

The West African enigma: Systematics, evolution, and palaeobiogeography of cardiid bivalve Procardium
JAN JOHAN TER POORTEN and RAFAEL LA PERNA
Poorten, J.J. ter and La Perna, R. 2017. The West African enigma: Systematics, evolution, and palaeobiogeography of cardiid bivalve Procardium. Acta Palaeontologica Polonica 62 (4): 729–757.
Procardium gen. nov. is proposed for a group of early Miocene to Recent large cardiids in the subfamily Cardiinae. The type species is Cardium indicum, the only living representative, previously assigned to the genus Cardium. It is a mainly West African species, with a very limited occurrence in the westernmost Mediterranean. Procardium gen. nov. and Cardium differ markedly with regard to shell characters and have distinct evolutionary and biogeographic histories. Six species, in the early Miocene to Pleistocene range, and one Recent species are assigned to the new genus: Procardium magnei sp. nov., P. jansseni sp. nov., P. danubianum, P. kunstleri, P. avisanense, P. diluvianum, and P. indicum. During the Miocene, Procardium gen. nov. had a wide distribution in Europe, including the Proto-Mediterranean Sea, Western and Central Paratethys and NE Atlantic, with a maximum diversity during the Langhian and Serravallian. Its palaeobiogeographic history was strongly controlled by climate. During the Langhian stage, warm conditions allowed the genus to reach its highest latitude, ca. 54° N, in the southern North Sea Basin. With cooling, its latitudinal range gradually retreated southward, becoming mainly Mediterranean in the Pliocene–Pleistocene, and West African at present.
Key words: Bivalvia, Cardiidae, systematics, Neogene, Quaternary, Africa, Europe.
Jan Johan ter Poorten [terpoorten@chello.nl], Integrative Research Center, Field Museum of Natural History, Chicago, IL 60605, USA.
Rafael La Perna [rafael.laperna@uniba.it], Dipartimento di Scienze della Terra e GeoAmbientali, Università di Bari, Via Orabona 4, 70125 Bari, Italy.
Received 22 March 2017, accepted 17 July 2017, available online 9 November 2017.
Copyright © 2017 J.J. ter Poorten and R. La Perna. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
Cardium indicum Lamarck, 1819 is a fairly well-known large cardiid with a mainly West African distribution, whose inclusion in the European molluscan fauna (ter Poorten and Gofas 2012) is based only on a limited occurrence off the African coast of the westernmost Mediterranean. This species differs markedly, in several shell characters, from the type species of the genus Cardium Linnaeus, 1758, viz. the West African species C. costatum Linnaeus, 1758. Cardium represents a small West African lineage (ter Poorten 2007), lacking a fossil record in Europe. Conversely, C. indicum belongs to a rather diverse group which was present in Europe since the early Miocene, but today has almost disappeared from this area. Another related genus, Bucardium Gray, 1853 (type species C. ringens Bruguière, 1789) ranges back to the early Miocene of the Aquitaine Basin and is today represented by a single species, living in the tropical waters of West Africa (La Perna 2016). The presence of three distinct groups of Cardiinae is well supported by molecular phylogenetics (Herrera et al. 2015), which identified C. costatum and B. ringens as sister taxa, and C. indicum as sister to the Cardium–Bucardium clade.
Based on these considerations, a new genus is herein proposed for C. indicum and its allied species from the Cenozoic of Europe. Several fossil species, sharing close similarities with C. indicum, and sometimes confused with the living species, have been described. With the exception of Cardium hians Brocchi, 1814, from the Mediterranean Pliocene–Pleistocene, these species are poorly known. These taxa have been interpreted in highly divergent ways (e.g., Smith 1945; Magne 1950; Zbyszewski 1957; Ghisotti 1971; Schultz 2003) and such a lack of consensus, together with the frequent poor state of preservation of their thin walled and fragile shells, has hampered a better knowledge of these cardiids.
Institutional abbreviations.—MPUR, Museo di Paleontologia dell’ Università di Roma “La Sapienza”, Italy; NHMW, Naturhistorisches Museum Wien, Austria; MNHN, Museum national d’Histoire naturelle, Paris, France; MPUB, Museo di Paleontologia, Università di Bari, Italy; MRSN BS, Bellardi and Sacco collection, Museo Regionale di Scienze Naturali, Turin, Italy; MSNM, Museo Civico di Storia Naturale di Milano, Italy; PH, Piet Hessel collection, Stichting Schepsel Schelp (collection associated with Naturalis Biodiversity Center), Rhenen, The Netherlands; RGM, formerly Rijksmuseum van Geologie en Mineralogie, now Naturalis Biodiversity Center, Palaeontology Department, Leiden, The Netherlands; RMNH, Naturalis Biodiversity Center, Leiden, The Netherlands; USNM, Smithsonian Institution, Washington DC, USA; ZMA, Zoölogisch Museum Amsterdam, now incorporated in Naturalis Biodiversity Center, Leiden, The Netherlands.
Other investigated collections.—EK, Eric Kaptein collection, Almelo, The Netherlands; JJTP, Jan Johan ter Poorten collection, Hilversum, The Netherlands; JV, Jaap van der Voort collection, Ostercappeln-Venne, Germany.
Other abbreviations.—H, shell height, L, shell length.
Systematic palaeontology
Class Bivalvia Linnaeus, 1758
Order Cardiida Ferussac, 1822
Family Cardiidae Lamarck, 1809
Subfamily Cardiinae Lamarck, 1809
Genus Procardium nov.
Etymology: From Latin, pro, before, early; and Cardium; in reference to genus origin, putatively ancestral to Cardium (gender neuter).
Type species: Cardium indicum Lamarck, 1819 (= C. darwini Mayer, 1866, C. delpeutum McGill, 1964), Recent, “Habite l’Ocean des grandes Indes” [Indian Ocean; erroneous, actually Western Africa].
Species included: Cardium diluvianum Lamarck, 1819; Cardium avisanense Fontannes, 1879; Cardium kunstleri Cossmann and Peyrot, 1912; Cardium danubianum Mayer, 1866; Procardium magnei sp. nov.; Procardium jansseni sp. nov.
Diagnosis.—Shell large, slightly longer than high, relatively thin and posteriorly gaping. 14–24 radial ribs, sculptured with horseshoe-shaped scales anteriorly and projecting spines posteriorly. Interspaces wider than ribs, often with a secondary riblet. Anterior cardinal blade like and posterior cardinal very small in left valve. Cardinal teeth erect, fully separated and similar in height in right valve.
Description.—Shell large (L 75–115 mm), generally slightly longer than high, weakly to strongly inequilateral, relatively thin, inflated, gaping posteriorly. Umbo orthogyrate to slightly prosogyrate. Anterior and ventral margins rounded, posterior margin truncated and nearly straight. Shell bearing 14–24 primary radial ribs, on anterior half each interspace with a weakly developed secondary riblet. Interspaces wider than primary ribs. Anterior ribs prominent with conical, ventrally hollow scales. Cross-section of anterior and median ribs roundly triangular or trapezium-shaped, sometimes with a radial groove on the rib tops. Ribs on postero-medial slope becoming asymmetrical in cross section, ultimately flattened posteriorly, with sculpture on posterior rib flanks gradually developing into pronounced, posteriorly directed spines. Rib impressions clearly visible from within, shell resorption present in rib impressions on postero-medial part. Hinge plate rather narrow and slightly sinuous, hinge angle very large (ca. 165–175°), cardinal teeth nearly horizontally rotated. Left valve: two cardinal teeth, the anterior one well defined and blade like, the posterior one tiny; both teeth connected at their bases and with a deep socket below. Right valve: two similar sized, erect, pointed cardinal teeth, not touching at their bases but separated by a deep socket. Anterior lateral sockets curved and extremely hollowed, lateral teeth of both valves thin, blade like. Tips of lateral teeth about equidistant from anterior cardinal. Ligament short. Lunule not well defined, dorsal margin slightly raised anterior of the umbo. Exterior colour dirty white, sometimes with commarginally arranged brownish areas, posteriorly pink or purple, except for white spines. Rusty-brown deposit present on interspaces of postero-medial part. Interior colour white with traces of cream, posterior margin purple.
Remarks.—Procardium gen. nov. can be separated from several related genera by a number of characters: the ribs are well developed, alternating with a tiny secondary riblet (Fig. 1B, D, F), anteriorly and medially carrying erect scales (Fig. 1F) and posteriorly strongly projecting spines. The shell is thin and gapes posteriorly (Fig. 1E). The hinge of the left valve has two very unequal cardinal teeth: the anterior one is elongate and blade like, located anterior to the very poorly developed posterior tooth. The right valve (Fig. 1A) has two projecting, equally sized and slightly diverging cardinals, that are fully separated and placed diagonally. It has an extremely excavated anterior lateral socket (Fig. 2A). The shell is white with a purple posterior slope.
In the related genus Cardium Linnaeus, 1758, consisting of two Recent species, the type species Cardium costatum Linnaeus, 1758 and C. maxicostatum ter Poorten, 2007, the ribs are very broad and flattened with a high projecting flange running on top, which is completely hollow. Additional rib sculpture is lacking, except for a poorly developed ridge running on the posterior ribs of C. maxicostatum. The shell is white, moderately solid and has a modest posterior gape. The interspaces never have a secondary riblet. The hinge of the left valve has two equally developed cardinal teeth, with the anterior tooth positioned ventral to the posterior tooth; the hinge of the right valve has two almost equal-sized projecting cardinals that are fully separated and placed along a horizontal axis. The hinge plate is straight (Fig. 2B).
A second related genus is Bucardium Gray, 1853, with its type species and sole Recent representative Bucardium ringens (Bruguière, 1789). A second species is known from the Burdigalian of the Aquitaine Basin, B. grateloupianum La Perna, 2016. Bucardium has a rounded, solid, slightly gaping shell. The ribs are broad, with narrow interspaces, almost smooth, and the posterior ones are strongly protuberant, resulting in a deeply incised margin. The left valve has two very unequal cardinal teeth, with the anterior tooth positioned ventral to the posterior tooth; the cardinal teeth of the right valve are equally sized, rather short and largely fused (Fig. 2C). The shell is white with a pinkish-purple posterior slope. With a length of 50 mm (Recent) or 65 mm (fossil), Bucardium remains considerably smaller than the other two genera. Ringicardium Fischer, 1887, mainly used in the literature for species now referred to Procardium gen. nov., is an objective Type of Bucardium (La Perna 2016).
A third related species group, distinct from Procardium gen. nov., Cardium, and Bucardium, but of uncertain generic position, includes some species from the late Oligocene and early Miocene of Europe (La Perna 2016). The best known of them is “Cardium” burdigalinum Lamarck, 1819 from the Burdigalian of the Aquitaine Basin (La Perna 2016: figs. 6, 7). These species have thin, slightly gaping shells, 22–33 well developed radial ribs with rather narrow interspaces and attain a maximum size of about 75 mm. The anterior ribs carry elongate, flattish tubercles or small conical scales similar to those of Procardium gen. nov., and the posterior ribs are spiny and protuberant. The cardinal teeth in the right valve are largely fused with a deep socket below; the anterior lateral socket is not significantly excavated and the narrow hinge plate is moderately curved. In the right valve, the anterior and posterior ventral lateral teeth are approximately equidistant from the cardinals (Fig. 2D). Based on shell characters, these species seem to be significantly closer to Procardium gen. nov. than to other cardiids, but we regard them as representatives of a separate genus.
Acanthocardia Gray, 1851 is a more distantly related European genus, with hardly or non-gaping shells, partly connected cardinal teeth in the left valve, a thicker shell and lacking a secondary riblet in the interspaces, in addition to other notable differences (La Perna and D’Abramo 2009, 2013).
An overview of the discriminating shell characters of Cardium, Bucardium, and Procardium gen. nov. is given in Table 1, while differences in the hinge regions between the three genera and “C.” burdigalinum are shown in Fig. 2.
Stratigraphic and geographic range.—Early Burdigalian or Aquitanian (early Miocene) to Recent. Miocene: Proto-Mediterranean, Western and Central Paratethys, NE Atlantic (Franco-Iberian coast to North Sea Basin); Plio-Pleistocene: southern Spain and Mediterranean; Recent: extreme southwestern Mediterranean and W Africa.
Table 1. Morphological differences between Bucardium, Procardium gen. nov., and Cardium.
| |
Bucardium |
Procardium gen. nov. |
Cardium |
|
Shape |
• rounded • slightly to moderately gaping • shell moderately solid to solid |
• oval, with slight posterior truncation • strongly gaping • shell very thin |
• oval, with slight posterior truncation • moderately gaping • shell moderately solid |
|
Size |
• L to 65 mm
(fossil); 50 mm |
• L to 150 mm
(fossil); 110 mm |
• L to 135 mm |
|
Ribbing |
• 23–28 radial ribs • well developed, rounded-triangular • posterior ribs
strongly protuberant, • sculpture: lacking
(anterior), • interspaces small, no radial threads • shell resorption absent |
• 14–24 radial ribs • well developed,
rounded- • posterior ribs
protuberant, • sculpture:
horseshoe-shaped • interspaces very
wide, often • shell resorption present |
• 14–17 radial ribs
(anteriormost • very low
quadrangular, with • posterior ribs hardly protuberant • sculpture: absent,
only a small and • interspaces rather
wide, never with • shell resorption present |
|
Hinge |
• cardinal teeth largely fused • cardinal teeth subequal in height |
• cardinal teeth separated • cardinal teeth subequal in height |
• cardinal teeth separated • anterior cardinal
higher than |
|
Hinge |
• anterior cardinal
tooth solid, strongly • posterior cardinal tooth small • anterior cardinal
tooth positioned |
• anterior cardinal
tooth elongate, • posterior cardinal tooth very small • anterior cardinal
tooth positioned |
• anterior cardinal tooth pointed • posterior cardinal
tooth well • anterior cardinal
tooth positioned |
Procardium indicum (Lamarck, 1819)
Figs. 1, 2A.
1819 Cardium indicum sp. nov.; Lamarck 1819: 4.
1844 Cardium hians Brocchi, 1814; Reeve 1844: pl. 5: 27 (non C. hians Brocchi, 1814).
1866 Cardium darwini; Mayer 1866: 69–71 (nomen novum for C. indicum Lamarck, 1819).
1950 Ringicardium schoelleri; Magne 1950: 123 (nomen novum for C. darwini Mayer, 1866).
1964 Cardium delpeutum sp. nov.; McGill 1964: 4, unnumbered figure.
1977 Cardium (Cardium s.s.) indicum Lamarck, 1819; Fischer-Piette 1977: 112, pl. 10: 4a, b.
2004 Cardium indicum Lamarck, 1819; Hylleberg 2004: 843, unnumbered figures.
Material.—Holotype of Cardium indicum Lamarck, 1819, MNHN-IM-2000-25143, one valve from “l’Ocean des grandes Indes” [= Indian Ocean: erroneous]. ZMA.MOLL. 3318, two valves from Algier, Algeria. ZMA.MOLL.210347 (Hessel collection), two shells from Agadir, Morocco. RMNH.MOL. 119659 (alive), one shell from Atlantic Ocean (depth 38–41 m), Banc d’Arguin, Mauritania. RMNH.5006726 (ter Poorten collection), one shell from Algeria. JJTP3288, one shell from Mediterranean Sea (dredged 300–380 m), Cherchell, Algeria. JJTP1923, one valve from Agadir, Morocco (from fishermen). JJTP3442, one shell, JJTP45, one shell, from Senegal. JJTP 2075, one shell from Dakar, Senegal. JJTP443, one shell from Mbour, Senegal. All Recent material.
