

New sauropodomorph and cynodont remains from the Late Triassic Sacisaurus site in southern Brazil and its stratigraphic position in the Norian Caturrita Formation
JÚLIO C.A. MARSOLA, JONATHAS S. BITTENCOURT, ÁTILA A.S. DA ROSA, AGUSTÍN G. MARTINELLI, ANA MARIA RIBEIRO, JORGE FERIGOLO, and MAX C. LANGER
Marsola, J.C.A., Bittencourt, J.S., Da Rosa, Á.A.S., Martinelli, A.G., Ribeiro, A.M., Ferigolo, J., and Langer, M.C. 2018. New sauropodomorph and cynodont remains from the Late Triassic Sacisaurus site in southern Brazil and its stratigraphic position in the Norian Caturrita Formation. Acta Palaeontologica Polonica 63 (4): 653–669.
Sacisaurus agudoensis is the only silesaurid known from the Triassic beds of the Santa Maria Supersequence and the correlation of its type locality to the other Triassic deposits of south Brazil has always been controversial. In an attempt to improve this, a handful of dinosaur and cynodont remains found associated to S. agudoensis are here described and compared. The anatomy of the sauropodomorph is more similar to that of Norian forms such as Pantydraco caducus and Unaysaurus tolentinoi than to that of Carnian taxa such as Saturnalia tupiniquim and Pampadromaeus barberenai. The cynodonts recovered based on isolated teeth include a brasilodontid and a Riograndia-like form. This assemblage is consistent with a Norian age, as is also suggested by local stratigraphic correlation, which positions the site in the Caturrita Formation.
Key words: Dinosauria, Sauropodomorpha, Dinosauriformes, Cynodontia, Triassic, Norian, Caturrita Formation, Brazil.
Júlio C.A. Marsola [juliomarsola@gmail.com] and Max C. Langer [langer.mc@gmail.com], Departamento de Biologia, FFCLRP, Universidade de São Paulo, Ribeirão Preto, SP, 14040-901, Brazil.
Jonathas S. Bittencourt [sigmaorionis@yahoo.com.br], Departamento de Geologia, Universidade Federal de Minas Gerais, Belo Horizonte, MG, 31270-901, Brazil.
Átila A.S. Da Rosa [atila@smail.ufsm.br], Laboratório de Estratigrafia e Paleobiologia, Departamento de Geociências, Universidade Federal de Santa Maria, Santa Maria, RS, 97.105-900, Brazil.
Agustín G. Martinelli [agustin_martinelli@yahoo.com.ar], Laboratório de Paleontologia de Vertebrados, Departamento de Paleontologia e Estratigrafia, Instituto de Geociências, Universidade Federal do Rio Grande do Sul, Porto Alegre, RS, 91540-000, Brazil.
Ana Maria Ribeiro [ana.ribeiro@fzb.rs.gov.br] and Jorge Ferigolo [jorgeferigolo@gmail.com], Seção de Paleontologia, Museu de Ciências Naturais, Fundação Zoobotânica do Rio Grande do Sul, Porto Alegre, RS, 90690-000, Brazil.
Received 23 April 2018, accepted 25 September 2018, available online 29 October 2018.
Copyright © 2018 J.C.A. Marsola et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
During the last decades, the study of dinosaur origins has advanced greatly. This includes newly described fossils (e.g., Apaldetti et al. 2018; Müller et al. 2018; Pretto et al. 2018), new geological data (Whiteside et al. 2015; Marsicano et al. 2016; Bernardi et al. 2018; Langer et al. 2018), quantitative macroevolutionary analyses (Brusatte et al. 2008a, b; Sookias et al. 2012; Benton et al. 2014), and challenging taxonomic re-evaluations (Agnolín and Rozadilla 2017; Baron 2017; Baron et al. 2017a, b; Langer et al. 2017). In much of these discussions silesaurids have played a pivotal role. These dinosauromorphs have been suggested to represent the sister-group of dinosaurs (e.g., Nesbitt et al. 2010; Nesbitt 2011; Martínez et al. 2011; Bittencourt et al. 2015), bearing features previously considered dinosaur synapomorphies (Dzik 2003; Langer et al. 2013). Alternatively, other silesaurid features may suggest their affinities with ornithischian dinosaurs (Ferigolo and Langer 2007; Langer and Ferigolo 2013; Cabreira et al. 2016).
