

Morphology and ontogeny of some Middle Ordovician gonambonitid brachiopods from Baltoscandia
ANNA A. MADISON
Madison, A.A. 2018. Morphology and ontogeny of some Middle Ordovician gonambonitid brachiopods from Baltoscandia. Acta Palaeontologica Polonica 63 (3): 585–594.
The Volkhovian–Kunda boundary deposits of the Leningrad Region contain two closely related genera, Antigonambonites and Anchigonites. The latter genus was previously known only by valve moulds; here its shell exterior and interior, and ontogeny are described in detail based on the collection of well-preserved disarticulated valves of Anchigonites conulus. Antigonambonites and Anchigonites share similar developmental and morphological features, and ecological strategies including the attachment by cementation by a “pedicle tube” and are thus possibly phylogenetically related. A new diagnosis for the genus Anchigonites and description of A. conulus are provided; other species of Anchigonites are briefly revised. The moulds of outer epithelial cells are described for the first time for the class Strophomenata.
Key words: Brachiopoda, Gonambonitidae, morphology, ontogeny, Ordovician, Russia.
Anna A. Madison [sunnyannmad@yahoo.com], Borissiak Paleontological Institute of Russian Academy of Sciences, 123, Profsoyuznaya ul., Moscow, 117997 Russia.
Received 17 April 2018, accepted 26 June 2018, available online 16 August 2018
Copyright © 2018 A.A. Madison. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
The Volkhovian–Kunda boundary deposits of the Leningrad Region contain two closely related brachiopod species, Antigonambonites planus (Pander, 1830) and Anchigonites conulus Öpik, 1939. Antigonambonites planus had been repeatedly described and depicted in many papers during the last two centuries and seems to be one of the best-documented billingsellide species in the world. However, A. conulus is very poorly known and represented in the literature only by shell moulds (Öpik 1939), latex casts of these moulds (Rubel and Wright 2000: 704, fig. 507: 2a–c), and one cross section (Vinn and Rubel 2000: figs. 2.3, 2.4). Anchigonites had been also reported from China, but the Chinese material is also represented by incomplete shell moulds, which probably belong to some other genus (see below). New material of well-preserved valves of A. conulus from the Leningrad Region allows careful description of its external and internal morphology; the developmental changes of ventral and dorsal valves exteriors and interiors of A. planus are also described here in detail in order to verify close relationship between these two genera.
Institutional abbreviations.—PIN, Borissiak Paleontological Institute of the Russian Academy of Sciences, Moscow, Russia; GIT, Tallinn University of Technology, Institute of Geology, Tallinn, Estonia.
Material and methods
The described brachiopods were obtained by washing the Middle Ordovician (lower part of Darriwilian, Volkhovian, and Kunda regional stages) clay from the section at the mouth of the Lynna River, western Leningrad Region (Figs. 1, 2). The Volkhovian and Kunda deposits there consist of interbedded clay and limestone and contain abundant and well preserved faunal remains (Dronov 1997; see also Madison 2013). Twelve ventral and 13 dorsal valves of A. planus and 10 ventral and 19 dorsal valves of A. conulus were studied using the SEM. All studied valves are disarticulated except for one complete shell of a juvenile A. planus. Each valve was photographed both from the interior and exterior. The collection also contains some other billingsellide shells with a well-developed and sealed “pedicle tube” that certainly do not belong to A. planus or A. conulus; possibly it is Gonambonites but additional study is needed in order to confirm their generic identity.

Fig. 1. Generalized stratigraphic column of the Middle Ordovician section from the western Leningrad Region (after Dronov 1997).
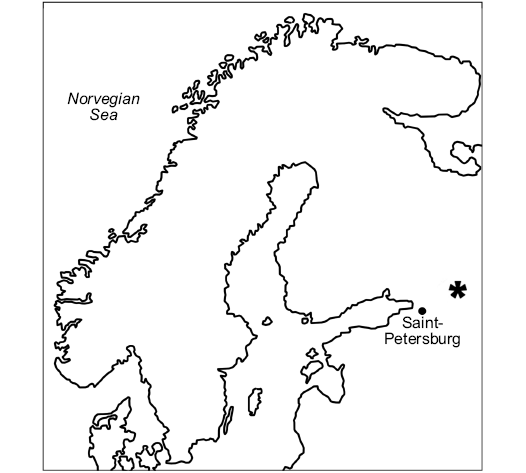
Fig. 2. Map of Northern Europe showing position of the studied section E 60.01053054594174, N 32.56286132296759 (asterisk) at the mouth of the Lynna River (Leningrad Region, Russia).
Systematic palaeontology
Phylum Brachiopoda Duméril, 1806
Subphylum Rhynchonelliformea Williams, Carlson, Brunton, Holmer, and Popov, 1996
Class Strophomenata Williams, Carlson, Brunton, Holmer, and Popov, 1996
Order Billingsellida Schuchert, 1893
Suborder Billingsellidina Schuchert, 1893
Family Gonambonitidae Schuchert and Cooper, 1931
Genus Anchigonites Öpik, 1939
Type species: Anchigonites conulus Öpik, 1939; Norway, Krekling; Darriwilian, Kunda regional stage (“Expansusschiefer”).
Emended diagnosis.—Shell small to medium sized, ventral valve high and conical, dorsal valve flattened or slightly convex. Deltidial plates form pedicle tube. Spondylium triplex; socket ridges almost subparallel to posterior margin; inner socket ridges and dorsal median septum present; dorsal muscle field radiate; posterior adductors considerably larger than anterior adductors.
Anchigonites conulus Öpik, 1939
Figs. 3, 4.