Description.—Shell large (L 75–115 mm), slightly longer than high, almost equilateral, relatively thin, inflated, moderately gaping posteriorly. Umbo slightly prosogyrate. Anterior and ventral margins rounded, posterior margin truncated and nearly straight. Shell bearing 20–24 primary radial ribs, on anterior half each interspace with a rather well defined, thin secondary riblet. Median primary ribs weakly delimited, rather broad, low rounded to weakly triangular and about as wide as interspaces. Anterior ribs prominent with roof tile-shaped, ventrally hollow scales, placed on rib top. Cross-section of anterior and median ribs trapezium-shaped, with a radial groove on the rib tops. Ribs on postero-medial slope asymmetrical in cross section, ultimately flattened posteriorly, with sculpture on posterior rib flanks gradually developing into close-set, pointed, posteriorly directed spines. Rib impressions clearly visible from within. Hinge plate rather narrow and slightly sinuous, hinge angle very large. Hinge and ligament: as for genus. Lunule not well defined, dorsal margin slightly raised anterior of umbo. Exterior colour dirty white, with commarginally arranged brownish areas, posteriorly pink or purple, except for white spines. Rusty-brown deposit present on interspaces of postero-medial part. Interior colour white with traces of cream, posterior margin purple.
Remarks.—As remarked by some authors (e.g., Mayer 1866; Fischer-Piette 1977; Hylleberg 2004), Cardium indicum is not from the Indian Ocean, in spite of the original mention of “l’Ocean des grandes Indes” by Lamarck (1819). The type material (MNHN-IM-2000-25413), a left valve illustrated by Fischer-Piette (1977: pl. 10: 4a, b) and Hylleberg (2004: 843, unnumbered figures), should be considered as holotype because Lamarck (1819) mentioned the presence of only one valve in his description.
Mayer (1866: 69–71), explicitly referring to the citation of C. indicum by Lamarck (1819: 18), Lamarck et al. (1835: 390, footnote) and to the figure of C. hians by Reeve (1844: pl. 5: 27), proposed Cardium darwini as a replacement name for C. indicum, because of the wrong specific attribute used by Lamarck (“Le nom de C. indicum impliquant une contre-vérité, les règles de la nomenclature […] m’ont obligé a changer cette dénomination”). As further discussed, the description of C. darwini was based on a distinct Miocene species, mistaken by Mayer (1866) for the living C. indicum, most probably due to the inaccurate, somewhat misleading illustration of the Recent species by Reeve (1844). However, C. darwini remains nominally a Type of C. indicum (ICZN 1999: Art. 72.7). Cardium delpeutum McGill, 1964, described from off Luanda, Angola is another Type of P. indicum (Ghisotti 1974).
Stratigraphic and geographic range.—Procardium indicum is essentially a tropical West African species, whose distribution ranges from Angola to Morocco, with a few deep water records from Tunisia and Algeria, in the westernmost Mediterranean (Ghisotti 1971; Fischer-Piette 1977; Delongueville and Scaillet 2011; Dailianis et al. 2016). A Canary Islands record (Gómez Rodríguez and Pérez Sánchez 1997) is based on a sole valve found in material dredged for artificial beach construction and needs confirmation. The species is mostly trawled or dredged, with deep water records up to 300–380 m. The only examined live taken specimen is from 38–41 m, collected from muddy sand in Mauritania.
Some old records from other Mediterranean areas (e.g., Toulon, Naples, Corinth) were discussed by Ghisotti (1971) and Fischer-Piette (1977). If not based on misidentified reworked fossil shells (admittedly unlikely, because of shell fragility), these records could represent the occurrence of scattered relict populations of P. indicum throughout the Mediterranean.

Fig. 1. Cardiid bivalve Procardium indicum (Lamarck, 1819), RMNH.5006725 (ter Poorten collection), Recent, Algeria. Articulated shell in internal (A, C), external (B, D), and umbonal (E) views, with detail of anterior sculpture (F). Scale bars: A–E, 50 mm; F, 20 mm.
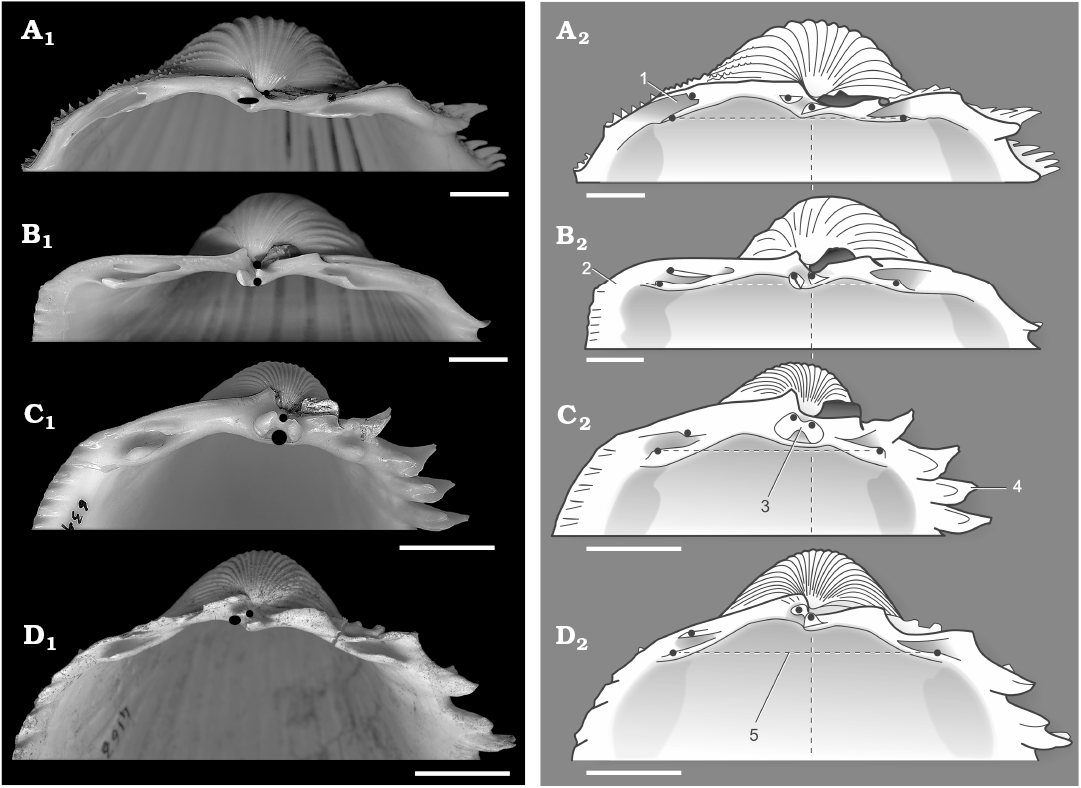
Fig. 2. Hinge region of the right valve of Cardiinae type species and a related “Cardium” species. A. Procardium indicum (Lamarck, 1819), RMNH.5006726 (ter Poorten collection), Recent, Algeria. B. Cardium costatum Linnaeus, 1758, RMNH.5006725 (ter Poorten collection), Recent, Angola. C. Bucardium ringens (Bruguière, 1789), RMNH.5006724 (ter Poorten collection), Recent, Senegal. D. “Cardium” burdigalinum (Lamarck, 1819), RGM.1309989 (ter Poorten collection), France, Léognan, Le Coquillat, early Miocene. Solid circles and ovals indicating positions of the tips of the teeth (A2–D2) and the positions and shapes of the cardinal teeth of the opposite valve (A1–D1). Note extremely hollowed anterior lateral socket in Procardium gen. nov. (1), angular antero-dorsal margin in Cardium (2), largely fused cardinals (3), strongly protuberant posterior ribs in Bucardium (4), and anterior and posterior ventral lateral teeth about equidistant from cardinals in “Cardium” (5). Scale bars 10 mm.
Procardium diluvianum (Lamarck, 1819)
Figs. 3, 4.
1814 Cardium hians sp. nov.; Brocchi 1814: 508, pl. 13: 6 (non C. hians Lightfoot, 1786 [nomen nudum], nec Spengler, 1799).
1819 Cardium diluvianum sp. nov.; Lamarck 1819: 18.
1908 Cardium (Ringicardium) hians var. rotundata; Cerulli Irelli 1908: 24, pl. 4: 7 (non Cardium rotundatum Carpenter, 1857).
1952 Cardium hians Brocchi, 1814; Rossi Ronchetti 1952: 73–75, fig. 28a–d.
2004 Cardium diluvianum Lamarck, 1819; Hylleberg 2004: 920, unnumbered figure.
2014 Cardium paleobrocchii; Hylleberg 2014: 43, figs. 8, 9B (nomen novum for C. hians Brocchi, 1814).
Material.—Syntype of Cardium hians Brocchi, 1814, MSNM i13317, one shell from Pliocene, northern Italy. Holotype of Cardium diluvianum Lamarck, 1819, MNHN. F.A50147 one valve from Pliocene, Siena, Italy. MRSN BS.131.03.001, two valves, MRSN BS.131.03.001/02, one valve, MRSN BS.131.03.001/03, three valves; from Pliocene, Asti Hills, Italy; MRSN BS.131.03.001/01, one valve from Pliocene, Masserano, Italy. MPUR no registration number (unnumbered Cerulli Irelli collection), one valve from Early Pleistocene, Farnesina, Italy. NHMW1836.XII.895, two fragments of one valve, NHMW1862.II.22, one shell; from Pliocene, Castell’Arquato, Italy. NHMW 1855.I.618, one valve, NHMW1855.I.619, one valve, NHMW 1855.I.620, one valve, from Early Pleistocene, Monte Mario, Italy. NHMW1877.XVIII.115, one valve from Pliocene, Valle Andona, Italy. NHMW1853.XXXVI.32, one valve, NHMW 1852.XXXII.143a, one valve, from Pliocene, Rhodes, Greece. RGM.794093 (van Nieulande collection), one valve, JJTP3383, one valve (nine fragments), JJTP2073, two valves, JJTP1001, two fragments, PH 42439, three valves; from Zanclean (Pliocene), Bonares, Spain. RGM.48583 (van Nieulande collection), one valve, RGM.794096 (van Nieulande collection), one shell, RGM.48582 (van Nieulande collection), one shell (one valve), JJTP1657, one fragment, PH 40489, one valve; from Piacenzian (Pliocene), Castell’Arquato, Italy. RGM.794097 (van Nieulande collection), one valve, RGM.794101 (van Nieulande collection), one valve; from Pliocene, Siena, Italy. USNM 82258, one valve from Pliocene, Asti, Italy. JJTP4736, one valve from Pliocene, Castelfiorentino, Italy. MPUB2017/1A, ca. 30 fragments from Early Pleistocene, Gravina in Puglia, Italy. MPUB 2017/1B, four valves from Pliocene, Cerignola, Italy. MNHN F.A50669, one fragment from Zanclean (Pliocene), Pichegu, France. EK uncatalogued specimen, two valves from Pliocene, Tétouan, Morocco. PH 40632, one valve from Pliocene, Belveglio, Italy. JV uncatalogued specimen, one valve from Piacenzian (Pliocene), Montezago, Italy.
Description.—Shell large (L up to 125 mm), slightly longer than high, almost equilateral, relatively thin, inflated, moderately gaping posteriorly. Umbo slightly prosogyrate. Anterior and ventral margins rounded, posterior margin truncated and nearly straight. Shell bearing 17–21 primary radial ribs, on anterior half each interspace with a weakly defined, rather broad secondary riblet. Median primary ribs sharply delimited, broad, high rounded and about as wide as interspaces. Anterior ribs prominent with conical, ventrally hollow scales, placed on rib top. Cross-section of anterior and median ribs trapezium-shaped, with a radial groove on rib tops. Ribs on postero-medial slope asymmetrical in cross section, ultimately flattened posteriorly, with sculpture on posterior rib flanks gradually developing into pointed, posteriorly directed spines. Rib impressions clearly visible from within. Hinge plate rather narrow and slightly sinuous, hinge angle very large. Hinge and ligament: as for genus. Lunule not well defined, dorsal margin slightly raised anterior of umbo.
Remarks.—Cardium hians Brocchi, 1814 is a primary junior homonym of C. hians Spengler, 1799 (Lamy 1941; Smith 1945; Fischer-Piette 1977; Hylleberg 2004), the latter being an objective junior Type of Fulvia aperta (Bruguière, 1789) by selection of the same specimen as lectotype for both taxa (Vidal 1994). The next available name, which has been mostly disregarded in the literature, is Cardium diluvianum Lamarck, 1819.
The description of C. diluvianum by Lamarck (1819: 18) was based on a fossil from Siena (Tuscany) collected by Georges Cuvier. To the short description “C. testâ cordatâ, antice angulatâ; costis 14, distantibus, convexis; vulvâ elevatâ, subcarinatâ”, Lamarck (1819) added the size of 80 mm. Unfortunately, the holotype, first illustrated by Hylleberg (2004: 920, unnumbered figure), is a poorly preserved left valve (Fig. 3B), apparently with at least 17–18 ribs.
Brocchi (1814: 508, pl. 13: 6) described C. hians from the areas of Piacenza, Siena and Asti, northern Italy, with the following diagnosis: “Testa subcordata tumida, costis 17 distantibus, antice depressis, aculeatis, postice tuberculis raris cochleariformibus instructis, valvis anterius hiantibus, margine hiatus profundissime serrato”. The figured syntype of C. hians, redescribed by Rossi Ronchetti (1952: 73–75, figs. 28a–d) who erroneously regarded it as the holotype, is an articulated shell bearing 17 ribs on the left valve and 18 on the right one (Fig. 3A).
The Typey between C. hians and C. diluvianum is based on the following considerations: (i) both types share a comparable size, rib morphology and number of ribs; (ii) both species were described from fine-grained deposits of Pliocene age, usually referred to as Argille Subappennine, widely cropping out in northern Italy. Study material and literature data (de Stefani 1874; de Stefani and Pantanelli 1879; Chirli 2016: 66, pl. 12: 1–4) confirm the occurrence of this large cardiid in the area of Siena, in Zanclean-Piacenzian deposits (Bossio et al. 2002).
The confusion between this fossil species and the living one was initiated by Lamarck (1819: 4), who suggested that the latter might appear to be the “analogue vivant” of C. hians. Consequently, Lamarck et al. (1835: 390, footnote) regarded C. indicum as a junior Type of C. hians, an opinion followed by many subsequent authors (Petit 1840; Weinkauff 1862; Smith 1945; Ghisotti 1971; Van Aartsen and Giannuzzi Savelli 1991; etc.). Recently, Hylleberg (2014) reported slight morphometric differences between C. hians and C. indicum, in addition to the occurrence of spines on the postero-dorsal margin in the fossil species. He thus kept C. hians distinct from the living species, proposing the replacement name C. paleobrocchii for the former.
Indeed, the fossil species and the living one are here considered distinct, Cardium paleobrocchii being therefore another Type of Procardium diluvianum, but the differences between the two species are mostly represented by sculpture (Table 2). The living species has a range of 20–24 ribs vs. 17–21 in P. diluvianum; the scales on the anterior slope of P. diluvianum are markedly funnell-shaped or broadly horseshoe-shaped (Figs. 4B, 16G), whereas they are more erect and rectangular, shingle-shaped in P. indicum (Figs. 1F, 16H). In addition, the radial ribs of P. diluvianum are broader, more pronounced and sharply delimited (Fig. 16G) and the secondary riblets are very weakly developed (Figs. 4A3, 16G), whereas in P. indicum the radial ribs are poorly delimited and the secondary riblets are more distinct (Figs. 1F, 16H). Up to five spines can be present on the postero-dorsal margin of P. diluvianum, as observed by Hylleberg (2014), but this character is variable, spines being distinct to almost absent. No appreciable differences in the hinge occur between the two species. The maximum recorded size is 125 mm in P. diluvianum, as can be extrapolated from Brambilla (1976: pl. 28: 2), while P. indicum is smaller (Table 2).
