinsky et al. 2015: figs. 10G, 12A3, B1, C2; SOM 1: video 3, 4, SOM 2: figs. 1–4). These canals are not found within the inner shell layer of tightly packed prismatic columns (Fig. 1A1, 2A, SOM 1: video 1). The X-ray tomography shows that the tangential canals have blunt terminations (Figs. 2A, SOM 2: figs. 1–4). Longitudinally striated structures consisting of < 1 µm thick parallel fibers (Fig. 3) are observed within two tangential canals. The fibers can be traced throughout the entire length of one of the canals (SOM 1: video 5–7) and show apparently hexagonal packing in cross-section of the bundle (Fig. 3B).
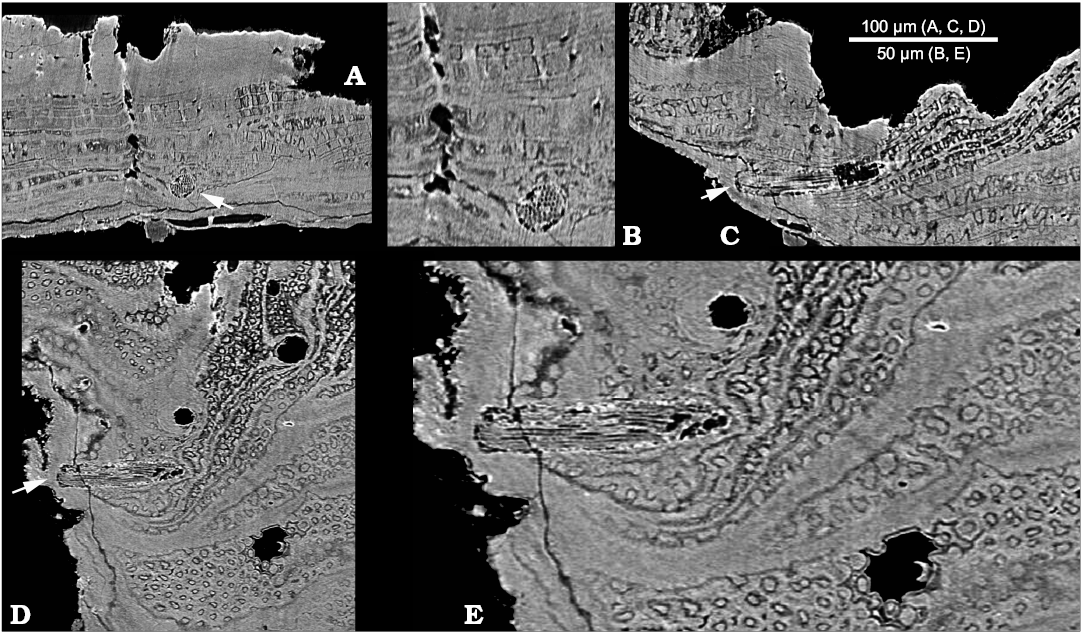
Fig. 3. Stem-group brachiopod Oymurania gravestocki Ushatinskaya in Kouchinsky et al., 2015 (SMNH X5302) from the lower Cambrian Perekhod Formation, Achchagyi-Kyyry-Taas section, Siberia. SRXTM slices showing a phosphatized seta inside a tangential canal (arrowed in A, C, and D). A. Section crossing perpendicularly to the tangential canal. B. Close-up of A showing cross-sections of filaments (microvilli). C. Longitudinal section in radial direction through the tangential canal. D. Longitudinal section oblique to the shell surface through the tangential canal. E. Close-up of D showing microvillar composition of the seta, lamellae consisting of acrotretoid columns perpendicularly penetrated by the Micrina–Setatella type canals.
Discussion
The two layers in the shell of Oymurania were first noticed in broken sections of the wall (e.g., Kouchinsky et al. 2015: fig. 10) and in specimens in which the outer layer consisting of the acrotretoid columnar microstructure had been abraded (Kouchinsky et al. 2015: figs. 11B, 12C1; Fig. 1B herein). The microstructure of a more massive inner layer was not, however, observed until the SRXTM supported the suggestion by Kouchinsky et al. (2015) on the original presence of spacious vs. more massive areas in the shell.
