


Evolution of postcranial skeleton in worm lizards inferred from its status in the Cretaceous stem-amphisbaenian Slavoia darevskii
MATEUSZ TAŁANDA
Tałanda, M. 2017. Evolution of postcranial skeleton in worm lizards inferred from its status in the Cretaceous stem-amphisbaenian Slavoia darevskii. Acta Palaeontologica Polonica 62 (1): 9–23.
Amphisbaenians are one of the most derived fossorial groups among Squamata. Studies of their evolution were hampered by their specialized limbless morphology and unrecognized early fossil record. This study presents a detailed description of the postcranial anatomy of the oldest known stem-amphisbaenian Slavoia darevskii. The skeleton shows an incipient adaptation to the fossorial mode of life, expressed in the early stages of limb reduction and elongation of the trunk, typical aspects of modern worm lizards. The forelimbs show a hyperphalangy of the first digit. They were probably strengthened this way to support the head in burrowing. Such an anatomy of the most ancient amphisbaenians implies that the forelimbs were lost multiple times in the amphisbaenians to be retained only in the Bipedidae. The hindlimb reduction was initiated early (before the split into modern families) but may have been completed independently. Despite these changes, S. darevskii still had the plesiomorphic 26 presacral vertebrae but the neck region was shorter, as shown by the ribs morphology and position.
Key words: Squamata, Amphisbaenia, limb loss, hyperphalangy, parallel evolution, Cretaceous.
Mateusz Tałanda [m.talanda@biol.uw.edu.pl], Department of Paleobiology and Evolution, Faculty of Biology, Biological and Chemical Research Centre, University of Warsaw, ul. Żwirki i Wigury 101, 02-089 Warszawa, Poland.
Received 9 July 2016, accepted 8 January 2017, available online 1 March 2017.
Copyright © 2017 M. Talanda. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
The amphisbaenians are one of several limbless, fossorial groups of lizards. They have an elongated trunk but short neck and tail. They show a rather unusual limb reduction pattern. Generally lizards tend to reduce their forelimbs before the hindlimbs but amphisbaenians are one of just a few exceptions to this rule (Caldwell 2003; Sakata and Hikida 2003; Miralles et al. 2015). While modern amphisbaenians are usually limbless, members of their family Bipedidae have well developed forelimbs and the Blanidae and Trogonophidae have retained some elements of the pectoral girdle. Members of the first two of these families have some vestiges of the hindlimb and all amphisbaenians have a pelvis, which is more or less reduced (Kearney 2002). In this respect, these three families might be considered primitive but the most basal group of modern amphisbaenians are Rhineuridae, which have the appendicular skeleton reduced to just a pair of small bony elements in the pelvic region (Kearney 2002). All this makes any consideration of amphisbaenian’s evolution difficult. How many times in this group have the hindlimbs or forelimbs been lost? Which were reduced first in the Rhineuridae, Amphisbaenidae, and Cadeidae? Does the reduction process resemble that of other limbless squamates or has it been different? Why did the Bipedidae retain nearly complete forelimbs and pectoral girdle?
Some light on these questions may be shed by Slavoia darevskii Sulimski, 1984, which was recently proposed to be a primitive amphisbaenian (Tałanda 2016). The skeleton of S. darevskii from the Barun Goyot Formation in Mongolia was originally described by Sulimski (1984). He suggested that the compact skull, reduction of size of the orbits, and structure of the pelvic girdle are adaptations of S. darevskii to a fossorial or semifossorial mode of life. He also noted that this lizard has some tendency toward limb reduction. Gao and Norell (2000) adhered to Sulimski’s (1984) interpretation.
This paper presents a description of the postcranial skeleton of S. darevskii. It is expected that the morphology of these bones may help in determining the mode of life of this lizard. Its basal position allows me to reconstruct the postcranial anatomy of amphisbaenians from before this group split into modern lineages.
Institutional abbreviations.—PIN, Institute of Paleontology, Russian Academy of Sciences, Moscow, Russia; ZPAL, Institute of Paleobiology, Polish Academy of Sciences, Warsaw, Poland.
Other abbreviations.—FLL, forelimb length; HLL, hind limb length; SE, snout-eye length; SVL, snout-vent length.
Material and methods
The specimens available for study are stored in ZPAL and PIN collections. The material was available to the original author (Sulimski 1984) except for ZPAL MgR-III/69, MgR-I/87, MgR-I/78, MgR-I/85, and PIN collection. Most of the material represents articulated skulls with mandibles. They are preserved three-dimensionally, although some specimens are more or less deformed. The contrast between bones and matrix in the CT scans is not strong, making their interpretation difficult. Because all specimens are more or less articulated, not all the sediment was removed to prevent them from the breakage. It is especially baffling in the case of the axial skeleton. Many anatomical details were covered by surrounding bones or sediment and are impossible to trace.
The specimens of Slavoia darevskii were already prepared and only minor mechanical preparation was carried out. They usually represent articulated partial skeletons. They were studied under the light microscope Olympus SZX10. Six CT-scans were done of the specimens: ZPAL MgR-I/8, MgR-I/9, MgR-I/87, MgR-III/77, MgR-III/79, and MgR-I/112. They provide information about the parts of the bones covered by sediment and other skeletal elements. The scanning was made by a GE Measurement & Control Solutions Phoenix/x-ray, model v/tome/x in the Faculty of Computer Science and Material Science, University of Silesia. This paper scopes on the postcranial morphology of S. darevskii that has been only preliminarily studied by previous researchers (Sulimski 1984; Gao and Norell 2000).
I included log-transformed measurements of S. darevskii (SVL = 69.3 mm; SE = 3.9 mm; FLL = 26 mm; HLL = 21.7 mm; five fingers, four toes) in the already published dataset (Lee et al. 2016) and performed a PCA analysis (the first variant). The measurements were based on specimens ZPAL MgR-I/8, PIN 3142/358, and ZPAL MgR-III/79 (see SOM 2, Supplementary Online Material available at http://app.pan.pl/SOM/app62-Talanda_SOM.pdf). I excluded tail lengths from the analysis because only the proximal part of the tail is known for S. darevskii. The analysis was performed in order to compare the body proportions of S. darevskii with lizards from different habitats.
Systematic palaeontology
Order Squamata Oppel, 1811
Superfamily Lacertoidea Oppel, 1811
Genus Slavoia Sulimski, 1984
Type species: Slavoia darevskii Sulimski, 1984; late Campanian, Khulsan, Gobi desert.
Slavoia darevskii Sulimski, 1984
Figs. 1–12.
Material.—Isolated skulls: ZPAL MgR-III/69, MgR-III/73, MgR-III/74, MgR-III/75, MgR-III/78, MgR-III/81, from Khermeen Tsav II. ZPAL MgR-I/10, MgR-I/11, MgR-I/93, MgR-I/96, MgR-I/97, MgR-I/98, MgR-I/100, MgR-I/101, MgR-I/102, MgR-I/103, MgR-I/104, MgR-I/105, MgR-I/107, MgR-I/108, MgR-I/109, MgR-I/110, MgR-I/111, MgR-I/113, MgR-I/114, MgR-I/115, MgR-I/116, from Khulsan. ZPAL MgR-I/1, MgR-I/2, MgR-I/3, MgR-I/4, MgR-I/5, MgR-I/6, MgR-I/7, MgR-I/87, from Nemegt. Specimens with postcranial material: PIN 3142/358, PIN 4487/14, from Khermeen Tsav. ZPAL MgR-III/76 (skull with partial skeleton), ZPAL MgR-III/77 (skull with cervical vertebrae), ZPAL MgR-III/79 (skeleton), ZPAL MgR-III/80 (skull with incomplete skeleton), ZPAL MgR-III/82 (pelvic girdle), from Khermeen Tsav II. ZPAL MgR-I/8 (skeleton, holotype), ZPAL MgR-I/9 (skeleton), ZPAL MgR-I/78 (partial skeleton), ZPAL MgR-I/94 (skull with cervical vertebrae), ZPAL MgR-I/99 (skull and two vertebrae), ZPAL MgR-I/106 (partial skeleton), ZPAL MgR-I/112 (skeleton), ZPAL MgR-I/117 (bone fragments), from Khulsan. ZPAL MgR-I/85 (skull and partial skeleton), no data about specific locality. All material from Barun Goyot Formation, Upper Cretaceous of Mongolia.
Description.—The skull of S. darevskii was described by Sulimski (1984). Numerous amphisbaenian traits in this species have been identified by Tałanda (2016), including the unique synapomorphies: a strong overlap of vomers and palatines or tight wrapping of pterygoid around the posteromedial surface of the quadrate. I suggested there that an enforcement of the snout preceded the elongation of the body and reduction of the limbs. In this paper, I will compare the S. darevskii skull to other burrowing lizards (Acontinae, Feylininae, Dibamidae, and the crown Amphisbaenia) only in the context of its fossoriality.
The axial skeleton: Sulimski (1984) described that S. darevskii possesses 26 presacral vertebrae (including seven cervical), two sacral and an unknown number of caudals. According to him all are procoelous. I confirm these observations.
Atlas (Fig. 1) consists of two neural arches fused together on the dorsal side and the intercentrum that closes the ring on its ventral side. The neural arches have a gabled form (Fig. 1A). They are wider than the intercentrum (expanded laterally) and slightly expanded laterally. The intercentrum is badly preserved in all specimens studied. Its diameter is similar to the centrum of the second vertebra but its length is shorter. The first hypapophysis is visible in ZPAL MgR-I/108 (Fig. 1C). It has a prominent, cranially directed ventral process like in Diplometopon zarudnyi (Trogonophidae; Gans 1960). It is fused to the first centrum. Other intercentra are not preserved. Apparently, they were rather small and probably not fused to the centra.