Harbouring some of the oldest dinosaur records worldwide (e.g., Cabreira et al. 2016; Pretto et al. 2018), the continental Late Triassic deposits of southern Brazil have also yielded other dinosauromorphs, like the lagerpetid Ixalerpeton polesinensis (Cabreira et al. 2016) and the silesaurid Sacisaurus agudoensis (Ferigolo and Langer 2007). The type locality of the latter dinosauromorph was explored during 2000–2001 in a series of field expeditions led by JF and the crew of Fundação Zoobotânica do Rio Grande do Sul, in the context of the IDB (Inter-American Development Bank) funded Pró-Guaíba Project. This work unearthed the rich material attributed to S. agudoensis (Ferigolo and Langer 2007), as well as cynodont (Ribeiro et al. 2011) and dinosauromorph remains (Langer and Ferigolo 2013). The cynodont remains include four isolated teeth and the dinosauromorph is represented by a handful of isolated bones (see Material and methods), which are larger than the fossils attributed to S. agudoensis. The morphology of some of the overlapping elements with those of S. agudoensis (i.e., ilium, femur) indicates that they correspond to a different taxon, with further non-duplicated larger elements (i.e., ectopterygoid, cervical vertebra, metatarsal I) also tentatively attributed to that taxon.
This work aims at fully describing these dinosauromorph and cynodont specimens, inferring their phylogenetic affinities and signal for faunal correlation. Indeed, the correlation of the Sacisaurus site to other tetrapod-bearing localities of the Santa Maria and Caturrita formations is not strongly constrained (Langer et al. 2007). As such, we also attempt here to provide geological and biochronological data to more strongly define the stratigraphic position of that site.
Institutional abbreviations.—AMNH FARB, American Museum of Natural History, New York, U.S.A; BPI, Evolutionary Studies Institute, Johannesburg, South Africa (formerly Bernard Price Institute); MB. R., Museum für Naturkunde, Berlin, Germany; MCN, Museu de Ciências Naturais, Fundação Zoobotânica do Rio Grande do Sul, Porto Alegre, Brazil; MCP, Museu de Ciências e Tecnologia, PUCRS, Porto Alegre, Brazil; NHMUK, Natural History Museum, London, United Kingdom; PULR, Universidad Nacional de La Rioja, La Rioja, Argentina; PVSJ, Museo de Ciencias Naturales, San Juan, Argentina; SAM-PK, Iziko South African Museum, Cape Town, South Africa; SMNS, Staatliches Museum für Naturkunde, Stuttgart, Germany; UFRGS, Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil; UFSM, Universidade Federal de Santa Maria, Santa Maria, Brazil; ULBRA, Museu de Ciências Naturais, Universidade Luterana do Brasil, Canoas, Brazil.
Other abbreviations.—M., muscle.
Geological setting
The Sacisaurus site is located at 19°43’12’’ S, 47°45’04’’ W; inside the western urban area of the city of Agudo, state of the Rio Grande do Sul, Brazil. Due to the urban expansion, the outcrop is now located on the western margin of Independência Street, north to Concordia Avenue. Little of the original outcrop remains, but we provide here a sedimentary profile based on previous studies of the authors (Fig. 1A).
The profile is more than eight meters deep and is composed of an intercalation of fluvial (CH) and overbank (CR) deposits. It starts with a fine sandstone where we recorded ex situ Exaeretodon specimens. Still in the lowest meter of the profile, reddish mudstone and a brownish fine sandstone are visible. The latter shows an upwards coarsening trend, with mud intraclasts at the top, forming a millimetric intraformational conglomerate. Above this conglomerate, there is an intercalation of reddish mudstone and greenish fine sandstone that preserved most fossils in the site. The rest of the profile is formed of yellowish to pinkish fine sandstone with millimetric intercalation of brown mudstone. These present a lobe or tabular geometry and are generally massive, but sometimes bear horizontal lamination and trough cross-bedding. Whereas the basalmost levels represent traction and suspension deposition probably in an oxbow lake, the uppermost sandstone are linked to crevassing (Da Rosa 2015). These lithologies correspond to the Caturrita Formation (sensu Andreis et al. 1980), i.e., upper portion of the Candelária Sequence, Santa Maria Supersequence (sensu Horn et al. 2014).