Microstructure of the massive shell layer.—The polygonal ornamentation on the inner shell surface (Kouchinsky et al. 2015: figs. 10E, 11A6, B; Fig. 1A4 herein) and in broken sections within the shell wall (Fig. 1B) represents opposite ends of prismatic columns of the inner shell layer (Fig. 1A). A polygonal framework penetrating the shell and interpreted to be originally organic is reported among paterinid brachiopods and derived tommotiids, such as Eccentrotheca Landing, Nowlan, and Fletcher, 1980; Paterimitra Laurie, 1986; and Micrina Laurie, 1986, but unknown from phylogenetically basal “camenellan” tommotiids and undoubted crown-group brachiopods (Balthasar et al. 2009; Topper et al. 2013; Skovsted et al. 2015). The polygonal framework can be either continuous through the wall or shallow, which may be largely depending on preservation (Balthasar et al. 2009; Topper et al. 2013). A probably homologous honeycomb microstructure is well expressed in the wall of a paterinid brachiopod, Salanygolina obliqua Ushatinskaya, 1987, from the lower Cambrian of Mongolia (Holmer et al. 2009). Salanygolina reveals slightly narrower stacked hexagonal prisms, 8–10 µm in diameter, perpendicularly oriented within 20–30 µm thick laminae of its supposedly originally organophosphatic shell (Ushatinskaya 1987; Holmer et al. 2009). It is not clear, however, whether the prismatic columns in Oymurania consist of stacked units similar to those found in Salanygolina. Stratification through the inner shell layer in Oymurania (Fig. 1A1, A4, SOM 1: video 1) suggests that this may be the case.
Perpendicular canals.—Canals penetrating the shell were originally known from Micrina and Setatella Skovsted et al. 2010 (Williams and Holmer 2002; Skovsted and Holmer 2003) and were subsequently termed Micrina–Setatella type canals (Kouchinsky et al. 2015). As in Micrina and Setatella, these canals penetrate the shell perpendicularly to its lamination and subparallel to the acrotretoid columns. One or two openings of the perpendicular canals in the same indentation on the outer shell surface suggest either bifurcation of a single canal or a pair of canals opening close to each other. The latter alternative is supported by the tomography images (Fig. 2A, SOM 2: figs. 1–4, SOM 1: video 1) showing closely spaced Micrina–Setatella type canals running parallel to each other throughout the wall. No bifurcating canals have been observed. The Micrina–Setatella type canals can be compared with punctae penetrating brachiopod shell, where they house mantle outgrowths (cecae) reaching the periostracum (Pérez-Huerta 2009). Pairs of canals penetrating the wall were found in sellate sclerites of Tannuolina pavlovi by Kouchinsky et al. (2010). Those canals coalesce at the edges of sclerites to open in a common aperture. The coalescing canals may be homologous to the Micrina–Setatella type canals, including those found in Oymurania.
Tangential canals and setae.—Tangential canals is a more appropriate term than “horizontal canals” used by Kouchinsky et al. (2015: figs. 10G, 12A3, B1, C2) and is adopted herein following Butler et al. (in press). These canals represent setal tubes known also from Tannuolina Fonin and Smirnova, 1967 (Skovsted et al. 2014; Butler et al. in press), Micrina (Williams and Holmer 2002), Mickwitzia Schmidt, 1888 and Setatella (Butler et al. 2015, in press). The difference in number of pore openings on the inner and outer shell surface of Oymurania (Kouchinsky et al. 2015) and the evidence from SRXTM show that the tangential canals do not open on the inner shell surface. Setigerous canals in Micrina are non-penetrative and non-branching (Murdock et al. 2014; Butler et al. in press), similar to the epipunctae described from Ordovician orthid brachiopods (Jin et al. 2007). Inner terminations of the setigerous canals in Oymurania are sealed within the outer shell layer in medium-sized shells or by succeeding shell increments within older shell growth stages characterized by the acrotretoid microstructure (Fig. 2A; SOM 1: video 3, 4, SOM 2: figs. 1–4). In addition to blunt inner terminations of the tangential canals, SRXTM of Oymurania revealed continuous fibrous structures interpreted herein as phosphatised setae (Fig. 3; SOM 1: video 5–7). The latter represent bundles of < 1 µm thick filaments interpreted herein, in accordance with previous studies (e.g., Jin et al. 2007; Butler et al. in press), as microvilli. The microvilli have the same diameter in the middle and outer portions of the setae (Fig. 3B).