The axis has a high and broad neural spine that is descending cranially (SOM 1: fig. 1A). The spine protrudesposterior to the centrum. Prezygapophyses and postzygapophyses are well developed, the latter being a little larger (Fig. 1A). They are flat and nearly horizontal with rounded distal margins. The axis has two hypapophyses visible on ZPAL MgR-III/77. Its synapophyses protrude laterally. They are large and inclined posteriorly (Fig 1E).
The following cervical vertebrae resemble each other in their shapes (Fig. 1A, E; SOM 1: fig. 1A). Their neural spines are narrow and directed posteriorly. Synapophyses are high and well developed, as in the crown amphisbaenians (Folie et al. 2013; Bolet et al. 2014). They are inclined posteriorly. Zygapopsyses are broad and similar to those of the axis (Fig. 1A). The cervical vertebrae are generally robust, broad and short.
The first trunk vertebrae are morphologically similar to cervicals (SOM 1: fig. 1A). They become more and more elongated and narrow posteriorward. Their neural spines are reduced to very small posterior projections. Their synapophyses are small. Only zygapophyses remain well developed. The last trunk vertebrae are less slender than others resembling sacral ones (Fig. 1D).
The two sacral vertebrae are robust (Fig. 1F3; SOM 1: fig. 2C). Both possess higher neural arches than surrounding vertebrae and large zygapophyses. They lack neural spines. The transverse processes are flat and broad anteroposteriorly, but do not touch each other. Processes of the first pair are of a more cylindrical appearance (Fig. 1F3; SOM 1: fig. 2C).
The tail is best preserved in specimen ZPAL MgR-I/8, in which thirteen proximal vertebrae are represented (SOM 1: fig. 2C). The caudals are long and slender and decrease in size posteriorly rather fast. Their neurapophyses are small and short. The transverse processes are well developed, long, flat and oriented cranially in the most proximal caudal vertebrae (Fig. 1F1, F2). They become shorter and shorter posteriorly. The length of hemapophyses remains unknown. The zygapophyses are small. The preserved caudal vertebrae lack the autotomy septa, but their presence on more distal vertebrae cannot be ruled out because the tail is incomplete.
The cervical ribs are preserved in ZPAL MgR-I/78 and MgR-I/112 (Fig. 1E; SOM 1: fig. 1A). The first cervical ribs begin from the third vertebra, as seen in ZPAL MgR-I/78 on the left side (cervical ribs from the opposite side are displaced) and left side of ZPAL MgR-I/112. They are robust, short and broad. They widen distally like in lacertids, unlike narrow ribs of the amphisbaenians (Gauthier et al. 2012). The number of cervicals is often difficult to determine in fossil taxa. They are usually defined by the rib shape or attachment to the sternum. The ribs on the sixth vertebra are the first one, which is long and slender, resembling the dorsal ribs (Fig. 1E). However, both they and the next pair were probably not attached to the sternum, like in most lizards (including the closely related Teiidae and Lacertidae; Hoffstetter and Gasc 1969). Thus, S. darevskii had seven cervicals. This is lower than the count primitive for lizards, which is eight (Hoffstetter and Gasc 1969; Gauthier et al. 2012). Also Sulimski (1984) described S. darevskii as having had seven cervicals.
The trunk ribs are poorly preserved in all specimens studied and the number of inscriptional, dorsal and lumbar ribs is hard to determine. The first two types were long, slender and medially curved (Fig. 1D, E).
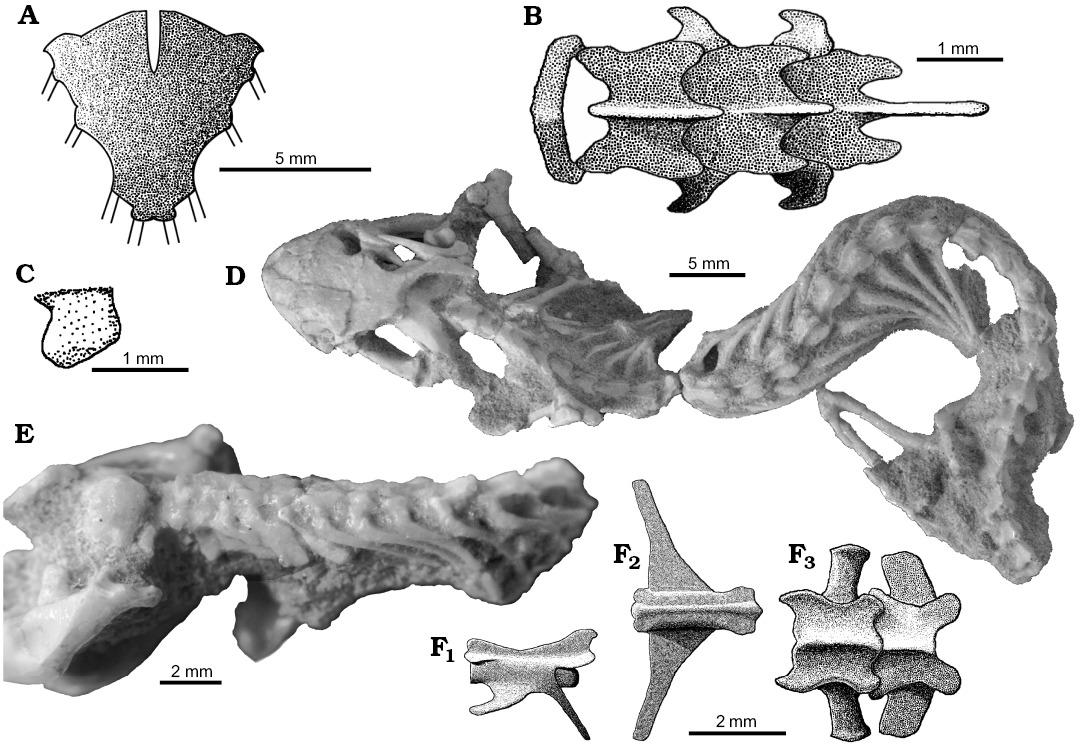
Fig. 1. The axial skeleton of the stem-amphisbaenid Slavoia darevskii Sulimski, 1984 from late Campanian of Mongolia. A. The reconstruction of the first four vertebrae in dorsal view (based on different specimens). B. The reconstruction of sternum in ventral view (based on PIN 4487/14). C. The reconstruction of first hypapophysis (based on ZPAL MgR-I/108). D. PIN 3142/358, complete presacral vertebrate column in dorsal view; photo courtesy of Vladimir Alifanov. E. ZPAL MgR-I/78, the neck with well preserved, blunt and broad cervical ribs. F. The reconstruction (based on ZPAL MgR-I/8) of the fifth caudal vertebra in lateral (F1) and dorsal (F2) views; sacral vertebrae in dorsal view (F3).
The appendicular skeleton: Several specimens have elements of pectoral girdle preserved (ZPAL MgR-III/76, MgR-III/79, MgR-III/82, MgR-I/8, MgR-I/9, MgR-I/85, MgR-I/101, MgR-I/106, MgR-I/112, PIN 4487/14, 3142/358; Figs. 2–4). Sulimski (1984) described and illustrated main bones (scapula-coracoids, interclavicle, and clavicles). He also mentioned the possible remains of a sternum in ZPAL MgR-I/108 and argued that this structure was not ossified in S. darevskii. During my investigation, I have also found ossified epicoracoids, prerequisites for the presence of suprascapulas and an ossified sternum (contra Sulimski 1984).
Sulimski (1984) did not find any remains of the sternum. He proposed that it was not ossified. However, it is preserved in specimen PIN 4487/14, where it is fully ossified (Fig. 1B). There are also visible remains of the sternum in ZPAL MgR-I/106. It was probably destroyed during preparation. The sternum has a shield-like form. It is ventrally convex. The sternum has at least four rib attachments and contacts the interclavicle antero-medially.
The interclavicle is cruciform (Figs. 2A, B, 4B, C), like in other lacertoids. The length and width of the interclavicle are similar. While the bone is flat in Timon lepidus, the interclavicle in S. darevskii is dorso-ventrally deep (Fig. 4B). It has very wide lateral processes. These processes form a distinct transverse crest. The crest delimits a transversal concavity faced antero-ventrally. The anterior and posterior processes are narrow and long. They are convex ventrally except for the concavity at the lateral processes level on the anterior process (Fig. 2A2). The process is pointed anteriorly. The bone is preserved in several specimens which enables us to observe its variation in proportions. It mainly concerns the relative length of the anterior and posterior processes (Fig. 2B).
The clavicles are curved, L- or J-shaped (Fig. 2C, D). They have variable morphology and do not depend on size. Holotype possesses slender, J-shaped clavicles that are rounded in cross-section. They become flat and wider medially and bifurcate at the medial end of the bone. They widen dorsally to form funnel-shaped endings, which suggest a clavicle-suprascapula contact. Other specimens of similar size, like ZPAL MgR-I/106 or MgR-I/101, have robust, L-shaped clavicles. They are wide and flat over the entire length. It is not clear whether they bifurcate medially. Both endings are badly preserved in all specimens with robust clavicles.