Nearby outcrops allow for a better stratigraphic correlation (Fig. 1), as only crevasse deposits are recorded at “Ki-Delicia” and “ASERMA” sites. The slightly more distant (about 1.8 km), and much better known, Janner outcrop (Pretto et al. 2015) has equivalent lithologies at its top, whereas the highly fossiliferous reddish mudstone of the middle and lower part of the outcrop correspond to the Santa Maria Formation (Da Rosa 2005, 2015).
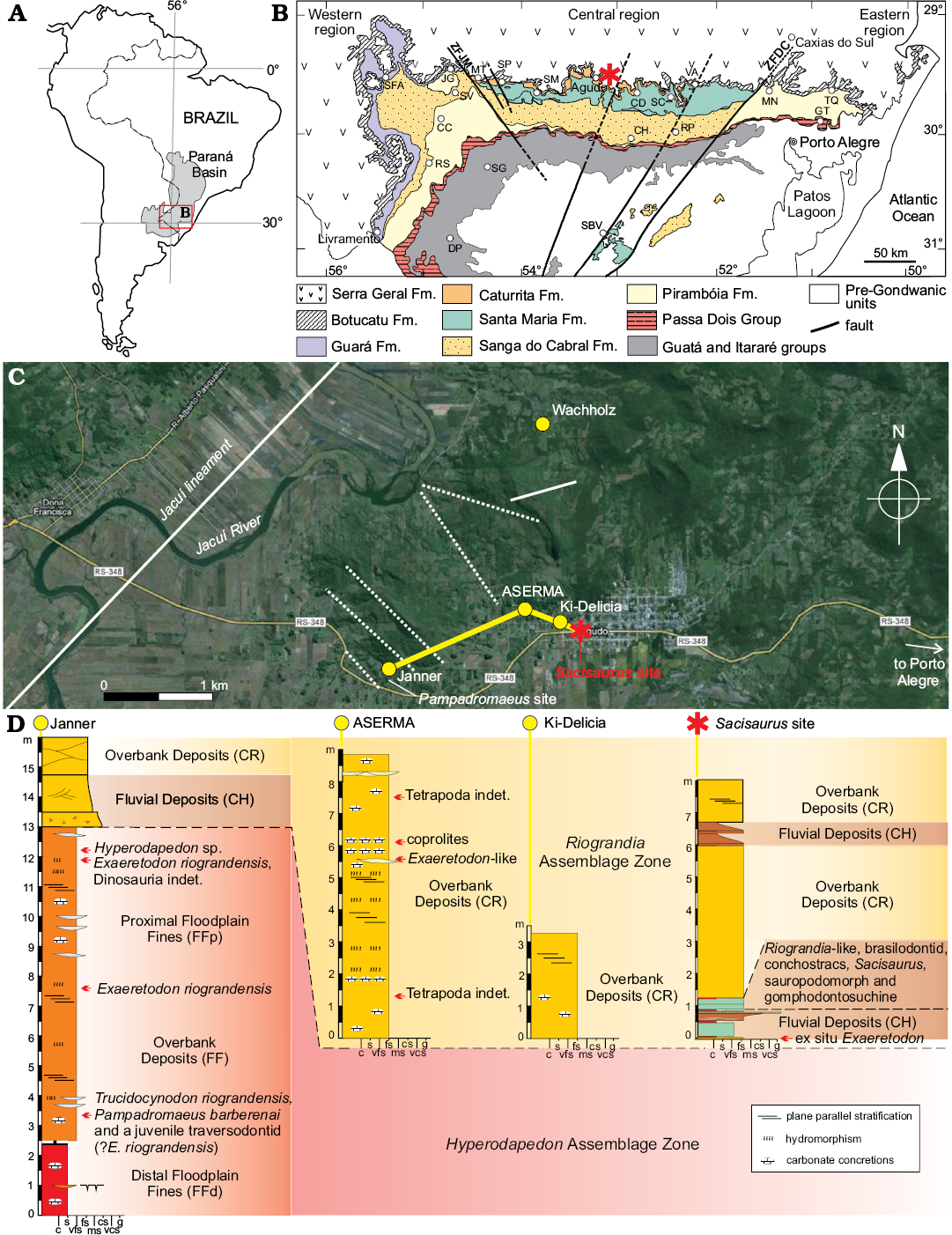
Fig. 1. A. Geographic location of Paraná Basin in South America. B. Geological map of Rio Grande do Sul State (modified from Da Rosa 2015), showing location of the Sacisaurus type locality (asterisk). C. Map of the study area showing location the Sacisaurus type locality and its correlations to neighboring sites. D. Sedimentary profile of the Sacisaurus type locality and neighboring sites. Abbreviations: CC, Cacequi; CD, Candelária; CH, Cachoeira do Sul; DP, Dom Pedrito; JG, Jaguari; GT, Gravataí; MN, Montenegro; MT, Mata; RP, Rio Pardo; RS, Rosário do Sul; SBV, Santana da Boa Vista; SC, Santa Cruz do Sul; SFA, São Francisco de Assis; SG, São Gabriel; SM, Santa Maria; SP, São Pedro do Sul; TQ, Taquari; VA, Venâncio Aires; ZFDC, Dorsal do Canguçu Fault Zone; ZFJM, Jaguari-Mata Fault Zone; c, clay; fs, fine sand; g, gravel; ms, medium sand; s, silt; vcs, very coarse sand; vfs, very fine sand.
Material
Bones of the larger dinosauromorph found at the Sacisaurus site were not recovered articulated or closely associated, so there is no clear evidence that they correspond to a single individual. This is corroborated by the preserved right and left femora, which share a very similar anatomy, but are of slightly different sizes. As a whole, the material has a similar taphonomic signature and includes a right ectopterygoid (MCN PV 10049), one cervical vertebra (MCN PV 10027), a right ilium (MCN PV10026), right (MCN PV10007) and left (MCN PV10008) femora, and the distal end of a metatarsal I (MCN PV 10049). As mentioned above, the attribution of these specimens to the same taxon is tentative, based on their similar phylogenetic signal (discussed below) and the application of topotypic principles. As for the cynodont teeth, these include two isolated right lower postcanines (MCN PV 10202-3), an isolated postcanine (MCN PV 10204), and an isolated left lower incisor (MCN PV 10205).