Conclusions
Oymurania gravestocki is remarkable in its distinct differentiation into two shell layers, with the outer layer showing the acrotretoid columnar microstructure and the inner layer consisting of continuous prismatic columns separated by a shell-penetrating polygonal framework. Together with non-branching tangential setigerous canals and the shell-perpendicular Micrina–Setatella type canals, these plesiomorphic microstructural characters are variably expressed in different combinations in the brachiopod stem group, including tannuolinids and mickwitziids. Although without comparable morphological differentiation of the inner shell surface, Oymurania is generally most similar to mitral sclerites of a tannuolinid Micrina. Presence of the Micrina–Setatella type and setal canals is shared by tannuolinids, as well as some mickwitziids. Oymurania is also similar to mickwitziids and crow-group linguliform brachiopods in having the acrotretoid columnar microstructure of the wall, whereas its prismatic columnar microstructure is known from paterinid brachiopods and some tommotiids. A unique set of morphological and microstructural features of Oymurania confirms its position within the brachiopod stem group, probably among tannuolinids, and provides an intriguing new perspective on the evolution within the stem- to crown-group transition.
Acknowledgements.—Federica Marone (Paul Scherrer Institute, Villigen, Switzerland) and Veneta Belivanova (Swedish Museum of Natural History, Stockholm, Sweden) helped with the SRXTM work. We also thank the organisational committee of the XIIIth International Field Conference of the Cambrian Stage Subdivision Working Group for the excellent fieldtrip to the Lena River area in 2008, during which the material was collected. The authors are grateful to reviewers Duncan Murdock (University of Leicester, UK) and Uwe Balthasar (University of Glasgow, UK) for their important comments on the manuscript. The research was supported by the Swedish Research Council (Grant No. 2013-4290) and the Paul Scherrer Institute (20130185).
References
Balthasar, U., Skovsted, C.B., Holmer, L.E., and Brock, G.A. 2009. Homologous skeletal secretion in tommotiids and brachiopods. Geology 37: 1143–1146. Crossref
Budd, G.E. and Jensen, S. 2000. A critical reappraisal of the fossil record of the bilaterian phyla. Biological Reviews 75: 253–295. Crossref
Butler, A.D., Garwood, R.J., Streng, M., Lowe, T., and Holmer, L.E. (in press). X-ray microtomography of the tommotiid Micrina reveals cellular and ultrastructural preservation, confirming a tommotiid stem-linguliform affinity. The Royal Society Open Science.
Butler, A.D., Streng, M., Holmer, L.E., and Babcock, L.E. 2015. Exceptionally preserved Mickwitzia from the Indian Springs Lagerstätte (Cambrian Stage 3), Nevada. Journal of Paleontology 89: 933–955. Crossref
Fonin, V.D. and Smirnova, T.N. 1967. New group of problematic Early Cambrian organisms and methods of preparing them [in Russian]. Paleontologičeskij žurnal 1967 (2): 7–18.
Holmer, L.E., Pettersson Stolk, S., Skovsted, C.B., Balthasar, U., and Popov, L.E. 2009. The enigmatic Early Cambrian Salanygolina—a stem group of rhynchonelliform chileate brachiopods? Palaeontology 52: 1–10. Crossref
Jin, J., Zhan, R., Copper, P., and Caldwell, W.G.E. 2007. Epipunctae and phosphatized setae in Late Ordovician plaesiomyid brachiopods from Anticosti Island, eastern Canada. Journal of Paleontology 81: 666–683. Crossref
Kouchinsky, A., Bengtson, S., and Murdock, D. 2010. A new tannuolinid problematic from the lower Cambrian of the Sukharikha River in northern Siberia. Acta Palaeontologica Polonica 55: 321–331. Crossref
Kouchinsky, A., Bengtson, S., Runnegar, B., Skovsted, C., Steiner, M., and Vendrasco, M.J. 2012. Chronology of early Cambrian biomineralisation. Geological Magazine 149 (2): 221–251. Crossref
Kouchinsky, A., Holmer, L.E., Steiner, M., and Ushatinskaya, G.T. 2015. The new stem-group brachiopod Oymurania from the lower Cambrian of Siberia. Acta Palaeontologica Polonica 60: 963–980.