1939 Anchigonites conulus n. sp.; Öpik 1939: 136, pl. 3: 9–11.
2000 Anchigonites conulus Öpik, 1939; Rubel and Wright 2000: text-fig. 507: 2a–c.
Material.—Over hundred specimens (collection number PIN 4921) from the Leningrad Region, section at the mouth of the Lynna River, lower Kunda substage, Darriwilian, Middle Ordovician.
Description.—Shell small, up to 5 mm long and 9 mm wide; with high ventral valve and slightly convex dorsal valve; maximum width at posterior margin; ventral interarea apsacline, with strongly elongated pointed endings. Pedicle tube well developed and usually not overtopping ventral umbone. Ornament of radial ribs of two orders and concentric growth lines forming scars on ribs. Ribs of first order start from umbone; smaller second order ribs intercalate along whole shell length. Ribs reflect on valves inner surfaces as radial grooves, which start almost from posterior margin in ventral valve; grooves, corresponding to first order ribs are deeper and wider than grooves of second order ribs. Fold on ventral valve slightly flattened; sulcus on dorsal valve slightly concave; both fold and sulcus well delineated by ribs of first order. At anterior margin, there are up to seven ribs of second order between two ribs of first order in sulcus and fold and up to five ribs of second order between two ribs of first order on lateral sides, numbering 6–7 ribs within 2 mm of anterior margin. In ventral valve, lateral septa of spondylium triplex rudimentary and well pronounced only most posteriorly; anteriorly lateral septa look like low ridges on inner part of spondylium. In dorsal valve, chilidial plates discrete and fused to cardinal process; socket ridges widely diverging, almost parallel to posterior margin, and bear prominent outgrowths on their anterior regions. Cardinal process may bifurcate posteriorly. Adductor impressions radiate and stretch anteriorly to middle valve; anterior adductors at least twice as big as posterior ones. Median septum low, wide and gentle in juveniles and high and prominent in senile specimens, and may bear groove in middle. Peripheral rim well pronounced in adults. Tubercles on inner surface developed only in most anterior valve region within peripheral rim or on trail outside peripheral rim.
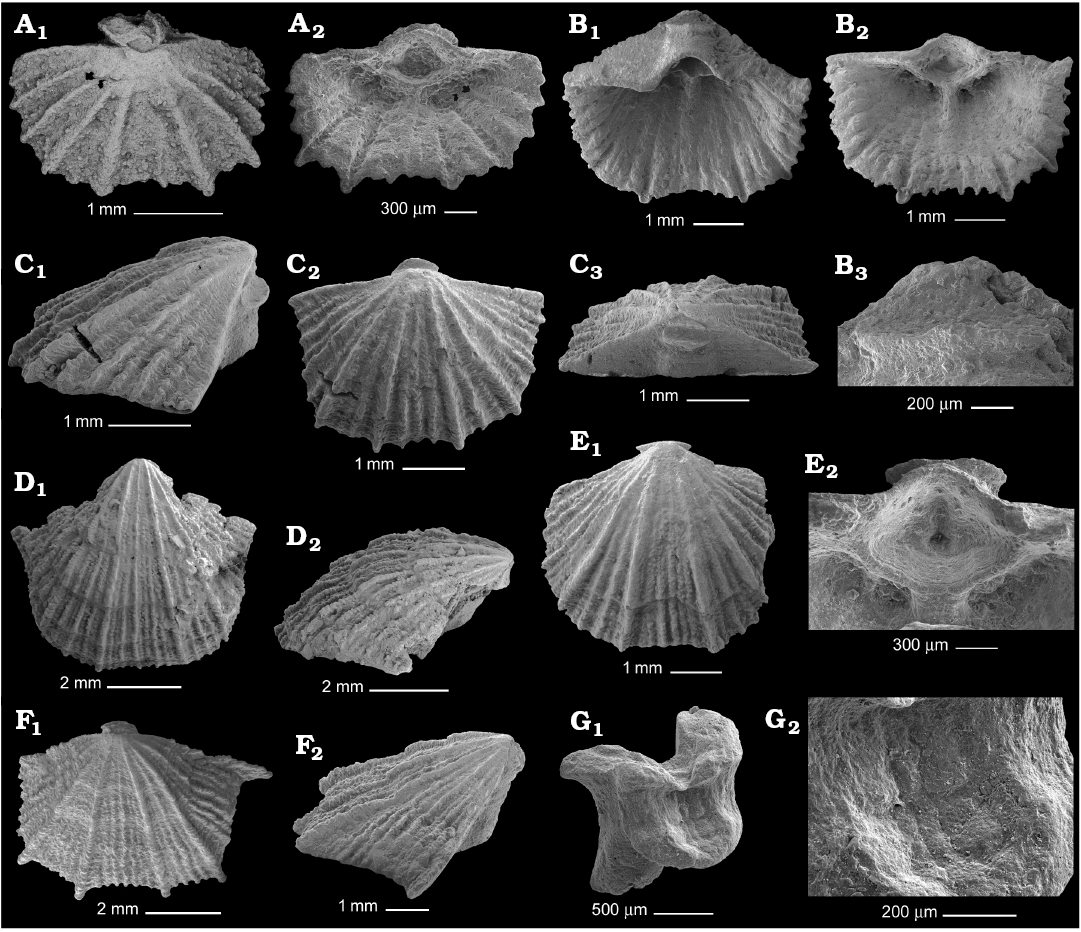
Fig. 3. Gonambonitid brachiopod Anchigonites conulus Öpik, 1939 from the Kunda regional stage, Darriwilian, Middle Ordovician of the Leningrad Region, Russia, ontogenetic series of ventral valves. A. PIN 4921/798, 3.5 mm wide valve in exterior (A1) and interior (A2) views. B. PIN 4921/891, 5 mm wide valve with sealed pedicle tube in interior view (B1), spondylium (B2), apical disc infilling the delthyrium apex (B3). C. PIN 4921/911, 4.2 mm wide valve in lateral (C1), ventral (C2), and posterior (C3) views. D. PIN 4921/908, 7 mm wide valve in general (D1) and lateral (D2) views. E. PIN 4921/807, 5.5 mm wide valve in general view (E1), spondylium with rudiment lateral septa (E2). F. PIN 4921/895, 8.3 mm wide valve in general (F1) and lateral (F2) views. G. PIN 4921/913, broken apical disc with imprint of the substrate, general view (G1), enlarged surface of the attachment region (G2).