Results
Ectopterygoid (MCN PV 10049).—The bone is nearly complete, missing only the rostral tip of the lateral process and the medial contour of the medial process (Fig. 2), precluding the proper assessment of its articulation with the pterygoid. As preserved, the ectopterygoid is slightly longer (rostrocaudally) than lateromedially wide. The lateral process is hook-shaped and arches rostrally, forming a pounded caudal margin with the medial process. Its lateral half flattens lateromedially, and it is dorsoventrally higher compared to its medial half. Its articulation with the jugal is marked by scars on the caudolateral margin. Although missing its rostral-most tip, it is clear that such articulation is tabular, differing from the T-shaped profile of Plateosaurus engelhardti (Prieto-Márquez and Norell 2011). The medial half of the lateral process expands rostrocaudally towards the medial process, which is much longer rostrocaudally than the former.
The medial process is flange-like, dorsoventrally expanded, and bears a medially directed dorsal margin. Its ventral surface is excavated by a semicircular depression, which has been considered pneumatic and typical of theropods (e.g., Rauhut 2003). Yet, Nesbitt (2011) noticed that this feature simply marks the articulation with the pterygoid, as seen in Eoraptor lunensis, Pantydraco caducus, Plateosaurus engelhardti, Liliensternus liliensterni, and Coelophysis bauri. The medial process has a long rostral projection resembling that of Pantydraco caducus (Yates 2003; Galton and Kermack 2010), and differing from that of “Syntarsus” rhodesiensis (Raath 1977), Allosaurus fragilis (Nesbitt 2011), Plateosaurus engelhardti (Prieto-Márquez and Norell 2011), and Lesothosaurus diagnosticus (Porro et al. 2015), which do not present such a projection.