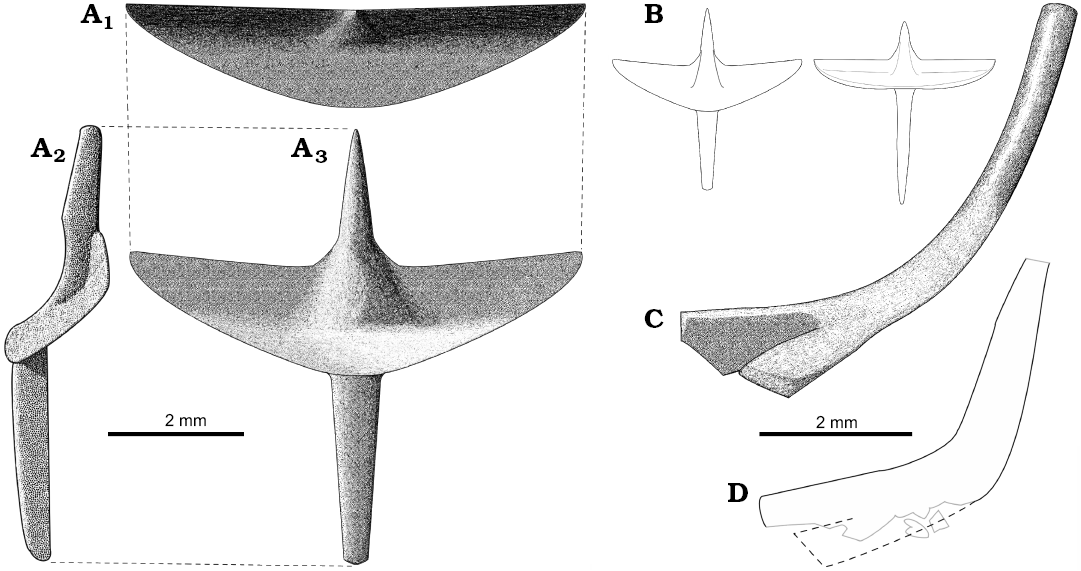
Fig. 2. Reconstructions of clavicles (C, D) and interclavicles (A, B) of Slavoia darevskii. A. Morphotype I based on ZPAL MgR-I/8, in anterior (C1), lateral (C2), and ventral (C3) views. B. Comparison of the morphotypes II and I in the same scale. C. Morphotype I based on ZPAL MgR-I/8. D. Outlines of the morphotype II preserved in ZPAL MgR-III/80; grey lines, damaged edges of the clavicle; dotted line, hypothetical reconstruction of missing part. Both in anterior view.
The scapula and coracoid are fused (in synostosis), like in lacertids (Müller 2001) and the suture is retained as a scar between the glenoid cavity and coracoid foramen (Figs. 3, 4). Their morphology is unique among Squamata because scapula is not fenestrated and forms a very thin subtriangular medial blade (Fig. 3A). It is often broken. Sulimski (1984: fig. 2) misinterpreted the damage of the blade as a scapular fenestra. The shaft of the scapula is narrow but robust. It is wider at the dorsal end (funnel-shaped), where it was in contact with the suprascapula. This extension reminds one of Bipes biporus Cope, 1894 (see Zangerl 1945). The variability of scapulae is difficult to determine due to the incompleteness of almost all the specimens. Some scapulae do not have the funnel-shaped dorsal ending but it might be a result of bad preservation. The scapulocoracoid fenestra is present. The coracoid of S. darevskii is flat and fan-shaped (Figs. 3, 4). There are no openings and the procoracoid is absent. The coracoid contributes to the ventral part of the glenoid cavity. The coracoid foramen is positioned medially to the glenoid. On specimen PIN 4487/14 a calcified epicoracoid is partially preserved (Figs. 3E, 4C).
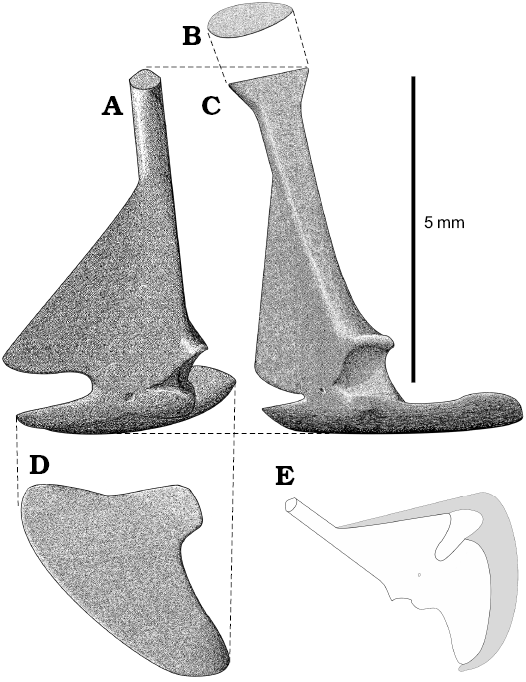
Fig. 3. Reconstruction of scapulocoracoid of Slavoia darevskii based on different specimens, in anterior (A), dorsal (B), lateral (C), and ventral (D) views. E. Reconstruction of epicoracoid assuming that it did not reach the suprascapula.
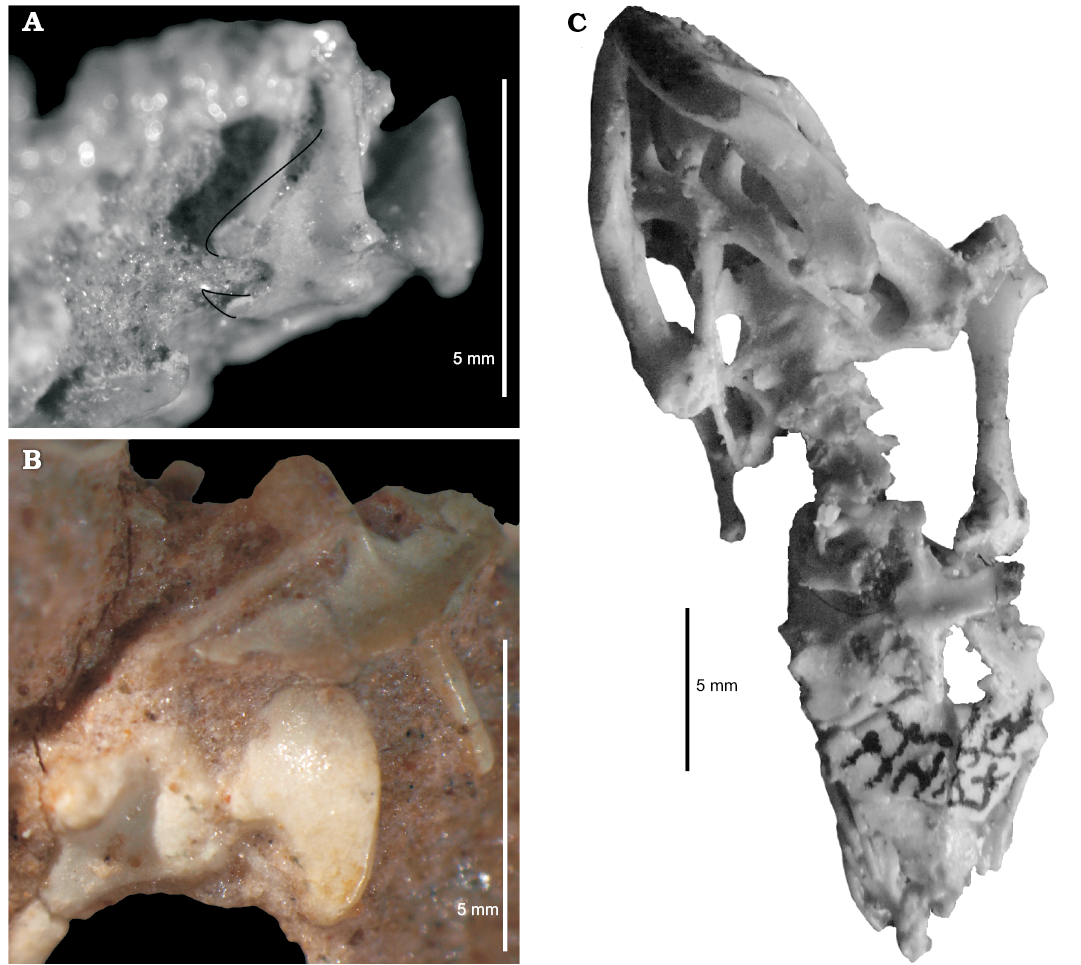
Fig. 4. Pectoral girdle of the stem-amphisbaenid Slavoia darevskii Sulimski, 1984 from late Campanian of Mongolia. A. ZPAL MgR-III/76, scapulacoracoid in anterior view; the black line marks missing parts seen in other specimens. B. ZPAL MgR-I/8, pectoral girdle with ventral edge of clavicle, interclavicle (morphotype I), coracoids, and proximal part of humerus. C. PIN 4487/14, sternum with rib attachments, coracoids with epicoracoids, interclavicle (morphotype I), humerus, cervical, and skull in ventral view.
The humerus of this lizard is well developed and elongated (Figs. 4–6), about 10 mm long in adult specimens. The proximal and distal ends are subtriangular in outline and rotated in respect to each other. The proximal end is concave ventrally. The humerus has a distinct deltopectoral crest oriented ventrally (Figs. 5B, 6). It extends for one fourth of the humerus length. The humerus epiphyses are weakly ossified or even not preserved in smaller individuals. The medial tuber is weakly developed. However, it is clear that the bone becomes wider towards the distal end and has a distinct attachments area with radial and ulnar condyles. Sulimski (1984) observed the presence of the ectepicondylar foramen. It is visible in ZPAL MgR-I/78.
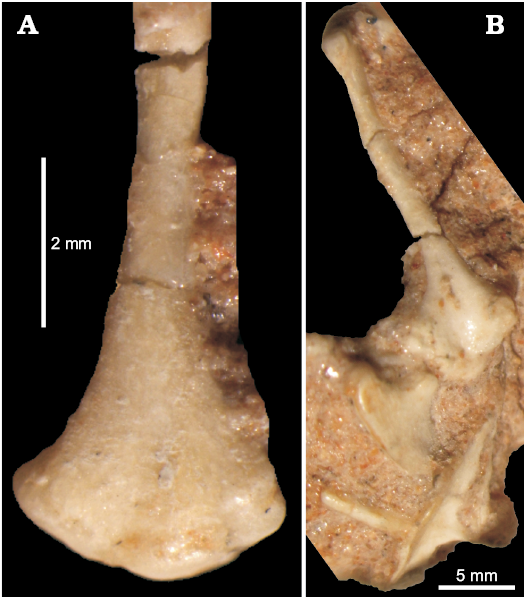
Fig. 5. Humerus of the stem-amphisbaenid Slavoia darevskii Sulimski, 1984 (ZPAL MgR-I/8) from late Campanian of Mongolia. A. Distal part in medial view. B. Humerus in ventral view, displaying also coracoid and interclavicule.