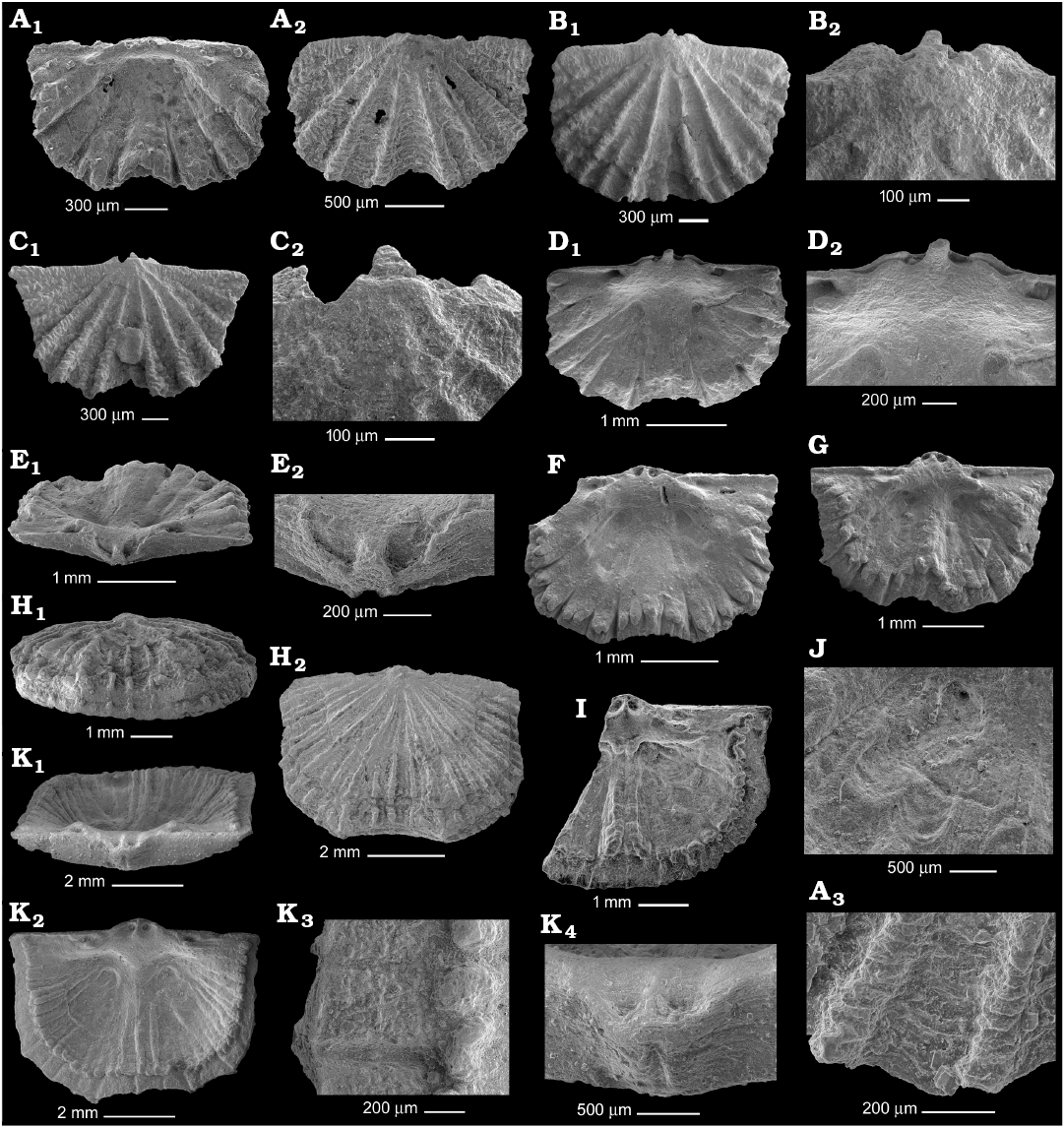
Fig. 4. Gonambonitid brachiopod Anchigonites conulus Öpik, 1939 from the Kunda regional stage, Darriwilian, Middle Ordovician of the Leningrad Region, Russia; ontogenetic series of dorsal valves. A. PIN 4921/528, 2.2 mm wide valve in interior (A1) and exterior (A2) views, fragment of ornamentation (A3). B. PIN 4921/689, 2.8 mm wide valve in general view (B1), umbone (B2). C. PIN 4921/688, 3 mm wide valve in general view (C1), umbone (C2). D. PIN 4921/897, 3 mm wide valve interior (D1), cardinalium (D2). E. PIN 4921/913, 3.3 mm wide valve in posterior view (E1), incomplete chilidium (E2). F. PIN 4921/912, interior of 3.5 mm wide valve. G. PIN 4921/917, interior of 4 mm wide valve. H. PIN 4921/526, 5.6 mm wide valve showing strong vertical growth in anterior (H1) and general (H2) views. I. PIN 4921/182, interior of 6 mm wide broken valve. J. PIN 4921/907, adductor scars of 7 mm wide valve. K. PIN 4921/906, 7.4 mm wide valve in posterior (K1) and interior (K2) views, fragment of inner surface near anterior margin (K3), incomplete chilidium (K4).