Table 2. Comparative shell characters for Procardium taxa. Lm, maximum shell length in the study material (* estimated); Rn, number of radial ribs (excluding secondary riblets); Rw/Ri, rib width to interspace width ratio on median shell region; median ribs, characters of ribs on median shell region; radial groove, characters of rib groove on median shell region; posterior gape, width of posterior shell gape; equilaterality, degree of shell equilaterality; granulations, occurrence and distribution of granulose microsculpture in radial interspaces.
| |
Lm [mm] |
Rn |
Rw / Ri |
Median ribs |
Radial groove |
Posterior gape |
Equilaterality |
Granulations |
|
Procardium |
115 |
20–24 |
~ 1:1 |
rather broad, low rounded to weakly triangular, not well delimited |
weakly developed |
moderately wide |
almost equilateral |
lacking |
|
Procardium |
125 |
17–21 |
~ 1:1 |
broad, high rounded, sharply delimited |
well developed |
moderately wide |
almost equilateral |
lacking |
|
Procardium |
85 |
15–19 |
~ 1:1 |
broad, low rounded, rather well delimited |
well developed |
moderately wide |
almost equilateral |
lacking |
|
Procardium |
144 |
14–17 |
~ 1:4 |
thin, high triangular, vaguely delimited on median-posterior part |
lacking |
wide |
moderately to strongly inequilateral |
lacking |
|
Procardium |
152 |
16–18 |
1:3–1:4 |
thin, high triangular, sharply delimited |
lacking |
very wide |
strongly inequilateral |
lacking |
|
Procardium |
~ 130 |
16–17 |
1:3–1:4 |
thin, rounded triangular, not well delimited |
lacking |
moderately wide |
weakly inequilateral |
poorly defined and very small |
|
Procardium |
95* |
19–24 |
1:5–1:6 |
very thin, rather high triangular, sharply delimited |
lacking |
very moderate |
? |
dense, forming well developed commarginal bands |
|
Procardium |
92 |
18–19 |
1:3–1:4 |
thin, high triangular, sharply delimited |
lacking |
moderately wide |
weakly inequilateral |
often forming commarginal bands |
Some variability, apparently unrelated to stratigraphic or geographic distribution, is observed with regard to the strength of the radial sculpture (e.g., Fig. 4A2, D2), with rare cases of unusually wide and strong ribs (Fig. 3G). Cerulli Irelli (1908: 24, pl. 4: 7) proposed Cardium (Ringicardium) hians var. rotundata, with more equilateral and equidimensional shell, the posterior margin not sloping, and 17–18 ribs, instead of 19–20, based on Calabrian (Early Pleistocene) material from Rome. The single valve present in the Cerulli Irelli collection (Fig. 3C) is similar to that illustrated as C. (R.) hians var. rotundata and provides no evidence for keeping it distinct. C. (R.) hians var. rotundata is therefore included in the Typey of P. diluvianum.
All records of C. hians from the Miocene of Europe were based on misidentifications. Conversely, all records those from the Pliocene and Pleistocene of Italy (Sacco 1899: 42, pl. 10: 11–13; Cerulli Irelli 1908: 24, pl. 4: 5–7; Gignoux 1913; Pavia and Demagistris 1971: 99, pl. 3: 1; Brambilla 1976: p. 110, pl. 28: 2; Chirli 2016: 66, pl. 12: 1–4), the Pliocene of southern France (Fontannes 1882: 80, pl. 5: 1; Chirli and Richard 2008: 96, pl. 21: 6), southern Spain (Andrés 1987: 112, pl. 3: 1, 2), Greece (Syrides 1995) and Cyprus (Moshkovitz 2012), should be referred to P. diluvianum. The studied material is representative of this distribution and also includes the early Pliocene of Tétouan, at the Mediterranean side of northern Morocco.
Stratigraphic and geographic range.—In the early Pliocene–Early Pleistocene interval, P. diluvianum occurred from the Atlantic Guadalquivir Basin in southern Spain to the eastern Mediterranean. Most probably, it also ranged further to south, along the coast of West Africa, similarly to the modern distribution of P. indicum.

Fig. 3. Cardiid bivalve Procardium diluvianum (Lamarck, 1819). A. Syntype of Cardium hians Brocchi, 1814, MSNM i13317, Pliocene, northern Italy (photo Giorgio Teruzzi); external (A1) and umbonal (A2) views. B. Holotype of Cardium diluvianum Lamarck, 1819, MNHN.F.A50147, Pliocene, surroundings of Siena, Italy; external view (photo Jacques Mouchart, http://coldb.mnhn.fr/catalognumber/mnhn/f/a50147). C. MPUR no registration number (unnumbered Cerulli Irelli collection), Early Pleistocene, Farnesina, Italy; external view. D. RGM.794093 (van Nieulande collection), Zanclean (Pliocene), Bonares, Spain; external view. E. RGM.48582.a, Piacenzian (Pliocene), Castell’Arquato, Italy; umbonal view. F. MRSN BS.131.03.001, Pliocene, Asti Hills, Italy; external view. G. RGM.48583, Piacenzian (Pliocene), Castell’Arquato, Italy; external view. Scale bar 50 mm.

Fig. 4. Cardiid bivalve Procardium diluvianum (Lamarck, 1819) from Italy. A. MPUB 2017/1B, Pliocene, Cerignola; internal (A1) and external (A2) views, detail of sculpture (A3). B. MPUB 2017/1A, Pliocene, Cerignola; detail of anterior sculpture. C. MPUB 2017/1A, Early Pleistocene, Gravina in Puglia; detail of posterior spines (inner view). D. PH 40489, Piacenzian (Pliocene), Castell’Arquato; internal (D1) and external (D2) views. Scale bars: A1, A2, D1, D2, 50 mm; A3, 10 mm; B, C, 5 mm.
Procardium avisanense (Fontannes, 1879)
Figs. 5, 6.
1878 Cardium avisanense; Fontannes 1878: 499, 502 (nomen nudum).
1879 Cardium avisanense sp. nov.; Fontannes 1879: 44.
1903 Cardium hians var. recta; Dollfus et al. 1903: 44, pl. 16: 4–6.
1912 Cardium (Ringicardium) hians var. recta Dollfus, Cotter, and Gomes, 1903; Cossmann and Peyrot 1912: pl. 21, figs. 3–5.
1950 Ringicardium avisanense (Fontannes, 1879); Magne 1950: 124.
Material.—RGM.794104, one shell, one valve, 16 fragments, RGM.794106 (van Nieulande collection), three shells, three valves; from Tortonian (Miocene), Cacela Velha Portugal. RGM.794107 (Visser collection), one valve, some fragments, RGM.794108 (Visser collection), one valve, one fragment; from Serravallian (Miocene), Salles “l’Argileyre”, France. PH19296, four valves from from Serravallian (Miocene), Salles “Le Minoy”, France. PH20376, two shells, four valves from Tortonian (Miocene), Cacela Velha, Portugal. PH20377, three shells, seven valves from Tortonian (Miocene), Fábrica/Cacela Velha, Portugal. JJTP4860, three fragments from Tortonian (Miocene), Lacem, Portugal.
Description.—Shell rather large (L up to 85 mm), slightly longer than high, almost equilateral, relatively thin, inflated, moderately gaping posteriorly. Umbo prosogyrate. Anterior and ventral margins rounded, posterior margin truncated and nearly straight. Shell bearing 15–19 primary radial ribs, on anterior half each interspace with a weakly defined, rather broad secondary riblet. Median primary ribs rather well delimited, broad, low rounded and about as wide as interspaces. Anterior ribs prominent with conical-rounded, ventrally hollow scales, placed on rib top. Cross-section of anterior and median ribs trapezium-shaped, with a radial groove on rib tops. Ribs on postero-medial slope strongly asymmetrical in cross section, ultimately flattened posteriorly, with sculpture on posterior rib flanks gradually developing into pointed, posteriorly directed spines. Rib impressions clearly visible from within. Hinge plate rather narrow and slightly sinuous, hinge angle very large. Hinge and ligament: as for genus. Lunule shallowly excavate, dorsal margin slightly raised anterior of umbo.
Remarks.—The original record of Cardium avisanense is from the Miocene of the Rhone Valley, southern France (Fontannes 1879: 44). The shell shape was described as “généralement un peu plus large que haute, légèrement oblique et inéquilatérale, globuleuse, largement arrondie en avant, subtronquée et comprimée en arrière”, while the radial sculpture was said to consist of 16–18 ribs, “assez élevées, presque arrondies dans la moitié supérieure, subquadrangulaires en avant et vers le bord palléal, divisées en deux par une strie profonde au milieu de laquelle se dressent, à des intervalles assez rapprochés et peu réguliers, de petites écailles”, with a poorly distinct rib sometimes present in the interspaces.
The species was again discussed by Fontannes (1882), while dealing with C. hians from the Pliocene of the Rhone Valley, remarking the separation of the Miocene species C. avisanense from the Pliocene one.
No precise locality was given in the original description, but the species was said to occur in marly-sandy deposits cropping out in the area of Mont Luberon, at Visan and Cucuron together with Pecten vindascinus Fontannes, 1878, up to the marly deposits with Ancillaria glandiformis Lamarck, 1810 at Cabrières d’Aiguës. From Cucuron and Cabrières d’Aiguës, the species had been reported, as a nomen nudum, by Fontannes (1878). Based on the original indications and on literature data on the stratigraphy of the Mont Luberon area (Demarcq 1960; Courme-Rault and Dubar 2011), the original records of C. avisanense were from Tortonian deposits.
Fontannes (1879: 45) noted several differences between C. avisanense and C. hians: “Elle [C. avisanense] en diffère par un contour palléal moins oblique, plus largement arrondi, par un crochet moins porté en avant, par un côté postérieur plus brusquement tronqué, laissant probablement un écartement beaucoup moins accusé que dans le type subapennin; les côtes sont moins élevées, plus larges, plus arrondies de chaque côté du sillon; celui-ci est plus étroit, moins profond et les épines sont notablement plus petites”.
The type material of C. avisanense was not located, but the careful description and comparison with C. hians by Fontannes (1879) leave no doubts about the identity of this species, which was studied on material from the late Miocene of the Algarve Basin, southern Portugal (Figs. 5, 6A, B, E) and from the Serravallian of the Aquitaine Basin (Salles, Le Minoy) (Fig. 6C, D). The Portuguese material is from Cacela (now Cacela Velha) and from the nearby locality of Fabrica, where the richly fossiliferous Cacela Formation, of Tortonian–Messinian age (Santos and Mayoral 2007; Pais et al. 2000; Studencka and Zieliński 2013) crops out. The poorly preserved shell from this formation, illustrated by Studencka and Zieliński (2013: fig. 4E–G) as C. indicum, can be referred to Procardium avisanense.
As remarked in the original description, P. avisanense is markedly similar to P. diluvianum. Both species share relatively wide radial ribs, convex in cross-section, well delimited, bearing a distinct median groove, from which conical scales rise, as well as poorly distinct secondary riblets (Table 2; Fig. 16F, G). The distinctive characters of P. avisanense from P. diluvianum are: (i) lower number of ribs (15–19); (ii) smaller size (L up to 85 mm); (iii) less oblique and less inequilateral shape; (iv) ventral margin more regularly convex, not or only weakly expanded postero-ventrally. Fontannes (1879) remarked further differences from C. hians, namely a sharper posterior truncation and less anterior umbo. However, in both species the sharpness of the posterior truncation is rather variable and the umbo is practically subcentral. More properly, in P. avisanense the anterior side is slightly larger and more convex than in P. hians, giving the impression of a less anterior umbo. The maximum size is slightly exceeding 80 mm, clearly smaller than P. diluvianum (Table 2).
A few specimens from the Tortonian of Cacela have particularly straight ribs, with slightly narrower interspaces (Fig. 5D, E). The shell is more inflated and with a narrower posterior gape, while the shape is more equilateral, largely lacking the typical obliquity of the genus. The rib number (16–17) is within the range of the other specimens from the same locality. At first, these shell characters seem to point to a distinct species, corresponding to Cardium hians var. recta Dollfus, Cotter, and Gomes, 1903, shortly described “Nous proposons pour les exemplaires figurés la variété recta, largeur 90 mm., hauteur 80. Les côtes sont assez serrées, presque droites, la forme générale moins brièvement tronquée en avant” and nicely illustrated from the Tortonian of Cacela (Dollfus et al. 1903: 44, pl. 16: 4–6). Probably, the “straight” ribs suggested the specific attribute rectum (= straight, Latin). However, these specimens seem to fall within the variability of P. avisanense, considering that other specimens occur (Fig. 5F), which are intermediate in shape and sculpture between the “typical” form with oblique ribs and the specimens matching C. hians var. recta. After all, similar variations also occur in P. diluvianum (Fig. 3D, G). Cardium hians var. recta is therefore considered a Type of P. avisanense, as also proposed by Magne (1950). The records of C. hians var. recta from the Tortonian of Cacela by Chavan (1940) are deemed to be based on P. avisanense.
The specimens from the Serravallian of the Aquitaine Basin (Fig. 6C, D) do not differ significantly from the Tortonian ones. It is also worth noting that these specimens show a certain variability in shape and radial ribbing, paralleling to some extent that of the Tortonian material. Cardium (Ringicardium) hians and C. hians var. recta reported by Cossmann and Peyrot (1912: 110, pl. 21: 3–5) from Salles are referred to P. avisanense.
Dollfus et al. (1903) mentioned the occurrence at Cacela of large fragments with wide radial interspaces and secondary riblets, together with fragments with long, sharp spines. These shell characters point to a distinct species, misidentified as the “typical” form of “C. hians”, namely Procardium kunstleri (Cossmann and Peyrot, 1912), which does occur in the Tortonian of Cacela. Also the records of C. (Ringicardium) hians mut. recta by Zbyszewski (1957: 135, pl. 5: 40, 41) from the Burdigalian of Portugal, based on internal moulds, point to a distinct species with secondary riblets, corresponding to P. kunstleri or to a similar species. For this reason, the wide distribution in Portugal reported by Dollfus et al. (1903), ranging from the Burdigalian to the Tortonian, is considered doubtful and in need of confirmation.
The record of C. hians from the Tortonian of Tuscany, northern Italy by Tavani and Tongiorgi (1963: 25, pl. 25: 10, 11), based on internal moulds, may represent P. avisanense or a closely similar species with convex, grooved radial ribs, but a clear identification is prevented by the poor preservation. Another Tortonian record from northern Italy (Venzo and Pelosio 1963: 174, pl. 25: 15), also based on internal moulds, may refer to a distinct Procardium species, markedly oblique in shape and with 16–17 convex ribs, probably grooved.
Stratigraphic and geographic range.—Procardium avisanense is known from the Serrravallian of the Aquitaine Basin and the Tortonian of the southern Rhone Valley and Algarve Basin, thus including the Franco-Iberian Atlantic coast and the Western Mediterranean. A wider, late Miocene distribution in the Mediterranean is possible.

Fig. 5. Cardiid bivalve Procardium avisanense (Fontannes, 1879) from Tortonian (Miocene) of Portugal, Cacela Velha (A, D, E) and Fabrica (B, C, F). A. RGM.794106.a (van Nieulande collection), internal (A1), external (A2), and umbonal (A3) views. B. PH 20377, internal (B1) and external (B2) views. C. PH 20377, umbonal view. D. RGM.794104.a, internal (D1) and external (D2) views. E. RGM.794104.b, external (E1) and umbonal (E2) views. F. PH 20377, external (F1) and internal (F2) views. Scale bar 50 mm.

Fig. 6. Cardiid bivalve Procardium avisanense (Fontannes, 1879) from Tortonian (Miocene) of Cacela Velha, Portugal (A, B, E) and Serravallian (Miocene) of France, Salles, “Le Minoy”(C) and “l’Argileyre” (D). A. PH 20376, external view. B. PH 20376, external (B1) and internal (B2) views. C. PH 19296, internal (C1) and external (C2) views. D. RGM.794107.a, internal (D1) and external (D2) views. E. RGM.794104.c, detail of anterior sculpture. Scale bars: A–D, 50 mm; E, 10 mm.
Procardium kunstleri (Cossmann and Peyrot, 1912)
Figs. 7–9.