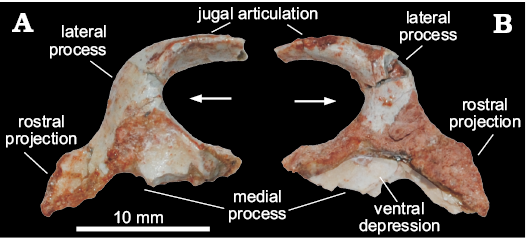
Fig. 2. Right ectopterygoid of a sauropodomorph from Sacisaurus site, Late Triassic Caturrita Formation, Agudo, Rio Grande do Sul, Brazil; MCN PV 10049 in dorsal (A) and ventral (B) views. Arrows point to cranial.
Cervical vertebra (MCN PV 10027).—The only preserved vertebra (Fig. 3) is incomplete and somewhat distorted, missing the cranioventral portion of the centrum, the zygapophyses, the neural spine, and most of the parapophyses and diapophyses. Together, the dorsal position of the parapophyses in the centrum, the well-developed diapophyses, the elongated centrum, and the presence of a ventral keel, indicate that MCN PV 10027 represents a cervical vertebra, possibly from the caudal region (8th or 9th element) of the neck. For descriptive purposes, the laminae and fossae nomenclature of Wilson (1999, 2012) and Wilson et al. (2011) will be adopted.
The centrum has an elongated profile, at least three times craniocaudally longer than dorsoventrally high. The ventral portion of the centrum bears a stout ventral keel, and only the caudal half of this keel is preserved. As such, the keel is mediolaterally thicker caudally, i.e., more than seven times thicker in its caudal portion than cranially at the middle of the centrum (Fig. 3D). That thicker portion projects further ventrally than the caudal articulation of the centrum. The surface surrounding that articulation is heavily scarred, mainly on its ventral margin. In ventral view, the centrum is spool-shaped, with its caudal rim 1.75 times wider than its middle portion. The caudal articular face of the centrum is deeply concave. In caudal view (Fig. 3B), it is far wider than high due to an exaggeration caused by taphonomic distortion. In lateral view, the margin of the caudal articular face is oblique to the long axis of the centrum, so that its ventral edge extents further caudally than the dorsal. The neurocentral suture is visible mainly along the caudal portion of the vertebra, suggesting an advanced closure stage, approaching somatic maturity (see Irmis 2007 for more details on ontogeny in archosaurs). The dorsal surface of the centrum has a well-developed fossa (Fig. 3), which is remarkably deep, dorsoventrally narrow, and craniocaudally elongated. Most of both parapophyses are missing due to breakages. Each is represented by a subtle and short oblique (cranioventrally to caudodorsally oriented) ridge set at the dorsal part of the cranial margin of the centrum, close to the neural arch, fading away caudodorsally in the direction of the anterior centrodiapophyseal lamina.
The neural arch bears well-developed laminae and fossae. The cranial portion of the prezygodiapophyseal laminae is missing, but its well-developed caudal part is clearly present in left lateral view (Fig. 3). This lamina roofs a deep prezygadiapophyseal centrodiapophyseal fossa and extends further laterally than the anterior centrodiapophyseal lamina (Fig. 3), which is short and does not reach the parapophysis. The centrodiapophyseal fossa is deep and craniocaudally elongated. It is set caudal to the prezygadiapophyseal centrodiapophyseal fossa, with its cranial half roofed by a lateroventrally directed, well-developed diapophysis, from the dorsal surface of which a well-developed postzygodiapophyseal lamina (Fig. 3) arises. This lamina extends towards the base of the postzygapophysis (which is not preserved), marking the craniodorsal margin of a deep postzygadiapophyseal centrodiapophyseal fossa. The fossa of the right side (Fig. 3C) is divided in two by a subtle ridge that expands cranioventraly from the middle of the postzygodiapophyseal lamina. An equivalent ridge is, however absent from the left side of the vertebra. The postzygadiapophyseal centrodiapophyseal fossa also overlaps the caudal half of the centrodiapophyseal fossa dorsally. The posterior centrodiapophyseal lamina roofs the dorsal and caudal portions of the centrodiapophyseal fossa, also forming the whole ventral edge of the postzygadiapophyseal centrodiapophyseal fossa.