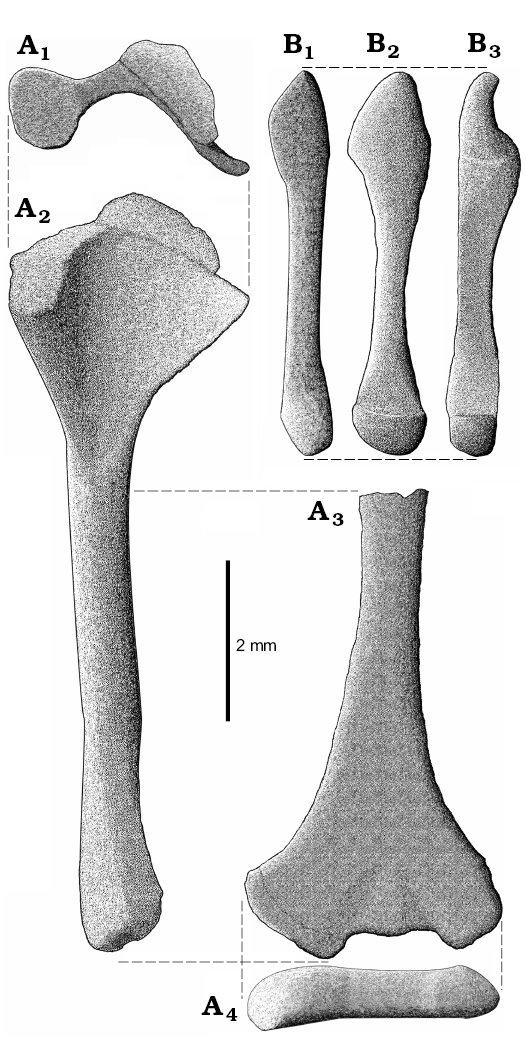
Fig. 6. Reconstruction of right humerus (A) and ulna (B) of Slavoia darevskii. A. ZPAL MgR-I/8, holotype. Proximal head with partially preserved epiphysis in proximal (A1) and ventral (A2) views, the shaft and distal head in medial view (A3), and distal head in distal view (A4). B. ZPAL MgR-I/9, subadult, in lateral (B1), posterior (B2), and medial (B3) views.
The ulna is longer and more massive than the radius but is shorter than the humerus (Figs. 6B, 7). The olecranon is prominent. The shaft is compressed laterally. Both ends of the bone are expanded. The proximal end has an indentation for the humerus. The distal end is probably convex and contacts the ulnare.
The radius is slender and delicate (Fig. 7B). It is shorter than the ulna because of olecranon on the latter bone. The proximal end is not well visible because of the surrounding bones but the distal end is distinctly expanded into two articulation surfaces (Fig. 7B2). In B. biporus the bone is also broader in this part but lacks a distinct lateral facet (Zangerl 1945).
The carpus is usually not well preserved but seems to be not reduced (Fig. 7). It is complete in ZPAL MgR- III/79 but its particular elements are indistinguishable (Fig. 7A1). In ZPAL MgR-I/8 and MgR-I/9, it is partly articulated but incomplete. The first specimen bears four elements around the distal head of the ulna (Fig. 7C). The large posterior element is probably the pisiform. The bone articulating with the ulna is the ulnare. The two bones next to the metacarpal are the distal carpals. The smaller and laterally-placed one is probably the distal carpal V. The larger, medially-positioned and contacting the ulnare one is probably the distal carpal IV. Four carpals are also visible in ZPAL MgR-I/9 (Fig. 7B). The first one is a small element between the ulna and radius. It is probably the carpal intermedium, the presence of which is also corroborated by a distinct articulation surface on the radius. More distally, there are two carpals articulated to each other and to the metacarpal II. They represent the lateral centrale and second distal carpal. The fourth element, placed next to IV metacarpal is probably the distal carpal IV. There is also another bone placed distally to the metacarpals. It is rectangular and has a relatively large distinct concave surface. Perhaps it is the disarticulated ulnare. To sum up, probably all ten carpals (including pisiform) were present in S. darevskii just like in the Lacertidae (Rieppel 1992; Maisano 2001). The B. biporus has nine elements, lacking an intermedium (Zangerl 1945; Kearney 2002).
The manus is fragmentary in ZPAL MgR-I/8 and MgR-I/9 but almost completely visible on the scan of ZPAL MgR-III/79 (Fig. 7A) and is well developed. All metacarpals are slender and have expanded ends. Contrary to typical lizards, their lengths are subequal. The metacarpal I and V are slightly shorter. The metacarpal III is probably the longest one. In B. biporus, the first three metacarpals are also subequal but the last two are shorter (Zangerl 1945). The phalanges are broad, especially the claws, which are also flat and large. This strongly resembles B. biporus (Zangerl 1945). The phalangeal formula is not easy to determine and proportions of most fingers are not typical for lizards (Shapiro et al. 2007). An examination of a CT-scan shows that the first and second fingers have their distal parts “cut off” in the same line (Fig. 7A1). The preserved part of digit I has the most proximal phalanx complete. The second phalanx is incomplete but the preserved part shows the condition of a regular phalanx—not like a claw. This corroborates the interpretation of the small remains of the third phalanx, which was probably the claw. The second digit has two proximal phalanges complete, and part of the third one. The third digit has three phalanges complete but lacks the claw. The next digit is complete and has four phalanges. The last of them forms a large, broad, flat claw. The fifth digit has an incomplete middle part but boundaries between the phalanges are preserved showing three distinct phalanges. The fifth claw is even larger than the previous one. Assuming that the second and third digits did not develop hyperphalangy, the most parsimonious phalangeal formula would be 3-3-4-4-3.
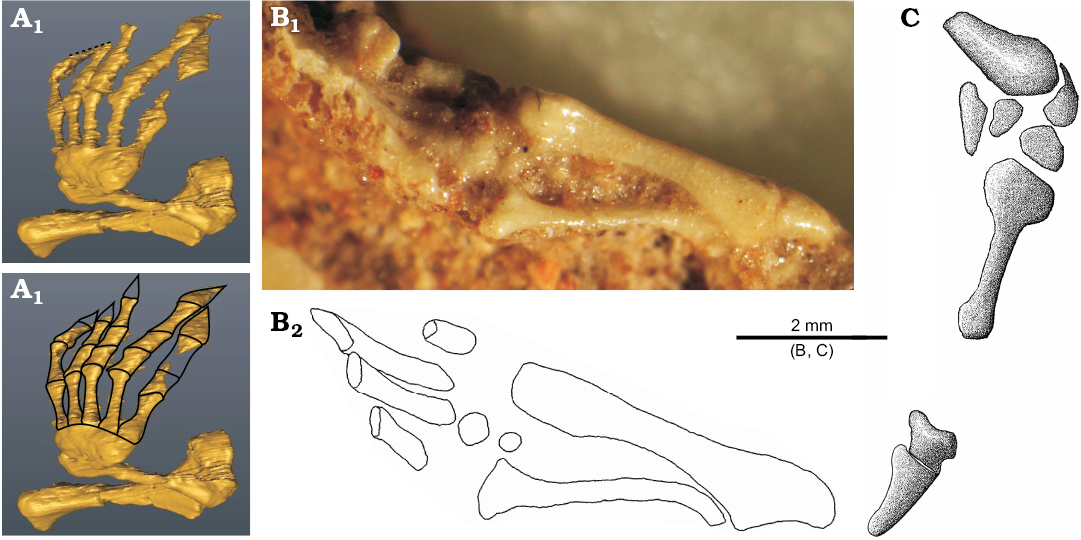
Fig. 7. Forelimb (with details of manus) of the stem-amphisbaenid Slavoia darevskii Sulimski, 1984 from late Campanian of Mongolia. A. ZPAL MgR-III/79, CT-scans. The dotted line in A1 marks surface where the digit I and II are eroded; outlines in A2 mark phalanges and metacarpals (preserved and reconstructed). B. ZPAL MgR-I/9; photograph (B1), outlines of the forearm and partially preserved hand (B2). C. ZPAL MgR-I/8, partially preserved hand.
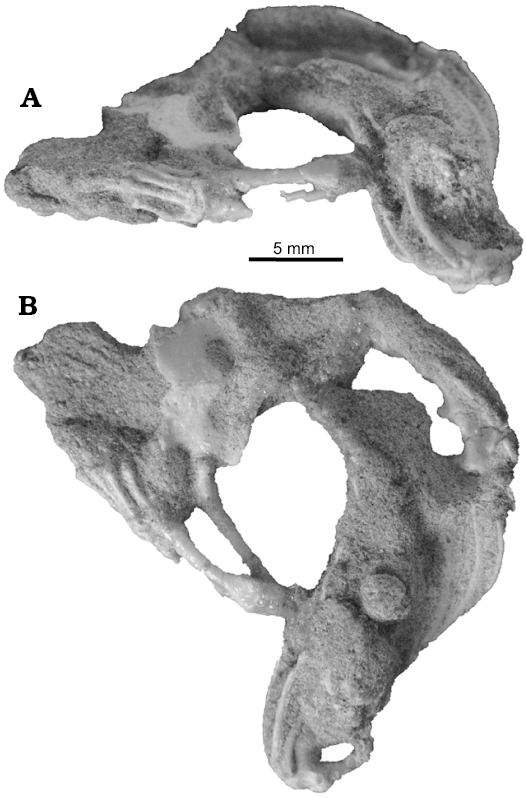
The pelvic and hindlimb anatomy were only briefly mentioned by Sulimski (1984). In the pelvis of S. darevskii (Fig. 8), the ilium is long and slightly curved caudally. It contacts the ischium and pubis ventrally to form the acetabulum. The dorsal part is slender but it expands antero-posteriorly in the mid part. The uppermost part tapers distally. Unlike lacertids, the preacetabular process is absent in S. darevskii.