1873 Cardium darwini Mayer, 1866; Fischer and Tournouër 1873: 45, pl. 20: 9 (non C. darwini Mayer, 1866).
1912 Cardium (Ringicardium) kunstleri sp. nov.; Cossmann and Peyrot 1912: 494, pl. 21: 28.
1950 Ringicardium kunstleri (Cossmann and Peyrot, 1912); Magne 1950: 123.
Material.—RGM.794109, one valve from Serravallian (Miocene), Salles “Moulin Débat”, France. RGM.794105, four fragments from Tortonian (Miocene), Cacela Velha, Portugal. RGM.794103, three moulds from Serravallian (Miocene), Akbogazi, Turkey. RGM.1309992 (Kaptein collection), one valve from Serravallian? (Miocene), Gironde, France. JJTP4774, four fragments from Serravallian? (Miocene), Gironde, France. JJTP4880, one valve from Serravallian? (Miocene), Gironde, France. PH11898, three valves from Serravallian (Miocene), Salles, France. PH20375, one shell, two valves, six fragments, PH20400, one mould, from Tortonian (Miocene), Cacela Velha, Portugal. EK unnumbererd specimen, one valve, five fragments from Serravallian? (Miocene), Gironde, France. P. aff. kunstleri: RGM.1309990 (ter Poorten collection) one valve from Zanclean (Pliocene), Lucena del Puerto, Spain; PH42438, one shell, one valve from Zanclean (Pliocene), Bonares, Spain.
Description.—Shell very large (L up to 144 mm), longer than high, strongly inequilateral, relatively thin, inflated, widely gaping posteriorly. Umbo prosogyrate. Anterior margin rounded, ventral margin almost straight, posterior margin truncated and nearly straight. Shell bearing 14–17 primary radial ribs, on anterior half each interspace with a weakly defined, rather broad secondary riblet. Median primary ribs not well delimited, thin, high triangular and about 4 times as small as interspaces. Anterior ribs prominent with horseshoe-shaped, ventrally hollow scales, placed on rib top and upper rib flanks. Cross-section of anterior and median ribs triangular to keel-like. Ribs on postero-medial slope asymmetrical in cross section, ultimately flattened posteriorly, with sculpture on posterior rib flanks gradually developing into pointed, strongly projecting, posteriorly directed spines. Rib impressions clearly visible from within. Hinge plate rather narrow and slightly sinuous, hinge angle very large. Hinge and ligament: as for genus. Lunule shallowly excavate, dorsal margin raised anterior of umbo.
Remarks.—In their study of the late Miocene molluscs from the Cabrières marls (Mont Luberon), Fischer and Tournouër (1873: 45, pl. 20: 9) illustrated a well preserved valve of a cardiid from the Serravallian of Salles (Aquitaine Basin), with the aim of providing a first illustration of Mayer’s Cardium darwini, avoiding to illustrate the poorly preserved material occurring at Cabrières. Instead, they illustrated a distinct Procardium species with a markedly oblique shape and narrow ribs, clearly the same species described, almost 40 years later, as C. (Ringicardium) kunstleri by Cossmann and Peyrot (1912: 494, pl. 21: 28).
The study material from the type area (Gironde, Serravallian) (Fig. 7) matches the syntype from Salles (a left valve) illustrated by Cossmann and Peyrot (1912), who oddly referred to it as “neotype”. The radial sculpture consists of 15 ribs (14–15 in the original description), which are anteriorly and medially narrow and triangular in cross-section. U- or V-shaped scales, mostly worn, are present on the median ribs, while they are larger and conical or horseshoe-shaped anteriorly (Fig. 16D). The radial interspaces are wide, flattish, with rather well defined secondary riblets (Fig. 16D). Procardium kunstleri differs markedly in sculpture from P. indicum, P. diluvianum, and P. avisanense, all with wider, convex ribs, bearing a median groove (Table 2; Fig. 16F–H). Also, the shell of P. kunstleri is more elongate, markedly oblique and with a poorly convex to almost straight ventral margin.

Fig. 7. Cardiid bivalve Procardium kunstleri (Cossmann and Peyrot, 1912) from Serravallian (Miocene) of France. A. RGM.794109, Salles, “Moulin Débat”; internal (A1) and external (A2) views. B. RGM.1309992 (Kaptein collection), Gironde; external view. C. PH 11898, Salles; internal (C1) and external (C2) views. Scale bar 50 mm.
The late Miocene specimens from Portugal (Fig. 8B–D), are very similar to the Serravallian specimens from France, but their posterior spines are particularly long and pointed (Fig. 8C2). In some specimens, the anterior scales are particularly well preserved: they are larger and more widely conical than those of P. diluvianum and P. avisanense (Fig. 8D).
The material from the early Pliocene of Cordoba and Bonares in the Gualdalquivir Basin, southern Spain (Fig. 9), differs from the Miocene specimens with regard to: (i) the higher number of anterior and median ribs (11 ribs in the Pliocene specimens vs. 9 in the Miocene ones); (ii) less projecting triangular ribs on the anterior-median slope (Fig. 16E); (iii) more elevated and sharply bordered posterior ribs; (iv) a stronger posterior keel separating the posterior slope. In other words, the sculptural change from the median to the posterior area is much more pronounced in the Miocene specimens, and accelerated by a keel-like rib, which results in a more angular postero-ventral transition. In addition, the siphonal gape in the Pliocene specimens is very wide (Fig. 9C). Additional material and information is needed to establish the taxonomic status of the early Pliocene specimens, which are here provisionally referred to Procardium aff. kunstleri.

Fig. 8. Cardiid bivalve Procardium kunstleri (Cossmann and Peyrot, 1912) from Serravallian (Miocene) of Salles, France (A) and Tortonian (Miocene) of Cacela Velha, Portugal (B–D). A. PH 11898, external (A1), internal (A2), and umbonal (A3) views. B. PH 20375, internal (B1) and external (B2) views. C. PH 20375, external view (C1) and detail of posterior sculpture (C2). D. PH 20375, detail of anterior sculpture. Scale bars A, B, C1, 50 mm; C2, D, 20 mm.

Fig. 9. Cardiid bivalve Procardium aff. kunstleri (Cossmann and Peyrot, 1912) from Pliocene of Spain. A. RGM.1309990 (ter Poorten collection), Cordoba, Lucena del Puerto; external (A1), internal (A2), and umbonal (A3) views. B. PH 42438, Bonares; internal (B1) and external (B2) views. C. PH 42438, Bonares; internal (C1), posterior (C2), and external (C3) views. Scale bar 50 mm.
The shells of P. kunstleri are large, up to about 125 mm in length in the Aquitaine Basin and 145 mm in the Cacela Basin. However, specimens of Procardium aff. kunstleri from the Guadalquivir Basin are even larger, reaching more than 150 mm in length (Table 2).
Apart from the middle Miocene and early Pliocene records from NE Atlantic, there are some records from the late Miocene of the Mediterranean, unfortunately all based on poorly preserved material. A group of records is from the Tortonian of northern Italy: the internal moulds referred to as Cardium kunstleri by Venzo (1933: 55, pl. 6: 4), Venzo and Pelosio (1963: 174, pl. 25: 16) and Tavani and Tongiorgi (1963: 26, pl. 25: 2, 4) show the main sculptural characters of P. kunstleri, but probably only the last record, from Tuscany, could be tentatively referred to P. kunstleri, while the others seem to differ markedly in shape.
Some literature records and material from the Mut Basin deposits (Fig. 10), cropping out in the area of Karaman, southern Turkey, and ranging in age from Burdigalian to Serravallian (Mandic et al. 2004; Ćorić et al. 2012; Landau et al. 2013), suggest the occurrence of several Procardium species. Scarce shell fragments from the Serravallian of Seyithasan (RGM.1309268, van der Voort collection) and Lale (PH 1634) show close similarities in sculpture with P. kunstleri, but apparently with different direction and curvation of ribs (Fig. 12B–F). Other material from the same area consists of internal moulds (Fig. 10D–G), all of them sharing narrow, wide spaced ribs, but differing from each other in shape. A notably elongate mould with 14 ribs (Fig. 10F), could be identified as Cardium subhians Fischer, 1866 (Fig. 10A), described form the area of Karaman (Fischer 1866: 283–284, pl. 18: 2). A few other moulds (RGM.794103, RGM.1309265, van der Voort collection) are almost equidimensional and equilateral, with straight ribs (Fig. 10E). Another mould is slightly elongate and oblique (Fig. 10D), while another, possibly distinct species, is almost equilateral, but markedly less globose than the others (Fig. 10G). In addition, from the area of Karaman, Toula (1901: 253, pl. 8: 5a, b) described Cardium cilicianum and reported Cardium aff. subhians, both on moulds of articulated shells. The holotype of C. cilicianum (RGM.16498; Fig. 10B) seems unusually elongate posteriorly, while the specimen of C. aff. subhians (RGM.16499; Fig. 10C) is markedly different in shape from the type material of C. subhians. The two moulds reported from the same area by Erünal-Erentöz (1958: 177, pl. 33: 18, 18a, pl. 34: 1) as Cardium (Ringicardium) cf. hians seem to correspond to Cardium aff. subhians, with its subrectangular, oblique shape. Also the internal mould reported by Venzo and Pelosio (1963: 174, pl. 25: 15) as C. hians from the Tortonian of northern Italy is somewhat similar to Toula’s specimen. The internal mould reported by Mandic et al. (2004: fig. 10-1) as Cardium hians danubiana Mayer, 1866 from the Burdigalian of Burunköy, in the Karaman area, could be another distinct species, as discussed below.
Other records of “C. kunstleri”, once again based on internal moulds, are from the Paratethys, namely from the middle Burdigalian of Bern, Switzerland (Rutsch 1928: 144, pl. 8: 36; Pfister and Wegmüller 1998: 476, pl. 10: 1–6) and from the Badenian (Langhian–middle Serravallian) of Poland (Studencka and Studencki 1988: pl. 7: 8). Although these moulds show some overlap in shape and sculpture with P. kunstleri, their definitive identification is not possible.
Stratigraphic and geographic range.—Procardium kunstleri is known from the Serravallian of France (Aquitaine Basin) and the Tortonian of Portugal (Algarve Basin), but a wider distribution (Tortonian of Mediterranean) is possible. Procardium aff. kunstleri is only known from early Pliocene of Spain (Guadalquivir Basin).

Fig. 10. Cardiid bivalve Procardium spp., internal moulds, from Serravallian? (Miocene) of Turkey. A. Syntype of Cardium subhians Fischer, 1866, MNHN.F.B32957, between Ermenek and Bostanesou (photo Peter Massicard, http://coldb.mnhn.fr/catalognumber/mnhn/f/b32957). B. Holotype of Cardium cilicianum Toula, 1901, RGM.16498, Karaman Basin, exact locality unknown; external (B1) and umbonal (B2) views. C. Cardium subhians Fischer sensu Toula, 1901, RGM.16499 (Claessens collection), exact locality unknown, Karaman Basin. D–G. Procardium sp. D. RGM.1309264 (van der Voort collection), 1–2 km E of Lale, Akboğaz. E. RGM.1309265 (van der Voort collection), Seyithasan. F. RGM.1309266 (van der Voort collection), 1–2 km E of Lale, Akboğaz. G. RGM.1309267 (van der Voort collection), slopes ENE of village, Akpınar; external (G1) and umbonal (G2) views. Scale bars: D–F, 50 mm; A–C, G, 25 mm.
Procardium danubianum (Mayer, 1866)
Fig. 11.
1862 Cardium hians Brocchi, 1814; Hörnes 1862: 181–182, pl. 26: 1–5.
1866 Cardium danubianum sp. nov.; Mayer 1866: 71.
1956 Cardium (Ringicardium) hians danubianum Mayer, 1866; Sieber 1956: 216, pl. 3: 11a, b.
2003 Cardium (Bucardium) ringens danubianum Mayer, 1866; Schultz 2003: 519, pl. 75: 2a, b; pl. 76: 9.
Material.—Syntypes of Cardium danubianum Mayer, 1866: NHMW 1853.XVI.44 (lectotype selected herein), 1 shell; NHMW 1853.XVI.43, 1 shell; NHMW 1853.XVI.41, one valve; from middle–late Badenian (Miocene), Enzesfeld, Austria.
Description.—Shell large (L up to 130 mm), longer than high, weakly inequilateral, relatively thin, inflated, widely gaping posteriorly. Umbo prosogyrate. Anterior and ventral margins rounded, posterior margin truncated and nearly straight. Shell bearing 16–17 primary radial ribs, on anterior half each interspace with a weakly defined, rather broad secondary riblet. Median primary ribs not well delimited, rounded triangular and about 3–4 times as small as interspaces. Anterior ribs prominent, nature of scales undefined (largely broken off), placed on rib top. Cross-section of anterior and median ribs rounded-triangular. Ribs on postero-medial slope asymmetrical in cross section, ultimately flattened posteriorly, with sculpture on posterior rib flanks gradually developing into pointed, posteriorly directed spines. Poorly defined microscopic granulations present on parts of interspaces of anterior half of shell. Hinge and interior undefined (hidden in matrix). Lunule not well defined, dorsal margin raised anterior of umbo.
Remarks.—The species reported by Hörnes (1862: 181–182, pl. 26: 1–5) as Cardium hians Brocchi, 1814 from the Miocene of Austria was recognized as a distinct species by Mayer (1866: 71), who proposed for it C. danubianum. The name was made available by reference to Hörnes (1862) (ICZN 1999: art. 12.1), so that the material illustrated by Hörnes (1862) should be considered the type material of C. danubianum. It consists of two articulated shells (NHMW 1853.X.44, Fig. 11A, B) from Enzesfeld. The larger specimen (NHMW 1853.XVI.44, Fig. 11A) is here selected as lectotype (ICZN 1999: art. 74.1), the remaining one becoming paralectotype (NHMW 1853.XVI.43, Fig. 11B).
Both type specimens were also illustrated by Schultz (2003: 519, pl. 75: 2a, b; pl. 76: 9). Apparently, the drawing of right and left hinges reported by Sieber (1956: pl. 3: 11a, b) is from the lectotype, whose valves are strongly shifted, showing in part their poorly preserved hinge. However, Sieber’s illustration seems to be based more on reconstruction than on their actual state of preservation.
The internal mould of a complete shell reported by Schaffer (1910: 66, pl. 30: 5, 6) as Ringicardium hians var. danubiana from Eggenburg is markedly different in shape and sculpture from the type material, appearing more similar to some internal moulds from the Serravallian of Turkey (e.g., Fig. 10E, G). Another internal mould from Eggenburg (NHMW 1869.III.16) seems similar to P. danubianum, but no clear identification is possible.
The type locality Enzesfeld, south-west of Vienna, is one of the fossiliferous localities in the Vienna Basin of middle–late Badenian age (late Langhian–middle Serravallian) (Strauss et al. 2006; Paulissen et al. 2011). The Eggenburg deposits, cropping out north-west of Vienna belong to the North Alpine Foreland Basin and are late Eggenburgian (early Burdigalian) in age (Mandic and Steininger 2003).
According to Mayer (1866), C. danubianum differs from “C. darwini” (i.e., the Miocene species misidentified by Mayer as the living P. indicum, representing P. magnei sp. nov., described below) by its larger umbo and its broader, lower and more closely set ribs, with secondary riblets in the interspaces. Indeed, these are diagnostic features of C. danubianum and “C. darwini”. However, Mayer’s (1866: 71) differential diagnosis was most probably based on more than one species, as he referred to C. danubianum specimens from the area of Ulm, southern Germany and Bern, Switzerland, preserved as internal moulds (“Ce type se retrouve aux environs d’Ulm et de Berne, et on le reconnaît facilement même à l’état de moule”). Evidently, his material originated from the Swiss and German Upper Marine Molasse, in the North Alpine Foreland Basin, of Eggenburgian–Ottnangian (early–middle Burdigalian) age (Heckeberg et al. 2010; Pippèrr 2011; Pfister et al. 2011), but the Procardium specimens known from this formation seem closer to P. kunstleri, as discussed above.
The lectotype, ca 130 mm in length, has 16 ribs in the right valve; the left valve is fragmentary. The median ribs are moderately strong and roughly triangular in cross section. A fairly distinct medial secondary riblet is present in the wide interspaces (Fig. 11A4). Scales on the anterior and median ribs are mostly abraded and apparently funnel-shaped (Fig. 16C), while the posterior spines are mostly lost. The paralectotype, ca. 70 mm long, has the same type of sculpture as the lectotype, with 16 main ribs in the right valve; the left valve is broken. The specimen is almost equidimensional, suggesting marked ontogenetic changes in shape. In both specimens, the shell surface is crossed by particularly well distinct growth striae. A microsculpture is present in the radial interspaces: it consists of indistinct granulations, mostly located on the growth striae, though not regularly aligned on them (Fig. 11B3).