Remarks.—This species resembles A. planus in the general morphology and ontogeny. Antigonambonites Öpik, 1934 of the same family Gonambonitidae is restricted to the Volkhovian–Kunda deposits in East Baltic. Eight species had been distinguished within this genus; however, some of them are possibly the synonyms and the generic composition of Antigonambonites needs revising. The best known is A. planus, which occurs in the upper Volkhovian up to the boundary with the lower Kunda, where A. conulus appears. The morphology of A. planus is well known as both its juvenile, relatively large adult shells and disarticulated valves are usually well-preserved and often occur in the Volkhovian clays. In the present paper, only the ontogenetic changes of the exterior and interior of its shells are discussed and hence only juvenile valves of A. planus are depicted; for the adults see Rubel and Wright (2000: fig. 508: 2).
The most peculiar feature of the shell of A. planus is its attachment structure, composed of shell substance and reflecting pedicle shape (Madison 2004; Popov et al. 2007; Bassett et al. 2008a, b). Here, it is called the “pedicle tube”. This tube probably surrounded the pedicle and has a widening at the apex called an “apical disc”, which was used for cementation to the substrate. The margins of apical disc may be strongly extended and variously curved corresponding to the substrate relief. The youngest specimens have open pedicle tube (Fig. 5A, C, F, G1, I); this pedicle opening may be sealed in 3 mm wide specimens (Fig. 5B) and open in 4.5 mm wide ones (Fig. 5I) but is sealed in all larger specimens (Fig. 5H3). The apical disc is about 700 µm in diameter in all measured juveniles, both with open and sealed pedicle openings. The apical disc of one measured adult, about 3 cm wide, is about 1 mm in diameter. The ventral and dorsal margins (“lips”) of the apical disc are variously shaped. The dorsal margin may be extremely long and curved that is possibly connected with the type of substrate (Fig. 5D). The ventral lip often replicates and almost infills the apical outline of delthyrium (Fig. 5E, F); therefore the apical disc of A. planus had been formed after the development of ventral interarea. The sealing of the pedicle opening occurred not peripherally, which would have been ideal, but the remaining opening is often slit-like and located closer to the umbone (Fig. 5F).
A. planus has a spondylium supported by three septa. Our collection includes a broken specimen, about 3 mm wide (Fig. 5A), which is probably A. planus as its shape and external ornamentation do not differ from those of other A. planus valves and its broken, open pedicle tube with thickened ventral margin strongly resembles that of A. planus though it seems to be slightly, more slender. The U-shaped spondylium simplex of this specimen is a free plate similar to those of juvenile Apomatella Schuchert and Cooper, 1931 (Vinn and Rubel 2000) or Palaeoneumania (Harper in Bruton and Harper, 1981) (see Egerquist 2003) and lacks any supporting septa. No dental plates are developed and the free plate joins the delthyrial margins at some distance from the teeth that support Vinn and Rubel (2000), who suggested that “the dental plates (in evolutionary terms) are unrelated to the spondylium both phylogenetically and functionally”.
The characteristic feature of adult A. planus is a resupinate shell; however, small specimens have a conical ventral valve and flattened or even slightly concave dorsal valve that are typical for billingsellides; the resupination develops later during ontogeny. The ears of juveniles are acute. The ribs are uniform and increase by intercalation.
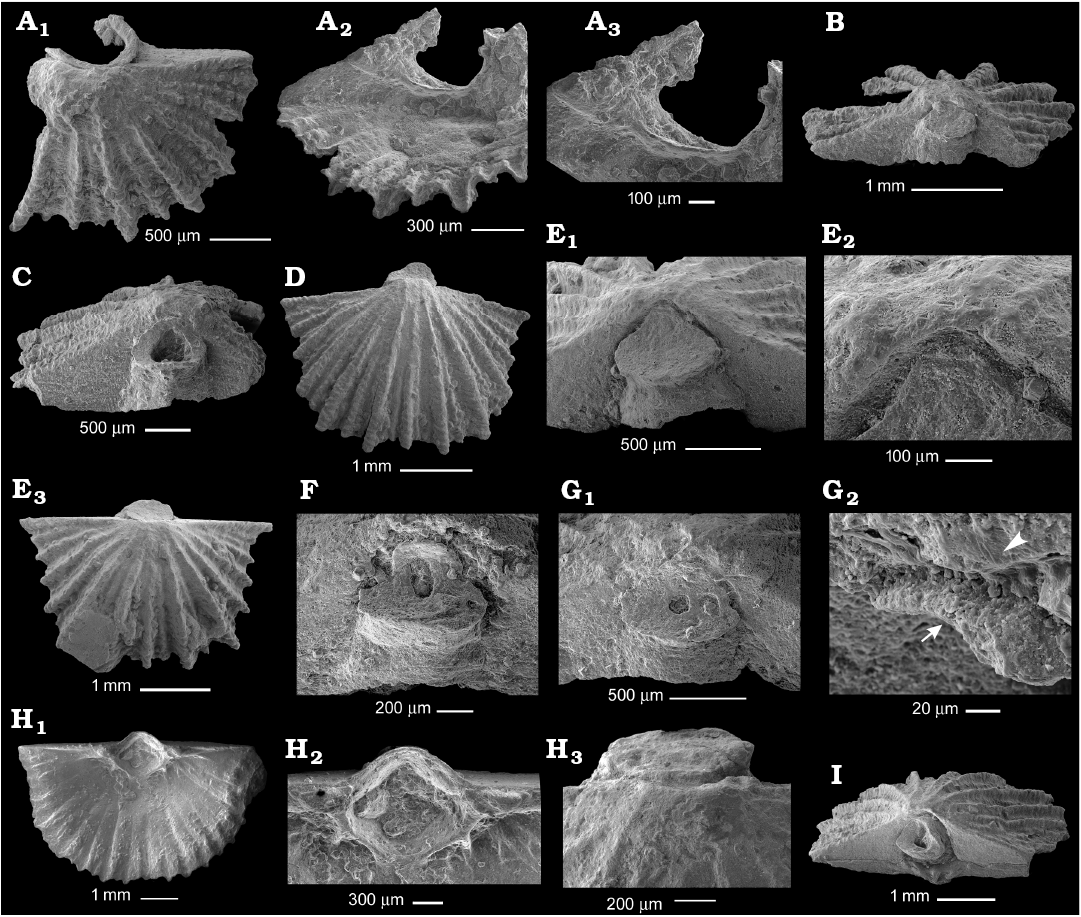
Fig. 5. Gonambonitid brachiopod Antigonambonites planus (Pander, 1830) from the Volkhovian regional stage, Darriwilian, Middle Ordovician of the Leningrad Region, Russia; ontogenetic series of ventral valves. A. PIN 4921/785, spondylium of 3 mm wide valve in exterior (A1), interior (A2), and inner (A3) views. B. PIN 4921/884, posterior view of 3 mm wide valve with sealed pedicle tube. C. PIN 4921/879, posterior view of 3.4 mm wide valve with open pedicle tube. D. PIN 4921/880, exterior view of 3.5 mm wide valve with sealed pedicle tube and ribs reaching the posterior margin. E. PIN 4921/882, 3.8 mm wide valve in posterior view (E1), upper margin of pedicle tube infilling the delthyrium apex (E2), general exterior view (E3). F. PIN 4921/885, posterior view of 4 mm wide valve with elongated remnant of pedicle opening. G. PIN 4921/885, posterior view of 4 mm wide valve with circular remnant of pedicle opening (G1), enlarged upper margin of apical disc (arrow) near ventral umbone (arrowhead) (G2). H. PIN 4921/805, interior of 8 mm wide valve (H1), spondylium supported by three septa (H2), for cell molds see Fig. 8A, exterior of ventral umbone with sealed apical disc and ribs reaching the umbone posterior margin (H3). I. PIN 4921/883, 4.4 mm wide complete shell in posterior view.