The pubis is only partially preserved in the holotype and absent in other specimens from the ZPAL collection. It is long and curved medially. It forms the antero-ventral one third of the acetabulum. The pubic tuber is oriented cranio-laterally and appears near the acetabulum.
The ischium is visible only in PIN 3142/358 from its ventral side and on the scan of the holotype pelvis. It generally resembles that of Ameiva (Teiidae). It is flat and broad. The ventral part has a parallelogram shape and horizontal symphysial border. It has a distinct ischiadic tuberosity and the notch for hypoischium. The dorsal part contacts the ilium and pubis and contributes to the postero-ventral part of the acetabulum.
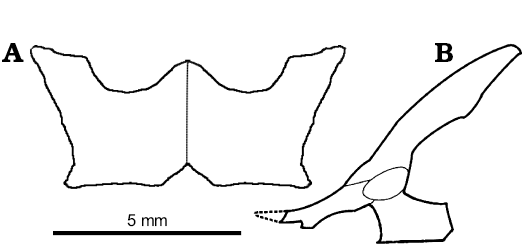
Fig. 8. Reconstruction of pelvis of Slavoia darevskii. A. Based on PIN 3142/358, paired ischia in ventral view. B. Based on ZPAL MgR-I/8, in lateral view; the dashed line marks missing part.
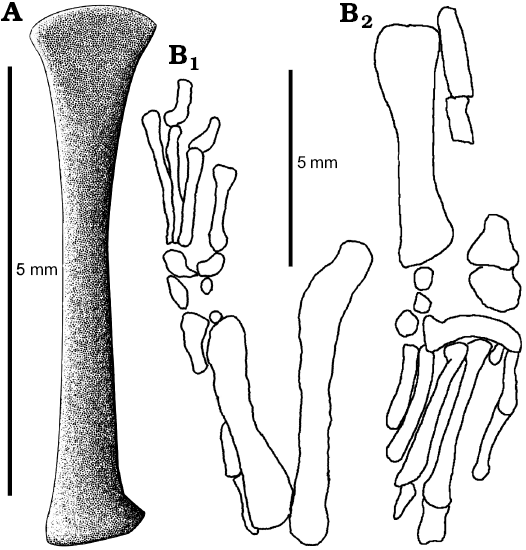
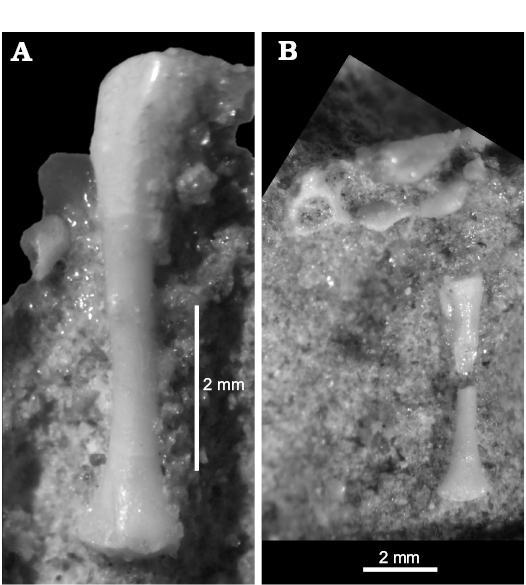
The femur is preserved in ZPAL MgR-I/8, MgR-I/9, and PIN 3142/358 (Figs. 9–11; SOM 1: fig. 2). It is long and slender. It is approximately 8 mm long in the holotype specimen, not 10 as measured by Sulimski (1984) and is longer than the tibia but shorter than the humerus. The shaft is rather smooth and rounded in cross-section. Both ends are expanded and rotated relative to each other (Fig. 10A). There is a well-developed crest next to the proximal head. This is probably the trochanter internalis (or fibular trochanter?). The distal end has a distinct tibial condyle.
The tibia is about 6 mm long as seen in ZPAL MgR-I/9 and PIN 3142/358 (Figs. 9, 10). It is more massive than the fibula. It has distinct heads on both its ends. The proximal end is broad. It has a flat proximal surface where the proximal epiphysis attached. A distinct crest is projecting dorsally to the proximal end of the tibia. It is a cnemial crest, which is also well developed in the Lacertidae (Parker 1900). The distal end is broad as well but compressed antero-posteriorly. It bears the remains of the distal epiphysis but they are too incomplete to determine the type of tibioastragalar articulation (Russell and Bauer 2008).
The fibula is only partially preserved. It is long and slender (Figs. 9, 11A). The proximal end was not expanded, contrary to the distal end.
The foot of S. darevskii is preserved only on PIN 3142/358 (Figs. 9B, 11). The tarsus is not well preserved. Particular bones are displaced. However, a distinct astragalocalcaneum is articulated to the fibula. Other elements are also present but difficult to distinguish. The rest of the foot is well preserved.
All the metatarsals are long and slender. They have expanded ends. Metatarsal V is the shortest and the IV is the longest one presenting the proportions typical for lizards. However, metatarsal V seems to be simplified and not hooked.
The phalangeal formula is uncertain but it was probably 0-1-1-2?-2?. The first digit is absent. The second and third are composed only by one phalanx each. Both are poorly ossified but resemble unguals in their shape. One complete, wide and short phalanx articulates the metatarsal IV and has a distal articulation surface for at least another phalanx. Similarly, digit V has only one phalanx preserved but it probably had more. The phalanx is long and slender unlike the one from digit IV.
Stratigraphic and geographic range.—Barun Goyot and Djadochta formations, Mongolia; late Campanian.
 |
 |
|
Fig. 9. Hindlimb of Slavoia darevskii. A. Based on ZPAL MgR-I/9, tibia in ventral view. B. Based on PIN 3142/358, outlines of hindlimb in dorsal (B1) and anterior (B2) views. |
Fig. 10. The stem-amphisbaenid Slavoia darevskii Sulimski, 1984 (ZPAL MgR-I/9) from late Campanian of Mongolia. A. Left femur in posterior view. B. Tibia in posterior view. |
|
|
|
Fig. 11. The stem-amphisbaenid Slavoia darevskii Sulimski, 1984 (PIN 3142/358, posterior half) from late Campanian of Mongolia; displaying nearly complete hindlimb and ischia. Photograph from the rear showing reduced pes (A), ventral side (B). Photos courtesy of Vladimir Alifanov. |
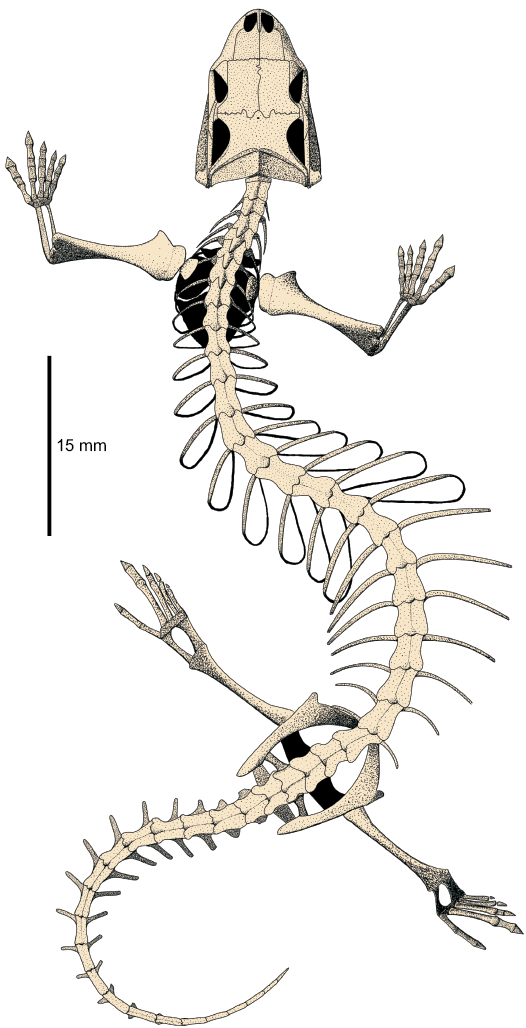
Fig. 12. Reconstruction of the whole skeleton of Slavoia darevskii in dorsal view. |
Discussion
Fossorial adaptations of Slavoia darevskii.—The PCA analysis found that two components explain more than 85% of the total variance (76.56% and 11.891%, respectively). They underline the unique morphology of S. darevskii among known lizards (Fig. 13). It does not group with any of them. The Principal Component (PC) 1 is influenced by almost all variables (loadings: -0.4254 for fingers; 0.4502 for forelimbs; 0.4532 for the hindlimb; -0.4394 for SVL; 0.4667 for toes). Snake-like, burrowing forms have usually lower values of PC1 than other lizards. Slavoia darevskii have a similar value as the second group. This is not surprising, as S. darevskii has a short body and still relatively well developed limbs. PC2 is influenced much more by SVL, the number of fingers, and forelimb length than by the negative values of the hindlimb length, and number of toes (loadings: 0.5801, 0.5215, 0.4867, -0.3181, -0.2314, respectively). Interestingly, Bipedidae have the highest values and S. darevskii is the closest lizard to them, despite its short SVL. This mirrors the transitional morphology of S. darevskii, which has more developed forelimbs than hindlimbs. It may also mean that S. darevskii and Bipedidae represent different forms of burrowing adaptations than lizards from the bottom of the diagram.