The dorsal surface of the neural arch (Fig. 3A) has several ridges, which we tentatively homologize with the laminae of later saurischians. The cranial surface of the neural spine has a deep, dorsoventrally short and craniocaudally long V-shaped spinoprezygapophyseal fossa (Fig. 3) at the base. Its lateral margins are formed by subtle spinoprezygapophyseal laminae that reach the medial surface of the prezygapophyses, where they bifurcate. Another pair of ridges (Fig. 3) form the lateral margin of the prezygapophyses, extending caudally along the dorsal surface of the neural arch to reach the lateral base of the neural spine caudal to the spinoprezygapophyseal fossa. Ventrolateral to that, there is another pair of faint craniocaudally directed ridges (Fig. 3). Also, subparallel to the neural spine, a pair of inconspicuous low ridges (Fig. 3) extend on the dorsal surface of the neural arch. Although its caudal end is missing, it most probably corresponds to the epipophyseal–prezygapophyseal lamina (Wilson 2012).
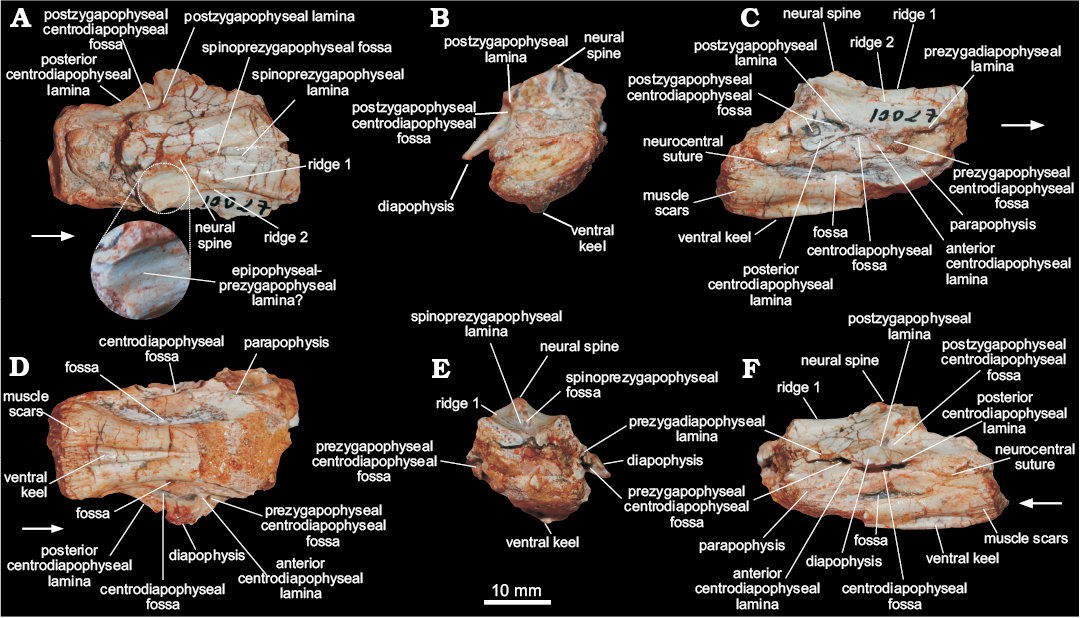
Fig. 3. Cervical vertebra of a sauropodomorph from Sacisaurus site, Late Triassic Caturrita Formation, Agudo, Rio Grande do Sul, Brazil; MCN PV 10027 in dorsal (A), caudal (B), right lateral (C), ventral (D), cranial (E), and left lateral (F) views. The zoomed area in A shows the epipophyseal-prezygapophyseal lamina. Arrows point to cranial.