Landing, E., Nowlan, G.S., and Fletcher, T.P. 1980. A microfauna associated with early Cambrian trilobites of the Callavia zone, northern Antigonish Highlands, Nova Scotia. Canadian Journal of Earth Sciences 17: 400–418. Crossref
Laurie, J.R. 1986. Phosphatic fauna of the Early Cambrian Todd River Dolomite, Amadeus Basin, central Australia. Alcheringa 10: 431–454. Crossref
Murdock, D.J.E., Bengtson, S., Marone, F., Greenwood, J.M., and Donoghue, P.C.J. 2014. Evaluating scenarios for the evolutionary assembly of the brachiopod body plan. Evolution & Development 16: 13–24. Crossref
Pérez-Huerta, A., Cusack, M., McDonald, S., Marone, F., Stampanoni, M., and MacKay, S. 2009. Brachiopod punctae: a complexity in shell biomineralisation. Journal of Structural Biology 167: 62–67. Crossref
Schmidt, F. 1888: Über eine neuendeckte untercambrische Fauna in Estland. Académie Impériale des Sciences, St. Petersbourg, Mémoires (Series 7) 36: 1–27.
Skovsted, C.B. and Holmer, L.E. 2003. The Early Cambrian (Botomian) stem group brachiopod Mickwitzia from Northeast Greenland. Acta Palaeontologica Polonica 48: 1–20.
Skovsted, C.B., Clausen, S., Alvaro, J.J., and Ponlevé, D. 2014. Tommotiids from the early Cambrian (Series 2, Stage 3) of Morocco and the evolution of the tannuolinid scleritome and setigerous shell structures in stem group brachiopods. Palaeontology 57: 171–192. Crossref
Skovsted, C.B., Streng, M., Knight, I., and Holmer, L.E. 2010. Setatella significans, a new name for mickwitziid stem group brachiopods from the lower Cambrian of Greenland and Labrador. GFF 132: 117–122. Crossref
Skovsted, C.B., Ushatinskaya, G., Holmer, L.E., Popov, L.E., and Kouchinsky, A. 2015. Taxonomy, morphology, shell structure and early ontogeny of Pelmanotreta nom. nov. from the lower Cambrian of Siberia. GFF 137: 1–8. Crossref
Topper, T.P., Holmer, L.E., Skovsted, C.B., Brock, G.A., Balthasar, U., Larsson, C.M., Petterson Stolk, S., and Harper, D.A.T. 2013. The oldest brachiopods from the lower Cambrian of South Australia. Acta Palaeontologica Polonica 58: 93–109. Crossref
Ushatinskaya, G.T. [Ušatinskaâ, G.T.] 1987. Unusual inarticulate brachiopods from the Lower Cambrian of Mongolia [in Russian]. Paleontologičeskij žurnal 1987 (2): 62–68.
Williams, A. and Holmer, L.E. 2002. Shell structure and inferred growth, functions and affinities of the sclerites of the problematic Micrina. Palaeontology 45: 845–873.
Artem Kouchinsky [artem.kouchinsky@nrm.se] and Stefan Bengtson [stefan.bengtson@nrm.se], Department of Palaeobiology, Swedish Museum of Natural History, Box 50007, SE-104 05 Stockholm, Sweden.
Received 26 October 2016, accepted 5 December 2016, available online 31 January 2017.
Copyright © 2017 A. Kouchinsky and S. Bengtson. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Acta Palaeontol. Pol. 62 (1): 39–43, 2017
https://doi.org/10.4202/app.00320.2016