Procardium danubianum is particularly similar to P. kunstleri in sculpture: both species share a relatively low number of ribs, not exceeding 17, which are narrow and roughly triangular in shape, with wide interspaces and well distinct secondary riblets. Some similarities are also seen in shape, both species being somewhat elongate, instead of almost equidimensional as P. indicum, P. diluvianum, and P. avisanense, and with a particularly large umbo.
Sacco (1899: 48, pl. 10: 15) reported C. (Ringicardium) cf. danubianum from Albugnano, “Helvetian” locality in the Turin Hills, northern Italy, now referred to the Langhian (Zunino and Pavia 2009). It is a poorly preserved valve, ca. 75 mm in length, somewhat oblique and with a large, prominent umbo (Fig. 12A). It has 12 or 13 main ribs, and well distinct, rather strong secondary riblets. Because of its poor condition, it was not possible to ascertain the occurrence of granulations. It is possibly a distinct species, apparently similar to P. danubianum. The same author described C. (Ringicardium) hians var. dertogigantea (Sacco 1899: 48, pl. 10: 14), from the Tortonian of Stazzano, Turin Hills, on a shell fragment showing the anteriormost median rib, square in cross section, and the first, asymmetrical anterior rib. Evidently, the poor preservation prevents from identification and var. dertogigantea is considered a nomen dubium.
The record of Cardium hians danubiana by Mandic et al. (2004) from the Burdigalian of southern Turkey, herein reported under P. kunstleri, seems to be based on P. danubianum, but its identification cannot be confirmed because of the poor preservation.
Stratigraphic and geographic range.—Procardium danubianum is known from the late Langhian–middle Serravallian of Austria (Central Paratethys).

Fig. 11. Cardiid bivalve Procardium danubianum (Mayer, 1866) from Badenian (Miocene) of Enzesfeld, Austria. A. Lectotype of Cardium danubianum Mayer, 1866, NHMW 1853.XVI.44, external (A1, A3) and umbonal (A2) views, detail of sculpture (A4). B. Paralectotype, NHMW 1853.XVI.43, external (B1) and umbonal (B2) views, detail of microsculpure (B3). C. NHMW 1853.XVI.41, external view. Photos Oleg Mandic. Scale bars: A1–A3, B1, B2, C, 50 mm; A4, 20 mm; B3, 1 mm.

Fig. 12. Cardiid bivalve Procardium spp. A. Procardium cf. danubianum (Mayer, 1866), MRSN BS.131.03.003 (Bellardi and Sacco collection), Langhian (Miocene), Albugnano, Italy; external view. B–F. Procardium sp., RGM. 1309268 (van der Voort collection), Serravallian (Miocene), Seyithasan, Turkey; external views. Scale bar 30 mm.
Procardium jansseni sp. nov.
Figs. 13, 14.
1925 Cardium (Ringicardium) danubianum Mayer; Kautsky 1925: 36.
1950 Cardium (Ringicardium) hians Brocchi, 1814; Heering 1950: 32.
1972 Cardium (Ringicardium) sp.; Nordsieck 1972: 38, pl. 8: 59.
1975 Bucardium sp.; Bosch et al. 1975: 82, pl. 12: 2.
1984 Cardium (Bucardium) sp.; Janssen 1984: 82, pl. 31: 3–5.
1994 Cardium (Bucardium) sp.; Moths 1994: 67, figs. 1–6.
1999 Cardium (Bucardium) sp.; Wienrich 1999: 240, pl. 57: 3a, b.
2010 Bucardium sp.; Moths et al. 2010: 21, fig. 13, pl. 7: 6, 7.
2016 Bucardium cf. danubianum (Mayer, 1866); Stein et al. 2016: 19, pl. 7: 5.
Etymology: After Arie W. Janssen, former curator of Cainozoic Mollusca at the National Museum of Geology and Mineralogy (now Naturalis Biodiversity Center, Leiden, The Netherlands), in recognition of his extensive contributions to our knowledge of Miocene molluscs of the North Sea Basin.
Type material: Holotype RGM.1309269 (van der Voort collection), top fragment of right valve (26 mm), Paratypes: RGM.225754, fragment of right valve (40 mm), RGM.225755, fragment of right valve (24.5 mm), RGM.225756, two fragments of right valves (largest 34 mm); all from the type locality.
Type horizon: Dark greenish black, variably silty, glauconite sand with many shells. Miste Bed, Breda Formation, lower part of the Aalten Member, Hemmoorian (Langhian), middle Miocene.
Type locality: Miste “Berenschot”, near Winterswijk, province of Gelderland, The Netherlands.
Material.—RGM.794091 (van den Bosch collection), 1 fragment; RGM.1309415, RGM.1309416, RGM. 1309417, RGM.1309418 (all van der Voort collection), 4 fragments; RGM.794092, 3 fragments; RGM.1309991 (ter Poorten collection), 1 fragment, JJTP2071, JJTP2072, 11 fragments; PH36255, 41 fragments; JV unnumbered specimens, > 100 fragments; from Langhian (Miocene), Miste, The Netherlands. RGM.794094 (Rijks Geologische Dienst collection), 3 fragments from Miocene, borehole Beeringen DB15 (B58B0008), De Peel, Beringe, The Netherlands. RGM.794095, Rijks Geologische Dienst collection), 4 fragments from Miocene of De Peel, Baarlo I, The Netherlands. JV unnumbered specimens, 18 fragments from Langhian (Miocene), Werder, Germany.
Diagnosis.—A rather large, inflated Procardium species with 19–24 sharply delimited high triangular radial ribs. Scales on anterior part close-set and also covering the rib flanks. Interspaces very broad with dense and commarginally alligned bands of minute granulations on anterior and median zones.
Description.—Shell large (L up to 95 mm, extrapolated), slightly longer than high (extrapolated), weakly inequilateral, thin shelled, inflated, with a moderately wide posterior gape along whole length of posterior margin. Umbo slightly prosogyrate. Anterior, ventral, and postero-ventral margins rounded; posterior margin weakly rounded and truncated. Shell bearing 19–24 sharply delimited, thin primary radial ribs, on anterior half alternating with a very weakly developed, broad and flattened secondary riblet. On median part, interspaces about 5–6 times wider than primary ribs. Radial ribs tiny and crowded on antero-dorsal slope, thin and rather pointed triangular on anterior and ventral slope, with broadly horseshoe shaped or funnel shaped close-set scales, also covering most of rib flanks and hollow ventrally. Ribs on posterior slope very low saw-tooth shaped with a sharp rib crest, sculptured with pronounced spines, placed on posterior rib flanks, directed posteriorly. Fine commarginal growth lines present in interspaces, combined with well developed, commarginally aligned bands of dense granulations on anterior and median zones. Rib impressions well visible from within. Hinge with nearly horizontally rotated cardinal teeth. Left valve: two cardinal teeth of which the anterior one well defined, posterior one tiny; both teeth connected at their bases, with a deep socket below. Right valve: two similar sized (extrapolated), erect, pointed cardinal teeth, not touching at their bases but separated by a deep socket. Anterior lateral sockets curved, extremely excavated; lateral teeth of both valves thin, blade like. Ligament rather short. Lunule not well defined, dorsal margin slightly raised anterior of umbo.
Remarks.—Fragments of Procardium from the early Miocene of the North Sea Basin, The Netherlands and northern Germany, are common in museum and private collections (Nordsieck 1972: 38, pl. 8: 59; Bosch et al. 1975: 82, pl. 12: 2; Janssen 1984: 82, pl. 31: 3–5; Moths 1994: figs 1–6; Wienrich 1999: 240, pl. 57: 3a, b; Moths et al. 2010: 21, fig. 13, pl. 7: 6, 7; Stein et al. 2016: 19, pl. 7: 5) as Cardium (Ringicardium) danubianum (Kautsky 1925: 36) or as C. (Ringicardium) hians (Heering 1950: 32) in the literature.
Records are mainly from the area of Winterswijk-Miste (province of Gelderland, The Netherlands) and to a lesser extent from the Peel region (bordering Noord-Brabant and Limburg provinces, The Netherlands), but there are also several records from northern Germany (Hemmoor-Basbeck, Itzehoe, Kevelaer, and Werder), all from deposits of Hemmoorian age, late Burdigalian to Langhian in the standard time scale (Janssen 2001; Gürs and Janssen 2002). The deposits in the area of Winterswijk-Miste most probably are late Hemmoorian (Oxlundian), i.e., Langhian in age (Arie W. Janssen personal communication 2017).
The study material, generally well preserved apart from fragmentation and invariably brownish-gray coloured, is mostly from Winterswijk-Miste, with the addition of few fragments from the coeval deposits at Beeringen (now: Beringe) and Baarlo, both in De Peel region, and Werder in northwestern Germany. The relatively abundant material (over 100 fragments), allowed the shell characters of the new species to be recognized and described. In order to give a realistic impression of a complete valve, a graphic reconstruction is herein given (Fig. 14), based on a photographic collage of 12 fragments, most of which are illustrated (Fig. 13), as well as examination of numerous additional fragments.
The shell of P. jansseni sp. nov. is particularly thin and fragile, and the fragmented condition in which this species is found is evidently due to its intrinsic shell fragility and weakness of radial ribs. The largest fragments are mostly from the anterior and posterior regions: in the former, scales are particularly crowded, giving some strength to shell, while the latter have wider and stronger ribs.
Further comments and comparisons are reported under the respective species.
Stratigraphic and geographic range.—Procardium jansseni sp. nov. is only known from the late Burdigalian to Langhian of the southern North Sea Basin.
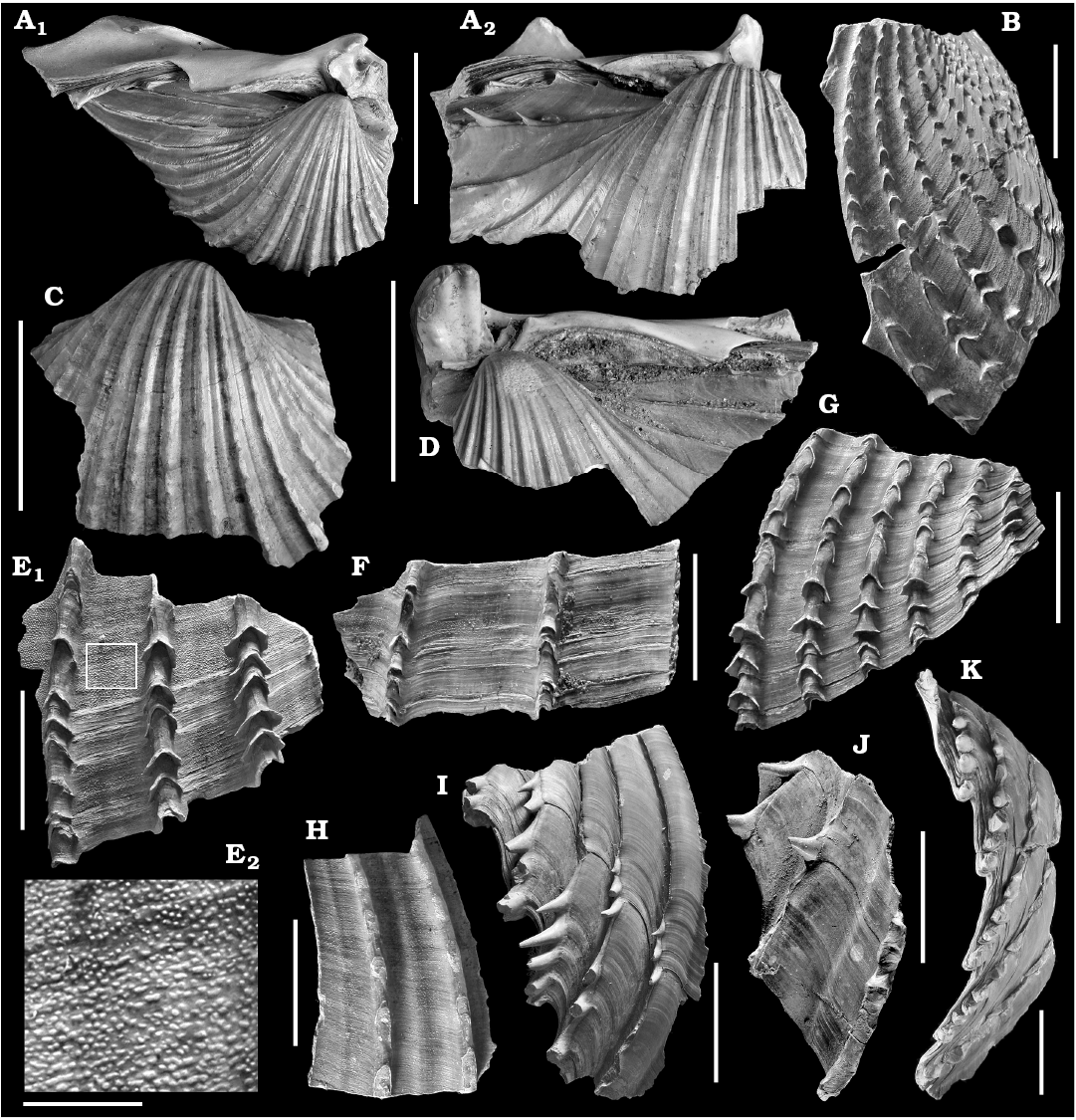
Fig. 13. Cardiid bivalve Procardium jansseni sp. nov. from Langhian (Miocene) of Winterswijk, Miste (except F that is from Limburg, Beringe, borehole Beeringen DB15 [B58B008]), The Netherlands. A. Holotype, RGM.1309269 (van der Voort collection), umbonal fragment. B. Paratype, RGM.225756, anterior fragment. C. RGM.1309415 (van der Voort collection), umbonal fragment, hinge missing. D. RGM.1309416 (van der Voort collection), umbonal fragment. E. Paratype, RGM.225755, anterior fragment (E1) and detail of microsculpture (E2). F. RGM.794094 (Rijks Geologische Dienst collection), median fragment. G. RGM.1309417 (van der Voort collection), anterior fragment. H. RGM.794092.a, median fragment. I. Paratype, RGM.225754, posterior fragment. J. RGM.1309991 (ter Poorten collection), posterior fragment. K. RGM.1309418 (van der Voort collection), posterior fragment. Scale bars: A–D, E2, F–K 10 mm; E1, 5 mm.
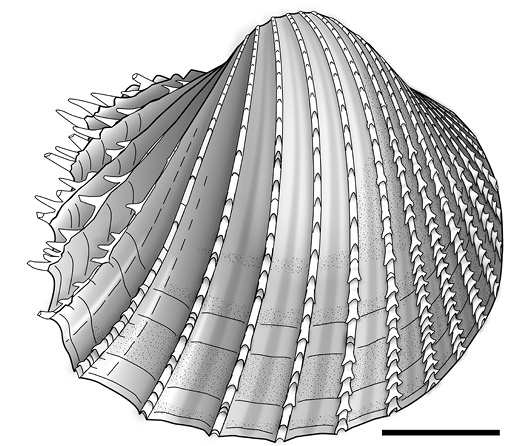
Fig. 14. Cardiid bivalve Procardium jansseni sp. nov. Graphic reconstruction of a right valve based on 12 fragments from Langhian (Miocene), The Netherlands, Miste, and Beringe. Fragments covering the majority of valve surface. Scale bar 20 mm.
Procardium magnei sp. nov.
Fig. 15.
1866 Cardium darwini; Mayer 1866: 69 (partim).
1912 Cardium (Ringicardium) darwini Mayer, 1866; Cossmann and Peyrot 1912: 492, pl. 21: 26, 27 (non C. darwini Mayer, 1866).
1950 Ringicardium schoelleri; Magne 1950: 123 (nomen novum for C. darwini Mayer, 1866).
1994 Cardium (Ringicardium) darwini Mayer, 1865 (sic!); Marcomini 1994: 15.
Etymology: After André Magne, paleontologist and malacologist from the geological department of the Faculty of Sciences at the University of Bordeaux in the 1940s–60s, who first tried to settle the nomenclature of this species.