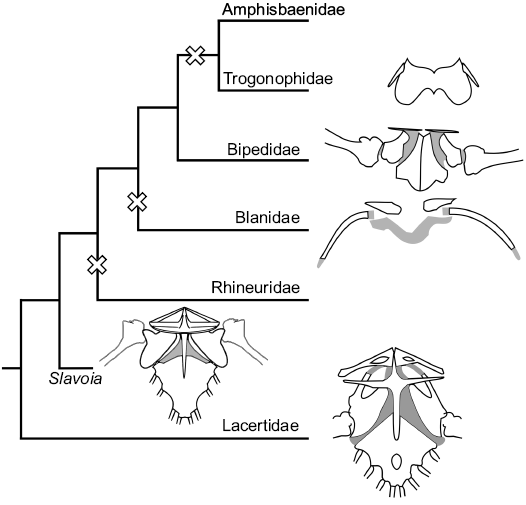
Fig. 13. Evolution of pectoral girdles of Lacertidae, Slavoia darevskii, and families of Amphisbaenia. Crosses mark at least three independent losses of the forelimbs among worm lizards.
The fossorial habit of S. darevskii was already proposed by Sulimski (1984). He mainly pointed out a compact skull and reduced orbits, which indicate such a mode of life. I confirm his observations and the new data added are congruent with his hypothesis. Indeed, the whole skull of the S. darevskii is massively built and compact, especially in the anterior part. This resembles other burrowing lizards, for example representatives of the families Scincidae (Acontinae and Feylininae; Rieppel 1981), Dibamidae (Greer 1985), and especially the crown Amphisbaenia (Gans 1960; Kearney et al. 2005; Hembree 2007; Tałanda 2016). However, S. darevskii differs from other burrowing forms (with the exception of A. plumbeus) in lacking the anterolateral processes of the frontals, which additionally enforce the snout structure. Slavoia darevskii had a short parietal segment and not as much integrated (in terms of braincase-parietal union) as it is in amphisbaenians and dibamids (e.g., Estes et al. 1988; Lee 1998). It also retains the temporal arch, which is usually lost in such lizards, with the exception of the most primitive burrowing skinks (Rieppel 1981).
Burrowing lizards have also more or less reduced bones that surround the orbit. Only the prefrontal and frontal remain well developed (Gans 1960; Greer 1985; Kearney et al. 2005; Hembree 2007). Slavoia darevskii, Acontinae and Feylininae (Rieppel 1981) have the frontal excluded from the dorsal margin of the orbit by a prefrontal-postfrontal contact. More advanced forms regained the frontal presence in the orbit by reducing the postfrontal (Gans 1960; Greer 1985). Slavoia darevskii have reduced the lacrimal and posterior process of the jugal but the postorbital and postfrontal have a primitive condition. To sum up, in the skull S. darevskii displays several features typical of burrowing lizards but they represent a very early stage of adaptation of this habit. This accords well with ancient geological time and the basal phylogenetic position of this lizard (Sulimski 1984; Tałanda 2016).
A shortened neck usually characterizes limbless, burrowing forms like the amphisbaenians and dibamids (Hoffstetter and Gasc 1969). This was probably connected with the modification of the axial musculature towards their specialized locomotion (cf. Wake 1993). As mentioned by Sulimski (1984), the neck of S. darevskii bears large areas of muscle attachment. Cervical ribs appear on the third vertebra and with the exception of the last, they are very broad (Fig. 1E). Relatively massive clavicles and the interclavicle delimit the neck (Fig. 2). They served for the attachments of relatively large muscles, which moved the head of S. darevskii. Moreover, this was accompanied by a reduction of the neck length. In most lizards the neck has eight vertebrae (Hoffstetter and Gasc 1969). This number was confirmed as plesiomorphic for lizards by phylogenetic studies (i.e., Gauthier et al. 2012). The neck of S. darevskii is shorter and bears only seven cervicals. This is underlined by the appearance of cervical ribs on the third—not on the fourth vertebra, as is in most other lizards.
The massive coracoids, the lower parts of the clavicles and calcified sternum served as strong adductors and abductors of the forelimb. In contrast, the scapula is less developed and is unfenestrated like in Bipes (Kearney 2002). This suggests a dominance of the antero-posterior forelimb movements. This anatomy becomes clear when we look at the forelimb. The humerus of S. darevskii is well developed and long (10 mm in the holotype)—longer than the femur (about 8 mm in the same specimen). This trait is quite unusual among Squamata. The hand is shovel-like: flat and broad. The large olecranon process reminds one of burrowing mammals (Goldstein 1972). It provides an efficient lever for the triceps brachii muscle which extend the elbow joint (Fujiwara 2009). All this strongly suggests that the forelimbs participated in tunneling, like in some modern lizards (Ananjeva 1977; Renous 1977) but to a higher degree. It was probably a kind of compensation for rather primitive head adaptations for burrowing (see above).
Lizards living in a complex habitat (like dense vegetation, litter or soil) tend to elongate their trunks and lose their limbs. Slavoia darevskii probably lived underground. This lizard had short limbs and a moderately elongated trunk (Figs. 1D, 13). The holotype is about 65 mm in length (SVL), including a 13–14 mm head, while the length of the limb elements are 10 mm for the humerus and about 8 mm for the femur. This is rather short for limbs if we look at other lizards (e.g., Pounds et al. 1983; Herrel et al. 2008). The elongation of the body is not evident because the number of presacral vertebrae—26, is typical for unspecialized lizards (Hoffstetter and Gasc 1969). But the elongation was expressed in the anterior shift of the cervical-thoracal border (see the description and the second part of the discussion). Moreover, body elongation might be achieved by that of the vertebral centra. Indeed, S. darevskii has the thoracic centra slender more elongated than usual in lizards, recalling amphisbaenian proportions (Hoffstetter and Gasc 1969). Dorsal vertebrae have low neural arches and reduced neural spines. It is also observed in modern burrowing lizards (Gans 1960). Probably pushing the body through a tunnel is more efficient with a lower trunk and living underground does not require vertical movements of the trunk.
The absence of a preacetabular process accompanies the reduced neural spines. These structures participate in lifting up the trunk (Snyder 1954) but are useless underground. The rest of the pelvis resembles that in a typical lizard. It is broad and connects the vertebrae in the dorso-posterior part of the ilium, where the bone is slightly broader. The acetabulum is positioned far away (ventro-anterior) from this contact. This type of anatomy characterizes lizards with greater involvement of the axial skeleton in locomotion (Borsuk-Białynicka 2008). Borsuk-Białynicka (2008) stated that it might be a preadaptation for limb reduction in lizards.
The hindlimb of S. darevskii favors this hypothesis. The femur is short when compared to the trunk and humerus (see above). The pes is partially reduced (Figs. 9, 11). The metatarsals are well developed, with the exception of the V, which is simplified. The phalanges seem to be reduced. It is a typical condition in primitive burrowing forms. The reduction of phalanges precedes the reduction of metatarsum. However, the digit of metatarsal V is better developed than others, whereas in other lizards it tends to be reduced as first (Greer 1991).
Evolution of the postcranium in amphisbaenians.—The fossil record of amphisbaenians is relatively good but it consists mainly of skulls and isolated vertebra. Given its age, phylogenetic position and completeness, S. darevskii offers a unique opportunity to study early amphisbaenian evolution. Lacertidae are the closest relatives of amphisbaenians. Because they have a very plesiomorphic postcranium, one may assume that it represents the state before these two groups split. Thus, lacertid morphology is the starting point in the considerations given below.
One of the most notable characteristics of amphisbaenians is their long, serpent-like body. It is a result of a high increase in the number of dorsal vertebrae, while the neck and tail have much less vertebrae than other lizards. The teiids and lacertids usually have 26 presacral vertebrae (Hoffstetter and Gasc 1969), so this is probably a primitive number for the whole Lacertoidea. This is supported by the same presacral vertebrae number in S. darevskii. It is surprising that at the beginning of their evolution, stem-amphisbaenians were not elongated. But the first changes towards such a body shape are already visible in S. darevskii. Usually Lacertoidea have eight cervical vertebrae (vertebrae which are not attached to the sternum; Hoffstetter and Gasc 1969). Slavoia darevskii probably had just seven, which opens the trend to shorten the neck in amphisbaenians. The change is not marked by any decrease or increase in the number of presacral vertebrae. It was just a shift of ribs specification towards the head. As a result, the first pair of ribs moved from the fourth to the third vertebra in S. darevskii which was inherited by all amphisbaenians (with a reversal in the Trogonophidae; Gans 1960). The cervical-trunk transition acted similarly. The ribs, starting from the sixth vertebra—not the seventh, are long and slender, resembling dorsal ones, so the trunk was probably one vertebra closer to the head. As a consequence, elongation of the trunk already began even if the overall number of presacral vertebrae remained the same. Modern forms have fewer cervicals than S. darevskii and extremely elongated trunks (Hoffstetter and Gasc 1969; Caldwell 2003). One cannot exclude that these modifications evolved independently in the main amphisbaenian lineages after their split. This would explain the differences in their vertebrate counts. The anterior cervical ribs of amphisbaenians taper distally, while in S. darevskii and other lacertoids they are blunt and wide at their ends (Gauthier et al. 2012). This means that the shape of cervical ribs in amphisbaenians is a derived condition, while S. darevskii still had an ancestral state. Another peculiar trait of amphisbaenians is their short tail (Zangerl 1945). The complete caudal series of S. darevskii is unknown but preserved parts suggest it was already shorter than in lacertids (Fig. 12).