Another peculiar feature of A. planus is the absence of the first-formed shell on both valves. Most of the rhynchonelliformean brachiopods have smooth or variously embossed regions on the umbones, which correspond in size to the larval mantle, may reflect some morphological features of the larva, lack adult ornamentation and are separated from the rest of the shell by the halo rings. However, A. planus lacks such region on both valves. The ribs on its ventral valve start immediately from the posterior margin and there are not any separated regions on the ventral umbone (Fig. 5D, E, H3). The umbone of the dorsal valve bears about 400–500 µm wide region well bordered with discontinual rings, but this region was unlikely to have been formed by the larval mantle as the best-preserved juvenile specimens have 6 or 8 adult ribs, which obviously cross these discontinual rings and seem to reach the posterior margin (Fig. 6A2, C, D2).
In the dorsal valve of A. planus, there is a simple cardinal process, radiate elongated posterior and anterior adductor imprints, which are usually well-pronounced in juveniles (Fig. 6F2), and numerous septa marking the vascular impressions in adults. The interior of juvenile dorsal valves is generally the same except for the undeveloped chilidial platform and the absence of vascular impressions, raised peripheral rim along the anterior margin, and tubercles in the anterior part of the valve (that possibly means that the pseudopunctae are not developed at these stages) (Fig. 6B, D3, F1). The muscle impressions are divided by a wide gentle elevation; the muscle field is deeply impressed.

Fig. 6. Gonambonitid brachiopod Antigonambonites planus (Pander, 1830) from the Volkhovian regional stage, Darriwilian, Middle Ordovician of the Leningrad Region, Russia; ontogenetic series of dorsal valves. A. PIN 4921/792, exterior of 2 mm wide valve (A1), ribs crossing the halo rings (A2). B. PIN 4921/905, interior of 2.7 mm wide valve. C. PIN 4921/793, umbone of 3.2 mm wide valve showing ribs crossing the halo rings. D. PIN 4921/902, exterior of 3.5 mm wide valve (D1), dorsal umbone (D2), valve interior (D3). E. PIN 4921/899, 4 mm wide valve in exterior view. F. PIN 4921/903, interior of broken 5.5 mm wide valve (F1), dorsal adductor scars (F2).
The genus Anchigonites, its type species A. conulus, and A. heintzi were established by A. Öpik in 1939 from the Kunda stage (“Expansusschiefer”) of the outskirts of Oslo (Norway) by the moulds of ventral and dorsal valves; the latex casts of his specimens of A. conulus were published by Rubel and Wright (2000: fig. 507: 2). Later, Vinn and Rubel (2000) published cross-sections through the spondylium of Anchigonites supported by three well-pronounced septa reaching the valve floor. Thus, until recently some morphological features of this genus remain controversial due to insufficient material and none of the papers listed above contain a comprehensive diagnosis of the genus; new diagnosis of the genus Anchigonites and description of A. conulus are given here. Our collection contains well-preserved disarticulated valves of A. conulus at different ontogenetic stages from the Kunda clayey deposits that making possible a thorough study of shell exterior and interior of this species.
The shell of A. conulus is small; 6 mm wide specimens stop growing in width and length and only the shell thickness increases; thus, the anterior margins of largest specimens show strong vertical growth (Fig. 4H1). The ornamentation consists of ribs of two orders; the larger ribs start immediately from the umbone and smaller ribs of the second order start to intercalate at about 1 mm from the umbone. The smallest studied specimen almost lacks second-order ribs (Fig. 3A). The ventral valve is high and conical (Fig. 3C1, D2, F2); the ventral interarea of the smallest specimens is normal to the commissural plane; in the largest specimens, it becomes apsacline.