Type material: Holotype PH11708, right valve (L 92 mm, H 86 mm). Paratypes: PH63240, left valve (L 68 mm, H ~ 57 mm), RGM.794098, right valve (L 76 mm, H 71 mm) from the type locality.
Type horizon: Yellow shelly sands, early Burdigalian, Miocene.
Type locality: Saucats “La Coquillière” (also known as “Coquilleyre”), Gironde department, France.
Material.—RGM.794100, one valve, PH63241, two fragments, from early Burdigalian (Miocene), Saucats “La Coquillière”, France. JJTP4779, JJTP4780, three fragments from early Burdigalian (Miocene), Léognan “Le Coquillat”, France.
Diagnosis.—A rather large, inflated Procardium species with 18–19 sharply delimited high triangular flat-topped radial ribs. Scales rather well spaced and also covering the upper part of the rib flanks. Interspaces broad with variably developed minute granulations on anterior and median zones.
Description.—Shell large (L up to 92 mm), slightly longer than high, weakly inequilateral, relatively thin, inflated, with a moderately wide posterior gape, covering two-thirds of posterior margin length. Umbo slightly prosogyrate. Anterior and ventral margins rounded, postero-ventral margin rather angular, posterior margin truncated and nearly straight. Shell bearing 18–19 sharply delimited primary radial ribs, on anterior half alternating with a very weakly developed, broad and flattened secondary riblet. On median part, interspaces about 3–4 times wider than primary ribs. Anterior ribs high triangular, flat-topped with rather well spaced, broadly horseshoe shaped scales, also covering upper part of rib flanks and hollow ventrally. Ribs on postero-ventral slope becoming even more prominent high triangular, carrying slender shingle-shaped scales. Ribs ultimately flattened triangular on postero-dorsal slope and sculptured with pronounced spines, placed on posterior rib flanks, directed posteriorly. Fine commarginal growth lines present in interspaces, combined with a variably developed pattern of minute granulations on anterior and median zones. Rib impressions well visible from within, with notable deep incisions at postero-medial marginal zone. Hinge plate rather narrow, slightly sinuous; cardinal teeth nearly horizontally rotated. Left valve: two cardinal teeth of which the anterior one well defined, posterior one tiny; both teeth connected at their bases, with a deep socket below. Right valve: two similar sized, erect, pointed cardinal teeth, not touching at their bases but separated by a deep socket. Anterior lateral sockets curved and extremely excavated, lateral teeth of both valves thin, blade like. Tips of lateral teeth about equidistant from anterior cardinal. Ligament short. Lunule not well defined, dorsal margin slightly raised anterior of umbo.
Remarks.—Because of the incorrect usage of Mayer’s (1866) name by Cossmann and Peyrot (1912), Magne (1950) proposed Cardium schoelleri as a nomen novum (ICZN 1999: Art. 72.7), evidently with the aim of giving a name to the fossil species from the Aquitaine Basin reported by Cossmann and Peyrot (1912). However, as a replacement name of C. darwini Mayer, 1866, C. schoelleri is a Type of C. indicum, while C. darwini sensu Cossmann and Peyrot (1912) remained an undescribed species.
Mayer (1866) referred some fossil cardiids from the Aquitaine Basin (Léognan, Saucats, Saint-Paul-lès-Dax, Saubrigues, Saint-Jean-de-Marsaq), from “Helvetian” localities in Switzerland, as discussed under P. danubianum, and from the Miocene and Pliocene of Algeria, to C. darwini. Evidently, as remarked by Cossmann and Peyrot (1912) and by Magne (1950), Mayer’s (1866) interpretation of C. darwini included more than one species. However, after Mayer’s (1866) description, the identity of the species on which he based his description is clear: it is the same as that reported from Léognan as C. (Ringicardium) darwini by Cossmann and Peyrot (1912: 492, pl. 21: 26, 27), who gave a more detailed description, including the hinge.
The main sculptural characters of Procardium magnei sp. nov. were clearly outlined by Mayer (1866: 70): 15–18 wide spaced ribs, thin, elevated and markedly sharp “distantes, très-minces, élevées et assez aiguës”. Scales, mostly worn out, are U- or V-shaped, straddling the ribs, as Mayer (1866: 70) tried to describe “côtes en forme de lamelles, qu’embrassent, pour ainsi dire, les écailles en cornets qui les surmontent” (Fig. 16A).
Types and the scarce additional material are from the early Burdigalian of the area of Saucats in the Aquitaine Basin. Geographic and stratigraphic details on these deposits were given by Cahuzac and Janssen (2010). Cossmann and Peyrot (1912) reported the present species from Léognan, another classical locality in the Aquitaine Basin, of early Burdigalian age (Poignant et al. 1997; Cahuzac and Janssen 2010); their figured specimen (Cossmann and Peyrot 1912: pl. 2: 26, 27) has been rediscovered and is deposited in the MNHN collection (Marcomini 1994). The other localities mentioned by Mayer (1866) include deposits whose age ranges from the Aquitanian (Saint-Paul-lès-Dax) to the Langhian (Saubrigues), including the late Burdigalian (Saint-Jean-de-Marsaq) (Cahuzac and Janssen 2010), which could imply the complete stratigraphic range of the species. The records from Switzerland by Mayer (1866), apparently based on internal moulds, are from the Upper Marine Molasse (middle Burdigalian). As discussed above, they were most probably misidentified, as also the records from the Neogene of Algeria.
Procardium magnei sp. nov. shares its main sculptural characters with P. jansseni sp. nov., P. danubianum and in part with P. kunstleri (Table 2). All of them have thin median ribs, triangular in cross-section, markedly different from the broad, convex ribs with a central groove of P. indicum, P. diluvianum, and P. avisanense. However, P. magnei sp. nov. is more similar in sculpture to P. jansseni sp. nov. than to the others: both species have sharply delimited radial ribs and weak secondary riblets (Fig. 16A, B), while in P. danubianum (Fig. 16C) the main ribs are not well delimited and the secondary riblets are more distinct. The rib separating the ventral and posterior slope is high triangular in P. magnei sp. nov., whereas it is thin and small in P. jansseni sp. nov. The scaly sculpture is not markedly variable among the Procardium species, but in P. jansseni sp. nov. the funnel-shaped scales wrap totally the rib (Figs. 13B, E1, G, 16B). Similar scales are also present in P. magnei sp. nov., but less markedly and mainly on the anterior ribs (Figs. 15A4, C2, 16A), while the scaly sculpture is poorly preserved in P. danubianum. Finally, a granular microsculpture in the radial interspaces occurs in P. magnei sp. nov., P. jansseni sp. nov., and P. danubianum. It is particularly well developed in P. jansseni sp. nov., being granulations larger and denser, often coalescent (Fig. 13E2). They form commarginal bands with variable density (Figs. 13E1, 14).
Procardium magnei sp. nov. and P. jansseni sp. nov. are also similar in shape, equidimensional and weakly inequilateral. Also P. danubianum has a similar shape, but only in the younger stages, becoming somewhat elongate and inequilateral with growth, while P. kunstleri is marked inequilateral and oblique.
Stratigraphic and geographic range.—Procardium magnei sp. nov. is known from the Burdigalian of the Aquitaine Basin, but probably ranging from Aquitanian to Langhian, Miocene.

Fig. 15. Cardiid bivalve Procardium magnei sp. nov. from early Burdigalian (Miocene) of Saucats, “La Coquillière”, France. A. Holotype, PH 11708, external (A1), internal (A2), and umbonal (A3) views, details of sculpture (A4) and right hinge (A5). B. Paratype, PH 63240, external (B1), internal (B2), and posterior (B3) views. C. Paratype, RGM.794098, external view (C1), detail of sculpture (C2) and microsculpture (C3). Scale bars: A1–A3, B, C1, 50 mm; A5, C2, 20 mm; A4, 10 mm; C3, 5 mm.
Discussion
As in many cardiid groups, in Procardium gen. nov. the most diagnostic characters are provided by sculpture (ribbing pattern, strength and shape of main and secondary ribs, shape of scales, etc.) (e.g., Vidal 1999; La Perna and D’Abramo 2009; ter Poorten 2013). The granular microsculpture in the radial interspaces, found in three species, seems similar to that known for Vepricardium Iredale, 1929 and Fulvia Gray, 1853 (Vidal 1994, 2000; ter Poorten 2009): in both cases, dense granulations are present in several species, also covering the ribs and sometimes commarginally aligned. Interestingly, Vepricardium is assigned to the subfamily Cardiinae (ter Poorten 2005), while Fulvia, traditionally placed in the Laevicardiinae, turned out to be a paraphyletic group forming a sister subclade of the Cardiinae (Herrera et al. 2015). Nevertheless, the presence of granular microsculpture amongst cardiid clades seems a case of morphological convergence rather than resulting from phylogenetic relationship, as similar structures have been found in Caribbean Laevicardium and Dallocardia taxa (both Trachyardiinae; personal observation JJtP).
Based on the rib shape, rib sculpture and presence of granulations, three species groups can be distinguished in the evolutionary history of Procardium gen. nov. (Fig. 16). The oldest one includes P. magnei sp. nov. (Fig. 16A), from the early Burdigalian of the Aquitaine Basin, but probably with a longer Aquitanian–Langhian range; P. jansseni sp. nov. (Fig. 16B), from the Langhian of the North Sea Basin; P. danubianum (Fig. 16C), from the early Burdigalian–middle Serravallian of Central Paratethys and, at least tentatively, Procardium sp. from the Langhian of the Liguria-Piedmont Basin. All of them share thin ribs, triangular in cross-section, and microgranulations in the radial interspaces. This group seems to include two morphologically and palaeogeographically distinct lineages: one with a NE Atlantic distribution (P. magnei sp. nov. and P. jansseni sp. nov., with subtriangular shells), the other with a Paratethyan-Mediterranean distribution (P. danubianum, Procardium sp., with slightly elongate shells).
Procardium kunstleri, with thin ribs but without granulations (Fig. 16D), may form part of a distinct lineage. The species is known from the Serravallian and Tortonian of NE Atlantic (Aquitaine Basin and Algarve Basin), but there is evidence that some similar species, only known from poorly preserved material, occurred in the middle and late Miocene of the Mediterranean. Procardium aff. kunstleri (Fig. 16E) from the Gualdalquivir Basin testifies the occurrence of this species group up to the early Pliocene. Burdigalian records from the Western Paratethys, all based on internal moulds, seem closer to P. danubianum than to P. kunstleri. Possibly, the group of P. kunstleri was the most diverse within the genus, but unfortunately it is the least known.
The close similarities among P. avisanense (Fig. 16F), P. diluvianum (Fig. 16G), and P. indicum (Fig. 16H) have been already noted: they share relatively wide, convex ribs with a median groove. Apparently, these three species form a NE Atlantic and Mediterranean lineage ranging from the middle Miocene to the Recent.
Fig. 16. Comparative rib cross-sections and rib sculpture for species of Procardium. Schematic drawings based on the third/fourth rib anterior to the midline, close to the margin of an adult shell. A. P. magnei sp. nov. B. P. jansseni sp. nov. (upper part of rib sculpure reconstructed). C. P. danubianum (rib sculpture undefined). D. P. kunstleri. E. P. aff. kunstleri. F. P. avisanense. G. P. diluvianum. H. P. indicum. Median rib, showing radial groove on rib top (H2). Note presence of microscopic granulations (white arrow) and secondary riblet in interspace (black arrow). Scale bar 5 mm.
Mediterranean and Paratethys were more or less continuously connected from the late Oligocene to the early Serravallian (Rögl 1998; Steininger and Wessely 2000; Studencka et al. 2016), allowing faunal exchange (Harzhauser and Piller 2007; Bartol et al. 2014; La Perna et al. 2017), including Procardium gen. nov. which spread all through the Mediterranean, Western and Central Paratethys. The northward diffusion, presumably from the Iberian and/or French coast to the North Sea, is supported by the strong affinities between the Miocene fauna of the North Sea Basin and that of the Aquitaine Basin, which are explained, implicitly or explicitly, with direct exchanges via the English Channel (Janssen 2001; Kowalewski et al. 2002; Lozouet 2014). According to some palaeogeographic reconstructions (Cahuzac et al. 1992; Van Vliet-Lanoë et al. 2010), the Strait of Dover was closed during the late Oligocene–Tortonian interval, but other reconstructions (Rögl 1998; Popov et al. 2004) reported an open Strait, at least in the Chattian to Burdigalian time slice.
With P. jansseni sp. nov., the genus reached the highest latitude (ca. 54° N) in its biogeographic history. This can be easily related with the warm climatic conditions, usually referred to as Miocene Climatic Optimum (Zachos et al. 2001; Böhme 2003), in the late Burdigalian–Langhian, when the boundary between the tropical region and the transitional zone to the Boreal region was close to The Netherlands and northwest Germany (Lauriat-Rage et al. 1993; Lozouet 2014). Although the stratigraphic ranges of species may be incomplete, the maximum diversity of Procardium gen. nov. seems to occur during the Miocene Climatic Optimum. Subsequently, the genus gradually retreated southward, from ca. 45° N in the Serravallian (Aquitaine Basin), to 37–36° N in the Tortonian (Algarve Basin) and in the early Pliocene (Gualdalquivir Basin), with P. avisanense and P. diluvianum, respectively. Once again, the relation to climate change is obvious: the southern withdrawal of Procardium gen. nov. can be referred to the cooling trend recorded after the Miocene Climatic Optimum (Raymo 1994; Zachos et al. 2001), which strongly affected the composition and palaeobiogeography of the European marine fauna, particularly since the middle Pliocene (Raffi et al. 1985; Monegatti and Raffi 2001).
The warm-water character of Procardium gen. nov., similar to that of its closest relatives, Cardium and Bucardium (ter Poorten 2007; La Perna 2016), is clearly suggested by the present-day distribution of P. indicum. The palaeobiogeographic history of this genus and its persistence in Europe, up to the Early Pleistocene, is similar to that of another cardiid genus, Europicardium Popov, 1977, represented by three living West African species but with a rather diverse record in the Neogene of Europe (La Perna 2017). Interestingly, the molecular phylogeny of Herrera et al. (2015) indicates Europicardium as a Cardiinae genus, sister to the Cardium–Bucardium clade. After the maximum diversity attained during the Miocene Climatic Optimum, Europicardium persisted in the Mediterranean up to the Early Pleistocene, in spite of the increasing cooling trend (La Perna 2017: fig. 16). Europicardium and Procardium gen. nov. should be therefore considered as rather tolerant in their thermal preferences.
No data are available on the fossil occurrence of Procardium gen. nov. in West Africa, but this area surely has a role in the evolution and diffusion of Cardium, Bucardium, and the new genus, in addition to Europicardium as discussed above. Cardium is a young West African offshoot, which originated in the late Miocene or considerably later, as suggested by a Bayesian estimation of divergence times (Herrera et al. 2015: fig. 3) and by its scant fossil record, only from the Pliocene of Angola (Gofas et al. 1985). The origin of Bucardium seems more or less coeval with that of Procardium gen. nov. in the early Miocene, not necessarily in the European area (La Perna 2016). It can be hypothesized that Procardium gen. nov. reached its southernmost outpost since the Early–Middle Pleistocene transition, in response to the increased icehouse condition (Head and Gibbard 2005), but it also can be assumed that the genus had already reached West Africa during the Miocene. Alternatively, it may have originated in West Africa, from which it spread northward, following favourable climatic conditions in the early Neogene. Noteworthy is that West Africa and southern Europe belonged to the same biogeographical province during the early Miocene.
The hypothesis of a northward diffusion of Procardium gen. nov. should take in consideration the occurrence of an intense cold upwelling in North-West Africa, separating the West African tropical waters from the Mediterranean since the latest Miocene, as discussed by Vermeij (2012). However, the distribution of P. indicum, mostly within the upwelling stretch in North-West Africa and including part of the warm waters of the Guinea Gulf, suggests a rather wide thermal tolerance, which could have characterized the extinct congeners too. The origin of Procardium gen. nov. somewhere in West Africa or Europe, remains an enigma.