The pectoral girdle is complete in S. darevskii and resembles that of the Lacertidae and Teiidae in having a cruciform interclavicle which is completely lost in modern amphisbaenians (Figs. 13, 14). It has no signs of reduction in S. darevskii . What is more, it is derived in respect of the lateral processes shape. They are antero-posteriorly wide and deep. This forms a large cavity, probably for the neck muscles. This is in great contrast with a flat and thin interclavicle of the lacertids and complete absence of this bone in the amphisbaenians. It is difficult to explain this phenomenon evolutionary. One may speculate that it was some kind of trade-off for S. darevskii . This lizard had still too primitive body for an efficient burrowing. Stronger muscles extending from the interclavicle could act as flexors of the head and thus play an important role in neck movement during digging. Later in the evolution, a more advanced head anatomy, shorter neck and other adaptations might have taken over that function, making the interclavicle redundant. A similar situation could be with the forelimbs. They are robust, relatively long and their movement was supported by muscles attached to a well-developed coracoid and sternum.
The phalanges are relatively broad resulting in its shovel-like appearance of the hand. As mentioned above, all this suggests that the forelimbs also contributed in digging, which might explain why the Bipedidae retained them. Again, further modification of the skull and axial column could also exclude forelimbs from tunneling with a subsequent reduction in most modern families. Despite these derived features, the pectoral girdle of S. darevskii shows the first signs of reduction. The scapulacoracoid lacks the coracoid emargination—present in the lacertids, and has only a scapulacoracoid emargination as in B. biporus (Kearney 2002). Slavoia darevskii resembles B. biporus also in hand digit morphology and count. Both taxa have broad digits and claws. While limbed lizards usually have the digital formula 2-3-4-5-3, B. biporus have it equalized to 3-3-3-3-3 (Kearney 2002). Slavoia darevskii perfectly fits between the two, probably having 3-3-4-4-3. It seems that hyperphalangy of the first digit and equalizing the digital formula in manus are apomorphies of the amphisbaenian lineage, not just Bipedidae. This strongly supports independent forelimb reduction in different amphisbaenian lineages (Figs. 13, 14), in the Rhineuridae, perhaps in Blanidae, and at least once after the split of Bipedidae (Kearney and Stuart 2004; Vidal and Hedges 2009). The position of Cadeidae is problematic but it would not be surprising if they also lost forelimbs independently.
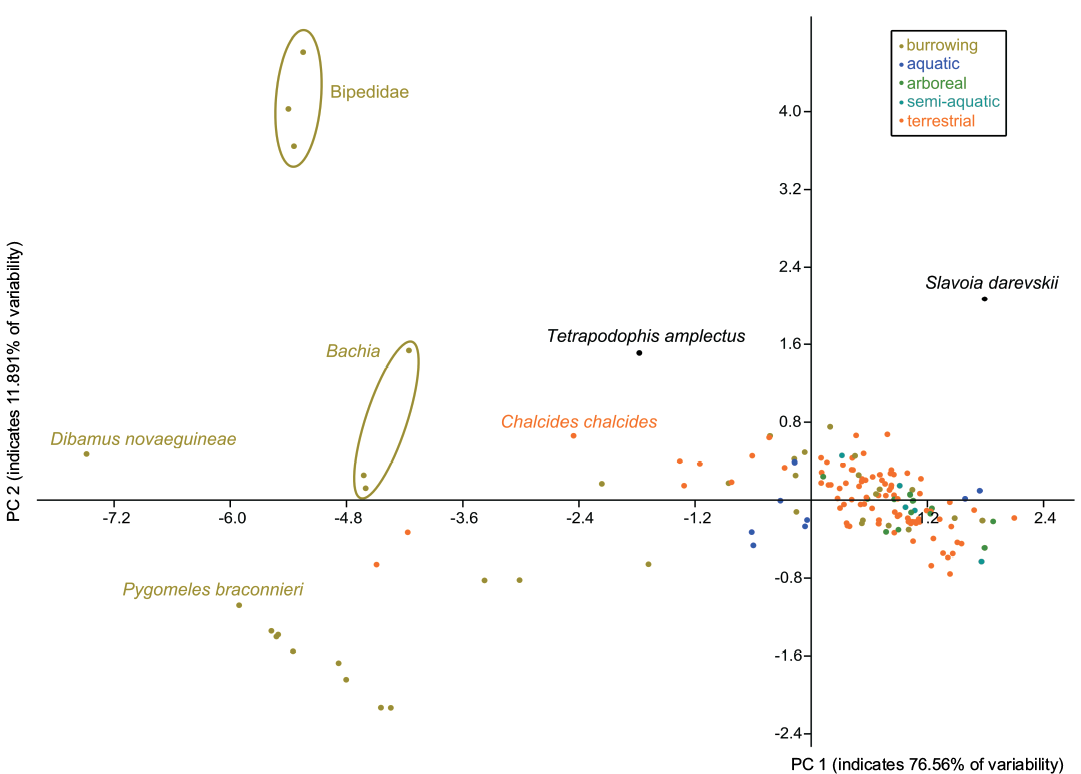
Fig. 14. Principal Component Analysis of body shape in lepidosaurian reptiles from different habitats. According to PC2, Slavoia darevskii is close to Bipedidae but distant according to PC1 (see Discussion). See Lee et al. (2016) for the data set.
The alternative scenario would be independent forelimb enforcement in S. darevskii and Bipedidae. This hypothesis considers the condition of S. darevskii as autapomorphy of this taxon—not a primitive condition of the amphisbaenian lineage. However, taking into account that (i) going deeper in time the probability of founding primitive traits increases; (ii) it is more parsimonious to assume that equalization of the digital formula occurred once rather than twice among amphisbaenians, especially that hyperphalangy is a rare phenomenon among Squamata (Greer 1992); (iii) gaining morphological complexity is generally more difficult than losing it—I consider this hypothesis less likely.
The partial hindlimb reduction in S. darevskii accords well with the condition seen in the Bipedidae. The loss of the preacetabular process and most digits in the pes of S. darevskii were the first steps in pelvis and hindlimb reduction. The phylogenetic position of S. darevskii (Tałanda 2016) suggests that this process was initiated before the split of the main amphisbaenian groups but could be continued independently by different lineages, as suggested by the pelvic anatomy of modern forms (Kearney 2002).
Such multiple losses of limbs among amphisbaenians show that this process might be even more common among the Squamata than previously thought. In the amphisbaenians this seems to be not directly linked with their diversification and dispersal, which were rather a result of a faunal exchange between continents (Tałanda 2016). The phenomena of limb reduction were widely discussed in the literature (Gans 1975; Greer 1991; Caldwell 2003; Kearney and Stuart 2004; Wiens et al. 2006; Skinner et al. 2008). One of the main topics of these discussions was the number of limb losses among Squamata and why in some groups this is a relatively frequent process and in others it is not (Greer 1991; Wiens et al. 2006; Skinner et al. 2008). The fossil record clearly shows that these considerations need paleontological evidence to be appropriately assessed. Until recently, the Lacertidae were among the lizard groups in which limb reduction was not observed (Greer 1991). The interpretation of Cryptolacerta—a lizard with partially reduced both fore- and hindlimbs, as a member of Lacertidae (Tałanda 2016), suggest that this group was also able to evolve such modifications. The absence of modern lacertids with reduced limbs might be a result of competition with amphisbaenians or climatic change in the late Neogene, which severely decimated species diversity in northern continents (i.e., Stanley and Ruddiman 1994; Ivanov 2007). Thus, the number of evolutionary lineages with limb reduction among lizards from these areas (i.e., Lacertidae, Amphisbaenia, Dibamidae) may be reduced by recent climatic events.
Conclusions
Slavoia darevskii bears clear adaptations for a burrowing mode of life in having a compact snout, short neck and strong forelimbs. However, it also has several primitive traits suggesting a very early stage of evolution towards this special ecology.
The postcranial skeleton of S. darevskii links the very different anatomies of the Amphisbaenia and Lacertidae. It shows that the dominance of forelimbs and hyperphalangy of the first manual finger were ancestral to all amphisbaenians. It was probably subsequently lost with all limbs in several lineages but retained in the Bipedidae. This forelimb specialization was accompanied by the hindlimb reduction. The latter was initiated before the split of modern families but continued later, probably independently. The shift in ribs specification toward the head and the reduction of neural spines preceded the elongation of the presacral column. This is summarized in Fig. 15.
Such transitional fossils as S. darevskii may offer an empirical background for developmental studies (Skawiński and Tałanda 2015).
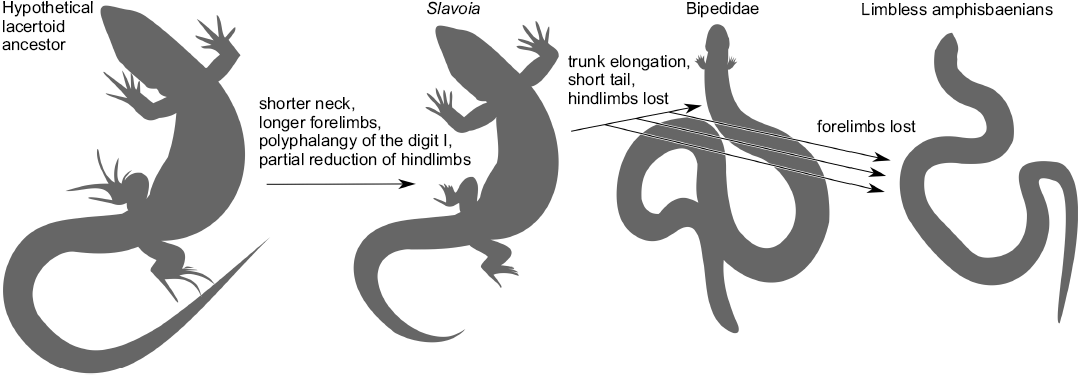
Fig. 15. Sequence of evolutionary changes in the amphisbaenian body plan proposed in this work. The phylogenetic relationships are taken from Kearney and Stuart (2004) and Tałanda (2016).