The pedicle tube on the ventral valve seems to be developed similarly to that of A. planus. The earliest developmental stage in our collection represents almost connected deltidial plates, which had already formed a wide lip around sealing pedicle opening (the cited specimen came from the lowest beds of the middle Kunda; in Madison [2004: pl. 7: 1] it was incorrectly noted as the upper Volkhovian). All other specimens except for Fig. 3A have a closed pedicle tube with a widened apical disc. The early closure of the pedicle tube is probably connected with the small size of these brachiopods. Unlike A. planus, the pedicle tube usually is short and does not overlap the umbone, but its apical disc is also variously curved corresponding to the relief of the substrate (Fig. 3A1, B3, C). One broken apical disc bears a distinct imprint of the substrate, to which it was attached (Fig. 3G). Ten 3–8 mm wide specimens were measured in order to find the correlation between the shell size and the diameter of apical disc but no such relationship was found. The discs of A. conulus are from 0.7 to 1 mm wide; their width does not depend on the shell size that indicates that growth of the pedicle tube ceased immediately after its formation.
In the ventral valve, the septa supporting the spondylium triplex differ from those of A. planus. The median septum is considerably thinner and higher than in A. planus and slightly projects anteriorly of spondylium (Fig. 3E2). The lateral septa of spondylium triplex reach the valve floor only in the umbone apex and are rudimentary anteriorly of it (Fig. 3B2, E1). The spondylium muscle field is clearly divided into three longitudinal regions: central wider adductor muscle scar and two narrower diductor attachments. The inner surface of deltidium bears a longitudinal scar at the place of the fusion of deltidial plates (Fig. 3E2).
The juvenile dorsal valve is flattened; adult dorsal valve is slightly convex (Fig. 4E1, K1). About 350 µm wide region on the umbone is separated from the rest of the valve by a few prominent concentric rings; the shell surface within this region lacks frequent growth lines, which cover the rest of the valve. However, as well as in A. planus, the better-preserved specimens clearly show that the ribs of the first order cross these concentric rings and reach the posterior margin (Fig. 4C2).
The dorsal valve interior is generally the same as in A. planus except for the lower chilidial platform and absence of ridges on vascular system. The sockets are internally bordered by curved ridges connecting the socket ridges to the posterior margin and separating the sockets from the notothyrial platform (Fig. 4D2). These ridges are interpreted here as inner socket ridges. The inner socket ridges are also developed in A. planus but are less-well pronounced. At the early stages, the muscle field is divided with wide gentle elevation, which transforms into the prominent median septum in adults (Fig. 4D1, F, I). The platform anteriorly of the muscle impressions bears thick and high ridges divided by very narrow grooves, which seem to correspond to the rib interspaces on the outer surface. The platform of adults is bordered by a high peripheral rim, which appears in 3–4 mm wide specimens (Fig. 4G, I, K2); the trail is short and densely covered with tubercles that emphasizes the pseudopunctate shell structure (Fig. 4K3). However, the study of the shell structure was unsuccessful as the shell substance of all studied specimens of A. conulus is strongly altered by diagenesis. The orthids from the same beds show well preserved fibers that possibly indicate the laminar shell structure of A. conulus similar to the laminar shell of A. planus (Madison 2017).
Another species of Anchigonites, A. heintzi is poorly known. It was distinguished by Öpik (1939: pl. 4: 1, 2) based on two internal moulds of dorsal and ventral valves; however, these moulds are incompletely preserved and the ventral umbone is absent. The latex casts prepared by Madis Rubel (Fig. 7) show a well-developed spondylium, possible lateral septa supporting the spondylium, dorsal muscle field divided by gentle elevation, socket ridges subparallel to the posterior margin, and cardinal process on the well-developed notothyrial platform with complete chilidium. These specimens are rather large, almost twice as big as A. conulus, the pattern of their dorsal muscle field and type of pedicle opening are uncertain; probably the species should be considered as a nomen dubium until new and better preserved material will be available.

Fig. 7. Latex casts of the gonambonitid brachiopod Anchigonites heintzi Öpik, 1939 from the Kunda regional stage, Middle Ordovician of Krekling, Norway. A. GIT 801-3, ventral valve. B. GIT 801-7, dorsal valve. Casts prepared by Madis Rubel (University of Tartu, Geology Department, Estonia).
One more controversial and poorly-known species of Anchigonites is A. minor Xu and Liu, 1984 from the Floian of South China. It was distinguished by a few inner and outer valve impressions showing uniformly ribbed shell, strong septa in the ventral and dorsal valves, and radiate scars of dorsal adductors. However, the ventral umbones of this species are unknown and its stratigraphic range is significantly older than that of A. conulus; therefore, the generic assignment of A. minor needs revising.
Cell imprints: The moulds of outer epithelial cells are well known in fossil brachiopods with calcium phosphate shells and are exceptionally rare in the brachiopods with calcium carbonate shells (Zhang et al. 2016). The cell imprints of strophomenate brachiopods are described here for the first time. The dorsal valves of A. planus and A. conulus bear variously preserved moulds of outer epithelial cells, which are allocated to the position of muscle attachment and vascular grooves near the anterior margin. Among the ventral valves, one juvenile 6 mm wide ventral valve of A. planus bears indistinct cell imprints on the bifurcating vascular ridges near its posterior region (Fig. 8A). The adductor imprints of A. planus also often bear variously-preserved cell imprints, but the best-preserved imprints are located on the deeply impressed adductor scars of A. conulus (Fig. 8B). They are indistinctly tetragonal or pentagonal in outline and usually 7–8 µm wide and 9–11 µm long. The best-preserved partitions between the moulds are up to 2.5 µm high. The cell molds are located also on the outer high vertical wall bounding the posterior adductor imprint that indicates that it was also used for muscle attachment (Fig. 8C). The lining of the coelomic canals in Recent brachiopods may contain epithelium-muscle cells (Tatyana Kuzmina, personal communication 2018) that may explain the presence of cell moulds in the vascular grooves of gonambonitids.