A more complete reconstruction of the evolutionary and biogeographic history of Procardium gen. nov. would require a deeper knowledge of some late Oligocene–early Miocene cardiids, evidently distinct from the new genus but with shared shell characters, as discussed by La Perna (2016). This poorly known group, herein exemplified by “Cardium” burdigalinum (Fig. 2D), includes Cardium (Ringicardium) buekkianum Telegdi-Roth, 1914 (La Perna 2016: fig. 8, as Cardium sp.) described from the Egerian (Chattian–early Aquitanian) of Hungary, reported from the Chattian of southeastern Turkey (Hüsing et al. 2008) and also known from the Chattian of the Aquitaine Basin (JJtP unpublished data). The record by Hüsing et al. (2008), from the eastern Tethys gateway, i.e., the seaway which connected Proto-Mediterranean and Indian Ocean, may open new views on the diffusion pathway of this species group, which appears to be the closest one to Procardium gen. nov., morphologically, geographically and stratigraphically.
This is not the place for dealing with the adaptive aspects of Procardium, which were discussed on a morphofunctional basis by Savazzi (1985) and Savazzi and Sälgeback (2004). However, this genus seems to have always been linked to relatively deep, quiet waters and muddy substrates, throughout its evolution. Procardium indicum is indeed a mud-dweller, distributed at depths largely exceeding 30 m (Petit 1840; Weinkauff 1862; Ghisotti 1971, Fischer-Piette 1977). A similar habit can be inferred for the fossil species, based on the shared shell morphology and on the occurrence of delicate, erect scales, sometimes even more developed than in P. indicum, incompatible with shallower water and coarser sediment.
Acknowledgements
We express our gratitude to the following people who kindly allowed study of institutional and private collections: Riccardo Manni (MPUR), Oleg Mandic and Mathias Harzhauser (both NHMW), Didier Merle and Jean-Michel Pacaud (both MNHN); Daniele Ormezzano (MRSN), Alessandro Garassino and Giorgio Teruzzi (both MSNM), Rob G. Moolenbeek (NCB Naturalis, ex ZMA); Ronald Pouwer and Jeroen Goud (both NCB Naturalis); Piet and Rudi Hessel (Stichting Schepsel Schelp, Rhenen, The Netherlands); Thomas R. Waller and Mark S. Florence (both USNM); Eric Kaptein (Almelo, The Netherlands) and Jaap van der Voort (Ostercappeln-Venne, Germany). David Jablonski (University of Chicago, USA) kindly assisted with literature. We would like to thank Mathias Harzhauser and Oleg Mandic (both NHMW) for comments and suggestions on an earlier draft of this work. Simon Schneider (University of Cambridge, UK), David Jablonski (University of Chicago, USA) and an anonymous reviewer are kindly acknowledged for their constructive remarks and linguistic assistance. Cardiid research and travel costs of JJtP were partly supported by the U.S. National Science Foundation under awards DEB-0919124/0918982/0919451. RLP (Fondi di Ricerca d’Ateneo 2014, Università di Bari) contributed to printing costs.
References
Andrés, I. 1987. Los Heterodonta (Bivalvia) en el Plioceno marino de Bonares (Huelva). Studia Geologica Salmanticense 24: 83–149.
Bartol, M., Mikuž, V., and Horvat, A. 2014. Palaeontological evidence of communication between the Central Paratethys and the Mediterranean in the late Badenian/early Serravalian. Palaeogeography, Palaeoclimatology, Palaeoecology 394: 144–157. Crossref
Böhme, M. 2003. The Miocene Climatic Optimum: evidence from ectothermic vertebrates of Central Europe. Palaeogeography, Palaeoclimatology, Palaeoecology 195: 389–401. Crossref
Bosch, M. van den, Cadée M.C., and Janssen, A.W. 1975. Lithostratigraphical and biostratigraphical subdivision of Tertiary deposits (Oligocene–Pliocene) in the Winterswijk-Almelo region (eastern part of The Netherlands). Scripta Geologica 29: 1–168.
Bossio, A., Mazzei, R., Salvatorini, G., and Sandrelli, F. 2002. Geologia dell’area compresa tra Siena e Poggibonsi (“Bacino del Casino”). Atti della Società Toscana di Scienze Naturali, Memorie, Serie A 2000–2002: 69–85.
Brambilla, G. 1976. I Molluschi pliocenici di Villalvernia (Alessandria). I. Lamellibranchi. Memorie della Società Italiana di Science Naturali e del Museo civico di Storia naturale di Milano 21: 79–128.
Brocchi, G. 1814. Conchiologia fossile subappennina con osservazioni geologiche sugli Appennini e sul suolo adiacente. 472 pp. Stamperia Reale, Milano. Crossref
Cahuzac, B. and Janssen, A.W. 2010. Eocene to Miocene holoplanktonic Mollusca (Gastropoda) of the Aquitaine Basin, southwest France. Scripta Geologica 141: 1–193.
Cahuzac, B., Alvinerie, J., Lauriat-Rage, A., Montenat, C., and Pujol C., 1992. Palaeogeographic maps of the Northeastern Atlantic Neogene and relation with the Mediterranean sea. Paleontologia i Evolució 24–25: 279–293.
Cerulli Irelli, S. 1908. Fauna Malacologica Mariana. Parte seconda. Leptonidae, Galeommidae, Cardiidae, Chamidae, Cyprinidae, Veneridae. Palaeontographia Italica 14: 1–64.
Chavan, A. 1940. Les fossiles du Miocène supérieur de Cacela. Comunicações dos Serviços Geológicos de Portugal 21: 61–106.
Chirli, C. 2016. Malacofauna Pliocenica Toscana. Vol. 12. Bivalvia. Heteroconchia Hertwig, 1895. Protobranchia Pelseneer, 1889. 211 pp. C. Chirli, Firenze.
Chirli, C. and Richard, C. 2008. Les Mollusques plaisanciens de la Côte d’Azur. 128 pp. C. Chirli, Tavarnelle Val di Pesa.
Ćorić, S., Harzhauser, M., Rögl, F., İslamoğlu, Y., and Landau, B. 2012. Biostratigraphy of some mollusc-bearing middle Miocene localities on the Karaman high plain (Turkey, Konya Province). Cainozoic Research 9: 281–288.
Cossmann, A.E.M. and Peyrot, A. 1909–1914. Conchologie néogénique de l’Aquitaine. Pélécypodes. Cardiacea: Vol. 1. 429–718, pls. 19–28. Published parallely in Actes de la Société Linnéenne de Bordeaux 65 (2): 51–98; 65 (3): 99–178; 65 (4): 179–333, pls. 19–28.
Courme-Rault, M.-D. and Dubar, M. 2011. Les formations cénozoïques dans le Sud-Est de la France (région située entre Grasse et Nice). Géologie de la France 1: 3–70.
Dailianis, T., Akyol, O., Babali, N., Bariche, M., Crocetta, F., Gerovasileiou, V., Ghanem, R., Gökoğlu, M., Hasiotis, T., Izquierdo-Muñoz, A., Julian, D., Katsanevakis, S., Lipej, L., Mancini, E., Mytilineou, Ch., Ounifi Ben Amor, K., Özgül, A., Ragkousis, M., Rubio-Portillo, E., Servello, G., Sini, M., Stamouli, C., Sterioti, A., Teker, S., Tiralongo, F., and Trkov, D. 2016. New Mediterranean biodiversity records (July 2016). Mediterranean Marine Science 17: 608–626. Crossref
Delongueville, C. and Scaillet, R. 2011. Présence de Cardium indicum Lamarck, 1819 sur la côte Est de Tunisie. Novapex 12: 3–8.
Demarcq, G. 1960. Contribution à l’étude des facies de Miocène de la vallée du Rhône. Mitteilungen der Geologischen Gesellschaft in Wien 52: 93–104.
De Stefani, C. 1874. Fossili pliocenici dei dintorni di S. Miniato (Toscana). Bullettino Malacologico 7: 5–89.
De Stefani, C. and Pantanelli, D. 1879. Molluschi pliocenici dei dintorni di Siena. Bullettino Malacologico 4: 49–112.
Dollfus, G.F., Cotter, J.C.B., and Gomes, J.P. 1903–1904. Mollusques tertiaires du Portugal. Planches de céphalopodes, gastéropodes et pélécypodes. Laissées par F.A. Pereira da Costa. Accompagnées d’une explication sommaire et d’une esquisse géologique. Mémoires de la Commission du Service Géologique du Portugal 34: 1–117.
Erünal-Erentöz, L. 1958. Mollusques du Neogène des bassins de Karaman, Adana et Hatay (Turquie). Publications de l’Institut d’Études et Recherches Minières de Turquie C4: 1–232.
Fischer, P. 1866. Faune tertiaire moyenne. In: P. Tchihatcheff (ed.), Asie Mineure. Description physique de cette contrée, 235–318. L. Guérin et C.ie, Paris.
Fischer, P. and Tourmouër, R. 1873. Étude sur les Invertébrés fossiles du Mont Léberon. In: A. Gaudry (ed.), Animaux fossiles du Mont Léberon (Vaucluse), 113–180. F. Savy, Paris.
Fischer-Piette, E. 1977. Révision des Cardiidae (Mollusques, lamellibranches). Mémoires du Museum national d’Histoire naturelle, n.s., A Zoologie 101: 1–212.
Fontannes, F. 1878. Les terrains néogènes du plateau de Cucuron (Vaucluse) (Cadenet; Cabrières-d’Aigues). Bulletin de la Société Géologique de France, 3e série 6: 469–529.
Fontannes, F. 1879. Études stratigraphiques et paléontologiques pour servir à l’histoire de la période tertiaire dans le bassin du Rhône. 5. Description de quelques espèces nouvelles ou peu connues. 56 pp. H. George, F. Savy, Lyon.
Fontannes, F. 1882. Les invertebres du bassin tertıaire du sudest de la France. Les mollusques pliocenes de la Vallee du Rhône et du Roussillon. Vol. 2, Acephales. 321 pp. H. Georg, Lyon.
Ghisotti, F. 1971. Un raro bivalve del Mediterraneo: Cardium hians Brocchi. Conchiglie 7: 73–82.
Ghisotti, F. 1974. La frenesia della nuova species. Conchiglie 10: 65–70.
Gignoux, M. 1913. Les formations marine Pliocènes et Quaternaires dans Italie du Sud et la Sicile. Annales Université de Lyon n.s. 36: 1–633.
Gofas, S., Pinto Afonso, J., and Brandão, M. 1985. Conchas e moluscos de Angola. Coquillages et mollusques d’Angola. 144 pp. Universidade Agostinho Neto, Elf Aquitaine Angola, Luanda.
Gómez Rodríguez, R. and Pérez Sánchez, J.M. 1997. Moluscos bivalvos de Canarias. 425 pp. Cabildo Insular de Gran Canaria, Las Palmas de Gran Canaria.
Gürs, K. and Janssen, A.W. 2002. Revised pteropod biostratigraphy for the Miocene of the North Sea Basin. In: K. Gürs (ed.), Northern European Cenozoic Stratigraphy. Proceedings of the 8th biannual meeting of the RCNNS/RCNPS. Landesamt für Natur und Umwelt des Landes Schleswig-Holstein, Flintbek, 117–131.
Harzhauser, M. and Piller, W.E. 2007. Benchmark data of a changing sea—Palaeogeography, palaeobiogeography and events in the Central Paratethys during the Miocene. Palaeogeography, Palaeoclimatology, Palaeoecology 253: 8–31. Crossref
Head, M.J. and Gibbard, P.L. 2005. Early–Middle Pleistocene transitions: an overview and recommendation for the defining boundary. In: M.J. Head and P.L. Gibbard (eds.), Early–Middle PleistoceneTransitions: The Land-Ocean Evidence. Geological Society, London, Special Publication 247: 1–18. Crossref
Heckeberg, N., Pippèrr, M., Läuchli, B., Heimann, F.U.M., and Reichenbacher, B. 2010. The Upper Marine Molasse (Burdigalian, Ottnangian) in Southwest Germany—Facies interpretation and a new lithostratigraphic terminology. Zeitschrift der Deutschen Gesellschaft für Geowissenschaften 161: 285–302. Crossref
Heering, J. 1950. Miocene Pelecypoda of The Netherlands (Peel-region). Mededelingen van de Geologische Stichting, ser. C, IV-1 10: 1–52.
Herrera, N.D., Poorten J.J. ter, Bieler, R., Mikkelsen P.M., Strong E.E., Jablonski D., and Steppan, S.J. 2015. Molecular phylogenetics and historical biogeography amid shifting continents in the cockles and giant clams (Bivalvia: Cardiidae). Molecular Phylogenetics and Evolution 93: 94–106. Crossref
Hörnes, M. 1851–1870. Die fossilen Mollusken des Tertiär-Beckens von Wien. II. Bivalven. Abhandlungen der Kaiserlich-Königlichen Geologischen Reichsanstalt 4: 1–477.
Hüsing, S.K., Zachariasse, W.-J, van Hinsbergen, D.J.J., Krijgsman, W., Inceöz, M., Harzhauser, M., Mandic, O., and Kroh, A. 2008. Oligocene–Miocene basin evolution in SE Anatolia, Turkey: constraints on the closure of the eastern Tethys gateway. In: van Hinsbergen, D.J.J., Edwards, M.A., and Govers, R. (eds.), Collision and Collapse at the Africa-Arabia-Eurasia Subduction Zone. The Geological Society, London, Special Publications 311: 107–132.
Hylleberg, J. 2004. Lexical approach to Cardiacea. 1. Literature. 2–3. Records of taxa A–Z. Illustrated and annotated bibliography of living and fossil shells, with emphasis on the families Cardiidae and Lymnocardiidae (Mollusca: Bivalvia). Phuket Marine Biological Center Special Publication 29: 1–352; 30: 353–940.
Hylleberg, J. 2014. Horizontal pivot axis and standardized shell outline: A new method to compare morphology of cardiids (Bivalvia: Cardiidae) exemplified by the genera Cardium, Acanthocardia and Vasticardium. The fossil Cardium hians Brocchi, 1814 replaced by Cardium paleobrocchi nom. nov. Phuket Marine Biological Center Special Publication 32: 37–51.
ICZN 1999. International Code of Zoological Nomenclature (4th ed.). 306 pp. The International Trust for Zoological Nomenclature, London.
Janssen, A.W. 1984. Mollusken uit het Mioceen van Winterswijk-Miste. Een inventarisatie, met beschrijvingen en afbeeldingen van alle aangetroffen soorten. 452 pp. Koninklijke Nederlandse Natuurhistorische Vereniging, Amsterdam.
, JanssenA.W. 2001. The age of the North Sea Basin Hemmoorian (Miocene): holoplanktonic molluscan evidence. Aardkundige Mededelingen 11: 45–50.
Kautsky, F. 1925. Das Miocän von Hemmoor und Basbeck-Osten. Abhandlungen der Preußischen Geologischen Landesanstalt NF 97: 1–255.
Kowalewski, M., Gürs, K., Nebelsick, J.H., Oschmann, W., Piller, W.E., and Hoffmeister, A.P. 2002. Multivariate hierarchical analyses of Miocene mollusk assemblages of Europe: Paleogeographic, paleoecological, and biostratigraphic implications. Geological Society of America Bulletin 114: 239–256. Crossref
Lamarck, M. de 1819. Histoire naturelle des animaux sans vertèbres, présentant les caractères généraux et particuliers de ces animaux, leur distribution, leurs classes, leurs familles, leurs genres, et la citation des principales espèces qui s’y rapportent […]. Tome 6, partie 1. 252 pp. Published by the author, Paris.
Lamarck, M. de, Deshayes, G.P., and H. Milne Edwards, 1835. Histoire naturelle des animaux sans vertèbres, présentant les caractères généraux et particuliers de ces animaux, leur distribution, leurs classes, leurs familles, leurs genres, et la citation des principales espèces qui s’y rapportent [...] Second edition. Tome 6. Histoire des mollusques. 600 pp. J.B. Baillière, Paris. Crossref
Lamy, E. 1941. Notes sur les espèces lamarckiennes de Cardium. (Moll. Lamellibr.). Bulletin du Museum national d’Histoire naturelle Paris, 2e sér. 13: 458–463.