Acknowledgements
I would like to thank Magdalena Borsuk-Białynicka and Jerzy Dzik (both ZPAL) for their comments on the manuscript. I also thank Vladimir Alifanov (Russian Academy of Sciences, Moscow, Russia) for access to specimens and photographs. X-ray microtomography imaging has been provided by Marcin Binkowski (University of Silesia, Katowice, Poland). The funding for this study came from the grant 2014/13/N/NZ8/02467 founded by National Science Centre. I thank Michael S.Y. Lee (Flinders University and South Australian Museum, Australia) for helpful review.
References
Ananjeva, N.B. 1977. Morphometrical analysis of limb proportions of five sympatric species of desert lizards (Sauria, Eremias) in the southern Balkhash lake region. Proceedings of the Zoological Institute, Academy of Sciences of the USSR 74: 3–13.
Bolet, A., Delfino, M., Fortuny, J., Almécija, S., Roblem, J.M., and Alba, D.M. 2014. An amphisbaenian skull from the European Miocene and the evolution of Mediterranean worm lizards. PLoS ONE 9: e98082. crossref
Borsuk-Białynicka, M. 2008. Evolution of the iliosacral joint in diapsid phylogeny. Neues Jahrbuch für Geologie und Paläontologie Abhandlungen 249: 297–311. Crossref
Caldwell, M.W. 2003. “Without a leg to stand on”: on the evolution and development of axial elongation and limblessness in tetrapods. Canadian Journal of Earth Sciences 40: 573–588. Crossref
Cope, E.D. 1894. On the genera and species of Euchirotidæ. American Naturalist 28: 436–437.
Estes, R., de Queiroz, K., and Gauthier, J. 1988. Phylogenetic relationships within Squamata. In: R. Estes and G. Pregill (eds.), Phylogenetic Relationships of the Lizard Families: Essays Commemorating Charles L. Camp, 119–281. Stanford University Press, Stanford.
Folie, A., Smith, R., and Smith, T. 2013. New amphisbaenian lizards from the Early Paleogene of Europe and their implications for the early evolution of modern amphisbaenians. Geologica Belgica 16: 227–235.
Fujiwara, S.I. 2009. Olecranon orientation as an indicator of elbow joint angle in the stance phase, and estimation of forelimb posture in extinct quadruped animals. Journal of Morphology 270: 1107–1121. Crossref
Gans, C. 1960. A taxonomic revision of the Trogonophinae, and a functional interpretation of the amphisbaenid adaptive pattern. Bulletin of the American Museum of Natural History 119: 129–204.
Gans, C. 1975. Tetrapod limblessness: evolution and functional corollaries. Integrative and Comparative Biology 15: 455–467. Crossref
Gao, K.-Q. and Norell, M.A. 2000. Taxonomic composition and systematics of Late Cretaceous lizard assemblages from Ukhaa Tolgod and adjacent localities, Mongolian Gobi Desert. Bulletin of the American Museum of Natural History 249: 1–118.
Gauthier, J.A., Kearney, M., Maisano, J.A., Rieppel, O., and Behlke, A.D.B. 2012. Assembling the squamate tree of life: perspectives from the phenotype and the fossil record. Bulletin of the Peabody Museum of Natural History 53: 3–308. Crossref
Goldstein, B. 1972. Allometric analysis of relative humerus width and olecranon length in some unspecialized burrowing mammals. Journal of Mammalogy 53: 148–156. Crossref
Greer, A.E. 1985. The relationships of the lizard genera Anelytropsis and Dibamus. Journal of Herpetology 19: 116–156. Crossref
Greer, A.E. 1991. Limb reduction in squamates: identification of the lineages and discussion of the trends. Journal of Herpetology 25: 166–173. Crossref
Greer, A.E. 1992. Hyperphalangy in squamates: insight on the reacquisition of primitive character states in limb-reduced lineages. Journal of herpetology 26: 327–329. Crossref
Hembree, D.I. 2007. Phylogenetic revision of Rhineuridae (Reptilia: Squamata: Amphisbaenia) from the Eocene to Miocene of North America. The University of Kansas Paleontological Contributions 15: 1–20.
Herrel, A., Vanhooydonck, B., and Irschick, D.J. 2008. Anatomical basis of differences in locomotor behavior in Anolis lizards: A comparison between two ecomorphs. Bulletin of the Museum of Comparative Zoology 159: 213–238. Crossref
Hoffstetter, R., and Gasc, J.-P. 1969. Vertebrae and ribs of modern reptiles. Biology of the Reptilia 1: 201–310.
Ivanov, M. 2007. Herpetological assemblages from the Pliocene to middle Pleistocene in Central Europe: palaeoecological significance. Geodiversitas 29: 297–320.
Kearney, M. 2002. The appendicular skeleton in amphisbaenians. Copeia 2002: 719–738. Crossref
Kearney, M. and Stuart, B.L. 2004. Repeated evolution of limblesness and digging heads in worm lizards revealed by DNA from old bones. Proceedings of the Royal B 271: 1677–1683. Crossref
Kearney, M., Maisano, J.A., and Rowe, T. 2005. Cranial anatomy of the extinct amphisbaenian Rhineura hatcherii (Squamata, Amphisbaenia) based on high resolution X-ray computed tomography. Journal of Morphology 264: 1–33. Crossref
Lee, M.S.Y. 1998. Convergent evolution and character correlation in burrowing reptiles: towards a resolution of squamate relationships. Biological Journal of the Linnean Society 65: 369–453. Crossref
Lee, M.S.Y., Palci, A., Jones, M.E., Caldwell, M.W., Holmes, J.D., and Reisz, R.R. 2016. Aquatic adaptations in the four limbs of the snake-like reptile Tetrapodophis from the Lower Cretaceous of Brazil. Cretaceous Research 66: 194–199. Crossref
Maisano, J.A. 2001. A survey of state of ossification in neonatal squamates. Herpetological Monographs 15: 135–157. Crossref
Miralles, A., Hipsley, C.A., Erens, J., Gehara, M., Rakotoarison, A., Glaw, F., Müller, J., and Vences, M. 2015. Distinct patterns of desynchronized limb regression in Malagasy scincine lizards (Squamata, Scincidae). PLoS ONE 10: e0126074. Crossref
Müller, J.N. 2001. Osteology and relationships of Eolacerta robusta, a lizard from the Middle Eocene of Germany (Reptilia, Squamata). Journal of Vertebrate Paleontology 21: 261–278.
Parker, T.J. 1900. A Course of Instruction in Zootomy (Vertebrata). xviii + 397 pp. Macmillan and Co., London.
Pounds, A.J., Jackson, J.K., and Shively, S.H. 1983. Allometric growth of the hind limbs of some terrestrial iguanid lizards. American Midland Naturalist 110: 201–206. Crossref
Renous, S. 1977. Musculature of the buccal floor of Bipes canaliculatus (Reptilia: Amphisbaenia). Copeia 1977: 464–471. Crossref
Rieppel, O. 1981. The skull and the jaw adductor musculature in some burrowing scincomorph lizards of the genera Acontias, Typhlosaurus and Feylinia. Journal of Zoology 195: 493–528. Crossref
Rieppel, O. 1992. Studies on skeleton formation in reptiles: III, patterns of ossification in the skeleton of Lacerta vivipara Jacquin (Reptilia, Squamata). Fieldiana (Zoology) 68: 1–25.
Russell, A.P. and Bauer, A.M. 2008. The Appendicular Locomotor Apparatus of Sphenodon and Normal-limbed Squamates. Biology of the Reptilia 21: 1–465.
Sakata, S. and Hikida, T. 2003. A fossorial lizard with forelimbs only: Description of a new genus and species of Malagasy skink (Reptilia: Squamata: Scincidae). Current Herpetology 22: 9–15. Crossref
Shapiro, M.D., Shubin, N.H., and Downs, J.P. 2007. Limb diversity and digit reduction in reptilian evolution. In: B.K. Hall (ed.), Fins into Limbs: Evolution, Development, and Transformation, 225–245. The University of Chicago Press, Chicago.
Skawiński, T. and Tałanda, M. 2015. Integrating developmental biology and the fossil record of reptiles. International Journal of Developmental Biology 58: 949–959. Crossref
Skinner, A., Lee, M.S.Y., and Hutchinson, M.N. 2008. Rapid and repeated limb loss in a clade of scincid lizards. BMC Evolutionary Biology 8: 310. Crossref
Snyder, R.C. 1954. The anatomy and function of the pelvic girdle and hindlimb in lizard locomotion. American Journal of Anatomy 95: 1–45. Crossref
Stanley, S.M. and Ruddiman, W.F. 1994. Neogene ice age in the North Atlantic region: climatic changes, biotic effects, and forcing factors. In: J.P. Kennet and S.M. Stanley (eds.), Effects of Past Global Change on Life, 118–133. National Academy Press, Washington, DC.
Sulimski, A. 1984. A new Cretaceous scincomorph lizard from Mongolia. Palaeontologia Polonica 46: 143–155.
Tałanda, M. 2016. Cretaceous roots of amphisbaenian lizards. Zoologica Scripta 45: 1–8. Crossref
Vidal, N. and Hedges, S.B. 2009. The molecular evolutionary tree of lizards, snakes and amphisbaenians. Comptes Rendus Biologies 332: 129–139. Crossref
Wake, M.H. 1993. The skull as a locomotor organ. The Skull 3: 197–240.
Wiens, J.J., Brandley, M.C., and Reeder, T.W. 2006. Why does a trait evolve multiple times within a clade? Repeated evolution of snakeline body form in squamate reptiles. Evolution 60: 123–141. Crossref
Zangerl, R. 1945. Contributions to the osteology of the post-cranial skeleton of the Amphisbaenidae. American Midland Naturalist 33: 764–780. Crossref
Acta Palaeontol. Pol. 62 (1): 9–23, 2017
https://doi.org/10.4202/app.00294.2016