Stratigraphic and geographic range.—Middle Ordovician, Kunda stage; Baltoscandia.

Fig. 8. Moulds of outer epithelial cells in gonambonitid brachiopods from the Volkhovian (A) and Kunda (B, C) regional stages, Darriwilian, Middle Ordovician of the Leningrad Region, Russia. A. Antigonambonites planus (Pander, 1830), PIN 4921/805, ventral valve with ridges of mantle canals system bearing cell molds (fragment of Fig. 5H2). B, C. Anchigonites conulus Öpik, 1939, dorsal adductor scars. B. PIN 4921/912, dorsal muscle field (B1), cell moulds covering adductor scars (B2–B4). C. PIN 4921/901, posterior wall of deeply impressed adductor scar with cell moulds.
Discussion
Structure of pedicle tube.—The development of the attachment structures of A. planus has been very differently interpreted (Madison 2004; Popov et al. 2007; Bassett et al. 2008a, b). Adult A. planus was cemented to the substrate by the shell substance, which reflects the pedicle in shape. It is referred here as a “pedicle tube” as the pedicle sheaths of strophomenids probably developed in a different way (Madison and Kuzmina 2017) and these structures are not considered in the present paper as homologues though a number of authors have inferred their homology (Bassett et al. 2008b; Bassett and Popov 2017). In the present paper, the scenario of the billingsellide pedicle tube formed by fused overgrown deltidial plates is supported (Madison 2004; for other interpretations see Popov et al. 2007). The developmental stages of the covering structures of the Antigonambonites pedicle opening are reconstructed here as follows. The first-formed pedicle opening was a delthyrium; the deltidial plates appeared quite early in the ontogeny. This earliest stage is absent in the collection; the following stages with discrete and almost fused deltidial plates are well shown in Popov et al. 2007: figs. 3A, M, 4D, F. Then the deltidial plates became completely fused and the carbonate shell was exuded as a continuous structure (Fig. 5C, I); this structure probably is homologous to the henidium of terebratulids as it completely lacks a line of junction on the outer side (in Madison 2004 it was called a symphytium, however, it has a junction suture on the inner side in A. conulus, Fig. 3E2). The henidium continued growing as a single plate both in ventral and dorsal directions, completely enclosing the delthyrium, and then grew ventrally to form a tube around the pedicle. This tube had a widening at the apex (referred to as “apical disc” here) that possibly started cementation immediately after formation (Fig. 5A1, C). For some time, the brachiopod attached both by the pedicle and widened margins of pedicle tube, but then the tube apex was gradually sealed with a cementing pad and the pedicle probably atrophied (Fig. 5D–G). Considering the rim size and shape it may be assumed that the brachiopod started cementation with this rim while the pedicle was still functioning. The summarized developmental stages of the pedicle tube of A. planus seem to be as follows: discrete deltidial plates, deltidial plates fused into a completely open at the apex tube, tube half-closed at the apex, and completely closed tube with wide apical disc.
Phylogenetic implications.—The comparison of A. planus and A. conulus shows that they are closely similar in their gross morphology, especially in the presence of pedicle tube, spondylium triplex, pattern of dorsal muscle field, and generally same type of cardinalium. The differences are in the shell shape, type of exterior ornamentation, less-well developed lateral septa of the spondylium and longer median septum in the adult dorsal valve of A. conulus, and variously pronounced ridges of vascular system in A. planus. However, their juvenile dorsal interiors are very similar (compare Figs. 4A1 and 6B). A. planus is unknown higher the Volkhovian–Kunda boundary, where A. conulus appears; probably Anchigonites originated from Antigonambonites and is its direct descendant.
Ontogenetic implications.—The dorsal umbonal apexes of A. planus and A. conulus bear well-pronounced wide first-formed regions, which lack growth lines and differ from the rest of the valve in the plane of growth. However, the better-preserved specimens of both species clearly show that the ribs of the first order cross this umbonal region and reach the posterior margin. As this region bears adult ornamentation, it cannot correspond to larval mantle activity and does not support the presence of a larval dorsal mantle lobe during the planktonic stage. These ribs also cannot correspond to the setal sacks of the larva as they start immediately from the posterior margin of the valve, while the larval setal sacks are located peripherally along the mantle margins. The ventral valve also lacks a region presumably formed by the larval mantle; thus the larva of these brachiopods completely lacked mantle lobes during the planktonic stage; possibly, its dorsal mantle lobe appeared earlier in the ontogeny than the ventral lobe but both of them developed after settlement.
It may be assumed that all billingsellide brachiopods, whose ventral and dorsal valves lack any first-formed areas, shared the same type of larva, which lacked rudimentary mantle lobes during the planktonic stage. This larva possibly had a well-developed pedicle lobe (as the smallest specimens show closing the delthyrium) and therefore it did not fold on the ventral side, as this is impossible for larvae with pedicle lobes. However, the presence of discontinuity rings on the dorsal valve needs further explanation.
Conclusions
Antigonambonites and Anchigonites share the same type of attachment structures, in the form of a cementing pedicle tube and formed by accreted deltidial plates.
Anchigonites possibly originated from Antigonambonites as they are very similar in morphology and occur sequentially.