Landau, B.M., Harzhauser, M., İslamoğlu, Y., and Marques da Silva, C. 2013. Systematics and palaeobiogeography of the gastropods of the middle Miocene (Serravallian) Karaman Basin, Turkey. Cainozoic Research 11–13: 3–584.
La Perna, R. 2016. Bucardium grateloupianum n. sp. from the Lower Miocene of Aquitaine with taxonomic comments on some fossil species from Europe (Bivalvia, Cardiidae). Zootaxa 4178: 568–582. Crossref
La Perna, R. 2017. A revision of the genus Europicardium Popov, 1977 (Bivalvia: Cardiidae) from the European Neogene: tracking palaeogeography and climate changes. Journal of Systematic Palaeontology 15: 287–312. Crossref
La Perna, R. and D’Abramo, M. 2009. Morphometric and systematic study on three Acanthocardia species from the Mediterranean Pleistocene (Mollusca, Bivalvia, Cardiidae). Geodiversitas 31: 669–682. Crossref
La Perna, R. and D’Abramo, M. 2013. Systematics of the Acanthocardia paucicostata group in the Mediterranean Plio-Pleistocene, with description of a new species (Bivalvia, Cardiidae). Journal of Conchology 41: 481–492.
La Perna, R., Mandic, O., and Harzhauser, M. 2017. Systematics and palaeobiogeography of Megacardita Sacco in the Neogene of Europe (Bivalvia, Carditidae). Papers in Palaeontology 3: 111–150. Crossref
Lauriat-Rage, A., Brebion, P., Cahuzac, B., Chaix, C., Ducasse, O., Ginsburg, L., Janin, M.-C., Lozouet, P., Margerel, J.-P., Nascimento, A., Pais, J., Poignant, A., Pouyet, S., and Roman, J. 1993. Palaeontological data about the climatic trends from Chattian to present along the Northeastern Atlantic frontage. Ciencias da Terra (UNL) 12: 167–179.
Lozouet, P. 2014. Temporal and latitudinal trends in the biodiversity of European Atlantic Cenozoic gastropod (Mollusca) faunas. A base for the history of biogeographic provinces. Carnets de Géologie (Notebooks on Geology) 14: 273–314. Crossref
Magne, A. 1950. Les Ringicardium néogènes du Bassin d’Aquitaine. Journal de Conchyliologie 90: 122–124.
Mandic, O. and Steininger, F. 2003. Computer-based mollusc stratigraphy—a case study from the Eggenburgian (Lower Miocene) type region (NE Austria). Palaeogeography, Palaeoclimatology, Palaeoecology 197: 263–291. Crossref
Mandic, O., Harzhauser, M., Schlaf, J., Piller, W.E., Schuster F., Wielandt-Schuster, U., Nebelsick, J.H., Kroh, A., Rögl, F., and Bassant, P. 2004. Palaeoenvironmental reconstruction of an epicontinental flooding—Burdigalian (Early Miocene) of the Mut Basin (Southern Turkey). Courier Forschungsinstitut Senckenberg 248: 57–92.
Marcomini, J.-L. 1994. La collection Léon Silvestre de Sacy (1867–1928). Cossmanniana 3: 13–29.
Mayer, C. 1866. Description de coquilles fossiles des terrains tertiaires supérieurs (suite). Journal de Conchyliologie 14: 67–76.
McGill, O. 1964. New Cardium from W. Africa. Hawaiian Shell News 12: 4.
Monegatti, P. and Raffi, S. 2001. Taxonomic diversity and stratigraphic distribution of Mediterranean Pliocene bivalves. Palaeogeography, Palaeoclimatology, Palaeoecology 165: 171–193. Crossref
Moshkovitz, S. 2012. The mollusca in the marine Pliocene and Pleistocene sediments of the south eastern Mediterranean Basin. Geological Survey of Israel Report 25: 1–159.
Moths, H. 1994. Die Muschel Cardium (Bucardium) sp. aus einem Hemmoor-Geschiebe der Kreidegrube Itzehoe-Kronsmoor. Der Geschiebesammler 27: 67–71.
Moths, H., Albrecht F., and Stein, G. 2010. Die Molluskenfauna (Hemmoorium, Untermiozän) aus der Kiesgrube Krinke bei Werder (Nordwest-Niedersachsen). Palaeofocus 3: 1–155.
Nordsieck, F. 1972. Die Miozäne Molluskenfauna von Miste-Winterswijk, NL (Hemmoor). 187 pp. Fischer Verlag, Stuttgart.
Pais, J., Legoinha, P., Elderfield, H., Sousa, L., and Estevens, M. 2000. The Neogene of Algarve (Portugal). Ciencias da Terra (UNL) 14: 277–288.
Paulissen, W.E., Luthi, S.M., Grunert, P., Ćorić, S., and Harzhauser, M. 2011. Integrated high-resolution stratigraphy of a Middle to Late Miocene sedimentary sequence in the central part of the Vienna Basin. Geologica Carpathica 62: 155–169. Crossref
Pavia, G. and Demagistris, L. 1971. Cardiacea del Pliocene medio e superior dell’Astigiano. Conchiglie 7: 94–112.
Petit, S. 1840. Notice sur l’habitat du Cardium hians de Brocchi; Card. Indicum Lamarck. Revue Zoologique, par la Société Cuvierienne 1840: 169–170.
Pfister, T. and Wegmüller, U. 1998. Bivalven aus der Oberen Meeresmolasse bei Bern. Beschreibung, Vergleich und Verbreitung der Bivalven-Arten aus den Belpbergschichten (Obere Meeresmolasse, mittleres Burdigalien) in der Umgebung von Bern, Schweiz. 2. Teil: Ostreacea, Heterodonta pro parte (Lucinacea, Chamacea, Carditacea und Cardiacea). Eclogae Geologicae Helvetiae 91: 457–491.
Pfister, T., Wegmüller, U., and Keller, B. 2011. Die Molluskenfauna der St. Galler Formation (Belpberg-Schichten, Obere Meeresmolasse) bei Bern (Schweiz): Taphonomie und Paläoökologie. Zitteliana 51: 153–208.
Pippèrr, M. 2011. Characterisation of Ottnangian (middle Burdigalian) palaeoenvironments in the North Alpine Foreland Basin using benthic foraminifera—A review of the Upper Marine Molasse of southern Germany. Marine Micropaleontology 79: 80–99. Crossref
Poignant, A., Pujol, C., Ringeade, M., and Londeix, L. 1997. The Burdigalian historical stratotype. In: A. Montanari, G.S. Odin, and R. Coccioni (eds.), Miocene Stratigraphy—An Integrated Approach. Developments in Palaeontology and Stratigraphy 15: 17–24.
Popov, S.V., Rögl, F., Rozanov, A.Y., Steininger, F.F., Shcherba, I.G., and Kovac, M. 2004. Lithological-Paleogeographic maps of Paratethys. 10 Maps Late Eocene to Pliocene. Courier Forschungsinstitut Senckenberg 250: 1–46.
Raffi, S., Stanley, S.M., and Marasti, R. 1985. Biogeographic patterns and Plio-Pleistocene extinction of Bivalvia in the Mediterranean and southern North Sea. Paleobiology 11: 368–388. Crossref
Raymo, M.E. 1994. The initiation of Northern Hemisphere glaciation. Annual Review of Earth and Planetary Sciences 22: 353–383. Crossref
Reeve, L.A. 1844. Conchologia Iconica or Illustrations of the Shells of Molluscous Animals. 2. Monograph of the Genus Cardium. 64 pp. (p. 47 excluded). Reeve Brothers, London.
Rögl, F. 1998. Palaeogeographic considerations for Mediterranean and Paratethys seaways (Oligocene to Miocene). Annalen des Naturhistorischen Museums in Wien, A 99: 279–310.
Rossi Ronchetti, C. 1952. I tipi della “Conchiologia fossile subapennina” di G. Brocchi. Parte I: Crostacei, Lamellibranchi. Rivista Italiana di Paleontologia e Stratigrafia, Memoire 5: 1–89.
Rutsch, R.F. 1928. Geologie des Belpbergs. Beiträge zur Kenntnis der Stratigraphie, Palaeontologie und Tektonik der Molasse südlich von Bern. Mitteilungen der Naturforschenden Gesellschaft in Bern 1927: 1–194.
Sacco, F. 1899. I molluschi dei terreni terziari del Piemonte e della Liguria, parte 27. Cardiidae. 25 pp. Carlo Clausen, Torino.
Santos, A. and Mayoral, E. 2007. Paleoecología de la malacofauna de bivalvos del Mioceno superior de Cacela (SE Portugal). Treballs del Museu de Geologia de Barcelona 15: 25–49.
Savazzi, E. 1985. Adaptive themes in cardiid bivalves. Neues Jahrbuch für Geologie und Paläontologie Abhandlungen 170: 291–321.
Savazzi, E. and Sälgeback, J. 2004. A comparison of morphological adaptations in the cardiid bivalves Cardium and Budmania. Paleontological Research 8: 221–239. Crossref
Schaffer, F.X. 1910. Die Bivalven der Miocänbildungen von Eggenburg. Abhandlungen der Kaiserlich-Königlichen Geologischen Reichsanstalt 22: 5–112.
Schultz, O. 2003. Bivalvia neogenica (Lucinoidea–Mactroidea). In: W.E. Piller (ed.), Catalogus Fossilium Austriae. Ein systematisches Verzeichnis aller auf österreichischem Gebiet festgestellten Fossilien 1 (2), 381–690. Österreichische Akademie der Wissenschaften, Vienna.
Sieber, R. 1956. Die mittelmiozänen Carditidae und Cardiidae des Wiener Beckens. Mitteilungen der Geologischen Gesellschaft in Wien 47 (for 1954): 183–234.
Smith, L.C. 1945. Notes on the family Cardiidae. Occasional Papers on Mollusks 1: 29–32.
Stein, G., Moths, H., Albrecht, F., Havekost, U., and Fehse, D. 2016. Revision der miozänen Molluskenfauna (Hemmoorium) von Werder bei Achim (Nordwest-Niedersachsen). Palaeofocus 5: 1–289.
Steininger, F.F. and Wessely, G. 2000. From the Tethyan Ocean to the Paratethys Sea: Oligocene to Neogene stratigraphy, paleogeography and palaeobiogeography of the circum-Mediterranean region and the Oligocene to Neogene Basin evolution in Austria. Mitteilungen der Österreichischen Geologischen Gesellschaft 92: 95–116.
Strauss, P., Harzhauser, M., Hinsch, R., and Wagreich, M. 2006. Sequence stratigraphy in a classic pull-apart Basin (Neogene, Vienna Basin). A 3D seismic based integrated approach. Geologica Carpathica 57: 185–197.
Studencka, B. and Studencki, W. 1988. Middle Miocene (Badenian) bivalves from the carbonate deposits of the Wójcza-Pińczów Range (southern slopes of the Holy Cross Mountains, Central Poland). Acta Geologica Polonica 38: 1–44.
Studencka, B. and Zieliński, G. 2013. Strontium isotope dating of bivalve faunas from the Upper Miocene Cacela Formation, eastern Algarve, Portugal: evidence from Messinian bivalve fauna. Geological Quarterly 57: 665–686. Crossref
Studencka, B., Popov, S.V., Bieńkowska-Wasiluk, M., and Wasiluk, R. 2016. Oligocene bivalve faunas from the Silesian Nappe, Polish Outer Carpathians: Evidence of the early history of the Paratethys. Geological Quarterly 60: 317–340. Crossref
Syrides, G.E. 1995. Neogene mollusk faunas from Strymon basin, Macedonia, Greece. First results for biochronology and palaeoenvironment. Geobios 28 (Supplement 1): 381–388. Crossref
Tavani, G. and Tongiorgi, M. 1963. La fauna miocenica delle “Arenarie di Ponsano” (Volterra, prov. di Pisa). Paleontographia Italica 58: 1–41.
Ter Poorten, J.J. 2005. Outline of a Systematic Index. Recent Cardiidae (Lamarck, 1809). Updated 2009 for WoRMS. http://www.conchology.be/en/shelltopics/visaya-net/date.php?year=2005.
Ter Poorten, J.J. 2007. Notes on the identity of Cardium costatum Linnaeus, 1758 with the description of Cardium maxicostatum spec. nov. from tropical West Africa (Bivalvia, Cardiidae). Basteria 71: 57–69.
Ter Poorten, J.J. 2009. The Cardiidae of the Panglao Marine Biodiversity Project 2004 and the Panglao 2005 Deep-Sea Cruise with descriptions of four new species (Bivalvia). Vita Malacologica 8: 9–96.
Ter Poorten, J.J. 2013. Revision of the Recent species of the genus Nemocardium Meek, 1876 (Bivalvia, Cardiidae), with the descriptions of three new species. Basteria 77 (4–6): 45–73.
Ter Poorten, J.J. and Gofas, S. 2012 Cardium indicum Lamarck, 1819. Accessed 2017 through WoRMS. http://www.marinespecies.org/aphia.php?p=taxdetails&id=138997
Toula, F. 1901. Eine marine Neogenfauna aus Cilicien. Jahrbuch der Kaiserlich-Königlichen Geologischen Reichsanstalt 51: 247–263.
Van Aartsen, J.J. and Giannuzzi Savelli, R. 1991. New names for well-known European marine Mollusca. Bollettino Malacologico 27: 1–8.
Van Vliet-Lanoë, B., Gosselin, G., Mansy, J.-L., Bourdillon, C., Meurisse-Fort, M., Henriet, J.-P., Le Roy, P., and Trentesaux, A. 2010. A renewed Cenozoic story of the Strait of Dover. Annales de la Société Géologique du Nord 17 (2 série): 59–80.
Venzo, S. 1933. I fossili del Neogene Trentino, Veronese e Bresciano. I. Pesci, Crostacei e Lamellibranchi. Palaeontographia Italica 34 (n.s. 4): 31–84.
Venzo, S. and Pelosio, G. 1963. La malacofauna Tortoniana del colle di Vigoleno (Preappennino Piacentino). Palaeontographia Italica 63 (n.s. 28): 43–213.
Vermeij, G.J. 2012. The tropical history and future of the Mediterranean biota and the West African enigma. Journal of Biogeography 39: 31–41. Crossref
Vidal, J. 1994. A review of the genus Fulvia Gray, 1853 (Mollusca, Cardiidae). Apex 9: 93–118.
Vidal, J. 1999. Taxonomic review of the elongated cockles: genera Trachycardium, Vasticardium and Acrosterigma (Mollusca, Cardiidae). Zoosystema 21: 259–335.
Vidal, J. 2000. Genus Vepricardium Iredale 1929 (Bivalvia, Cardiidae) with description of a new species from Thailand, Vepricardium albohamatum Hylleberg and Vidal. Phuket Marine Biological Center Special Publication 21: 447–464.
Weinkauff, H.C. 1862. Catalogue des coquilles marines recueillies sur les côtes de l’Algérie. Journal de Conchyliologie 10: 301–371.
Wienrich, G. 1999. Die Fauna des marinen Miozäns von Kevelaer (Niederrhein). Band 2 Bivalvia, Scaphopoda, Cephalopoda, Bryozoa, Annelida, Brachiopoda. 196 pp. Backhuys Publishers, Leiden.
Zachos, J., Pagani, M., Sloan, L., Thomas, E., and Billups, K. 2001. Trends, rhythms, and aberrations in global climate 65 Ma to present. Science 292: 686–693. Crossref
Zbyszewski, G. 1957. Le Burdigalien de Lisbonne. Comunicações dos Serviços Geológicos de Portugal 38: 91–215.
Zunino, M. and Pavia, G. 2009. Two new species of Rissoina (Gastropoda: Rissoidae) in the Lower Miocene of Valle Ceppi (Torino, NW Italy). Bollettino della Società Paleontologica Italiana 48: 51–57.
Acta Palaeontol. Pol. 62 (4): 729–757, 2017
https://doi.org/10.4202/app.00363.2017