The gonambonitid larva possibly lacked mantle lobes during the planktonic stage and possessed a pedicle lobe; thus its metamorphosis excludes folding on the ventral side after settlement.
Acknowledgements
The author is sincerely grateful to Ursula Toom for the help with collections of the Department of Geology, Tallinn University of Technology. The referees David Harper (Durham University, UK), Jisuo Jin (Western University, London, Canada), and Ren-Bin Zhan (Nanjing Institute of Geology and Paleontology, CAS, PRC) provided helpful comments that greatly improved the manuscript. This paper was supported by the Russian Foundation for Basic Research, project no. 18-04-01046 A.
References
Bassett, M.G. and Popov, L.E. 2017. Earliest ontogeny of the Silurian orthotetide brachiopod Coolinia and its significance for interpreting strophomenate phylogeny. Lethaia 50: 504–510. Crossref
Bassett, M.G., Popov, L.E., and Egerquist, E. 2008a. Early ontogeny of some Ordovician–Silurian strophomenate brachiopods: Significance for interpreting evolutionary relationships within early Rhynchonelliformea. Fossils and Strata 54: 13–20.
Bassett, M.G., Popov, L.E., and Egerquist, E. 2008b. Pedicle preservation in a Silurian rhynchonelliformean brachiopod from Herefordshire, England: soft-tissue or an artefact of interpretation? Earth and Environmental Science Transactions of the Royal Society of Edinburgh 98: 303–308. Crossref
Bruton, D.L. and Harper, D.A.T. 1981. Brachiopods and trilobites of the early Ordovician serpentine Otta Conglomerate, south central Norway. Norsk Geologisk Tidsskrift 61: 153–181.
Dronov, A.V. (ed.) 1997. Russian and International Bryozoan Conference “Bryozoa of the World”, A Field Excursion Guide. St. Petersburg, Terra Nostra 1997: 56.
Duméril, A.M.C. 1806. Zoologie analytique, ou méthode naturelle de classification des animaux, rendue plus facile à l’aide de tableaux synoptiques. xxiii + 344 pp. Allais, Paris.
Egerquist, E. 2003. New brachiopods from the Lower–Middle Ordovician (Billingen–Volkhov stages) of the East Baltic. Acta Palaeontologica Polonica 48: 31–38.
Madison, A.A. 2004. Delthyrial coverings of Middle Ordovician clitambonitidines (Clitambonitidina, Brachiopoda) and their significance for attachment to the substrate. Paleontological Journal 38: 627–634.
Madison, A.A. 2013. New brachiopod subfamily Anechophragmiinae (Strophomenida) from the Ordovician of the Leningrad Region. Paleontological Journal 47: 24–37. Crossref
Madison, A.A. 2017. Laminar shell structure of Antigonambonites planus (Pander, 1830). (Brachiopoda, Billingsellida). Estonian Journal of Earth Sciences 66: 183–187. Crossref
Madison, A.A. and Kuzmina, T. 2017. Possible structure of larva of the earliest strophomenids (Bachiopoda) In: E.N. Temereva (ed.), International Congress on Invertebrate Morphology, ICIM-4, Moscow, 2017, Abstracts, 18. Pero, Moscow.
Öpik, A. 1934. Über Klitamboniten. Acta et Commentationes Universitatis Tartuensis (series A) 26 (5): 1–239.
Öpik, A. 1939. Brachiopoden und Ostrakoden aus dem Expansusschiefer Norwegens. Norsk Geologisk Tidsskrift 19: 117–142.
Pander, C.H. 1830. Beiträge zur Geognosie des Russischen Reiches. 165 pp. Gedruckt bei K. Kray, St.-Petersburg.
Popov, L.E., Egerquist, E., and Holmer, L.E. 2007. Earliest ontogeny of Middle Ordovician rhynchonelliform brachiopods (Clitambonitoidea and Polytoechioidea): implications for brachiopod phylogeny. Lethaia 40: 85–96. Crossref
Rubel, M. and Wright, A. 2000. Suborder Clitambonitidina. In: R.L. Kaesler (ed.), Treatise on Invertebrate Paleontology, Pt. H: Brachiopoda Revised. Vol. 3, 692−707. The Geological Society of America, Boulder and The University of Kansas, Lawrence.
Schuchert, C. 1893. Classification of the Brachiopoda. American Geologist 11: 141–167.
Schuchert, C. and Cooper, G.A. 1931. Synopsis of the brachiopod genera of the suborders Orthoidea and Pentameroidea, with notes on the Telotremata. American Journal of Science (Series 5) 22: 241–255.
Vinn, O. and Rubel, M. 2000. The spondylium and related structures in the Clitambonitidine brachiopods. Journal of Paleontology 74: 439–443. Crossref
Williams, A., Carlson, S.J., Brunton, C.H.C., Holmer, L.E., and Popov, L.E. 1996. A supra-ordinal classification of the Brachiopoda. Philosophical Transactions of the Royal Society of London B 351: 1171–1193. Crossref
Wright, A.D. and Rubel, M. 1996. A review of the morphological features affecting the classification of clitambonitidine brachiopods. Palaeontology 39: 53–75.
Xu, H.K. and Liu, D.Y. 1984. Late Lower Ordovician brachiopods of southwestern China [in Chinese with English summary]. Bulletin of Nanjing Institute of Geology and Palaeontology 8: 147–235.
Zhang, Z.L., Zhang, Z.F., and Wang, H. 2016. Epithelial cell moulds preserved in the earliest acrotretid brachiopods from the Cambrian (Series 2) of the Three Gorges area, China. GFF 138: 455–466. Crossref
Acta Palaeontol. Pol. 63 (3): 585–594, 2018
https://doi.org/10.4202/app.00488.2018