Neck vertebrae with epipophyses have been regarded as a dinosaur synapomorphy (Langer and Benton 2006; Nesbitt 2011). Although the epipophyses are not preserved in MCN PV 10027, the ridge that we hypothesize to be the epipophyseal–prezygapophyseal lamina is an indirect evidence of their presence. A similar condition is present in the 8th cervical vertebra of Panphagia protos (PVSJ 874), in which a craniomedially directed incipient ridge comes from the epipophysis. This condition is clearer in later saurischians, such as the sauropodomorphs Adeopapposaurus mognai (PVSJ 610) and Massospondylus carinatus (BPI 4934, SAM PK K 388), and neotheropods like Elaphrosaurus bambergi (Rauhut and Carrano 2016). No epipophyses have been observed in the caudal cervical vertebrae of ornithischians (Sereno et al. 1993; Langer and Benton 2006; Nesbitt 2011), but these are present in non-dinosauromorph archosauromorphs, such as Batrachotomus kupferzellensis and Tanystropheus longobardicus (Langer and Benton 2006; Nesbitt 2011; Ezcurra 2016). Yet, the morphology of MCN PV 10027, with well-developed fossae and laminae, shows a closer resemblance to dinosaurs than to other archosaurs.
Carnian dinosaurs, such as Eoraptor lunensis (Sereno et al. 2012), Panphagia protos (Martínez and Alcober 2009), and Herrerasaurus ischigualastensis (Sereno and Novas 1993) lack the well-developed fossae and laminae seen in MCN PV 100027. These are also mostly absent in pseudosuchians, although deep fossae and prominent laminae are present in some poposaurids and crocodyliforms (Nesbitt 2005, 2007; Wedel 2007). Among later saurischians, neotheropods are widely recognized by their pneumatic vertebrae, as seen in early forms like Coelophysis bauri and Liliensternus liliensterni (see Benson et al. 2012). This condition is characterized by cervical vertebrae with well-developed laminae and deep fossae pierced by large foramina (Britt 1993; O’Connor 2006, 2007). On the other hand, early sauropodomorphs lack the deep fossae seen in neotheropods (Wedel 2007; Adeopapposaurus mognai, PVSJ 610) and in MCN PV 100027, although the caudalmost vertebra of the cervical series of Plateosaurus engelhardti (SMNS 13200) has well-developed fosssae and prezygodiapophyseal, centrodiapophyseal, and postzygodiapophyseal laminae, resembling the condition of MCN PV 100027. Neotheropods differ from MCN PV 100027 because they bear at least one pair of deep pleurocoels in the cranial portion of the centrum, which are set either caudodorsal or dorsal to the parapophyses (Tykoski 2005). Although the cranial portion of the centrum of MCN PV 100027 is damaged, the remaining morphology does not suggest the presence of such a feature (Fig. 3C, F). Furthermore, a second pair of pleurocoels may be present caudal to the aforementioned pleurocoels in the cranial portion of the centrum, as in the neotheropod Coelophysidea (Tykoski 2005) and in Elaphrosaurus bambergi (Rauhut and Carrano 2016), although not in the neotheropod Liliensternus liliensterni (MB. R. 2175) this is also not present in MCN PV 100027.
Ilium (MCN PV10026).—The right ilium (Fig. 4) is nearly complete, missing the cranial tip of the preacetabular ala, as well as small portions of the cranial part of the supracetabular crest, the cranioventral part of the acetabular medial wall, and the cranioventral part of the postacetabular ala. The iliac length (7.75 cm) suggests an individual smaller than those referred to Saturnalia tupiniquim (9.73 cm, MCP 3844-PV), Guaibasaurus candelariensis (14.1 cm, UFRGS PV0725T), and Pampadromaeus barberenai (8.9 cm, ULBRA-PVT016). The preacetabular ala is short and subtriangular in lateral view and laterally directed due to the inwards arching of the entire blade (Fig. 4C). The subtriangular shape of the preacetabular ala resembles that present in sauropodomorphs such as Riojasaurus incertus (PULR 56) and Efraasia minor (SMNS 12354), but clearly differs from that of other sauropodomoph remains from the Caturrita Formation (Bittencourt et al. 2012), in which the preacetabular ala is cranially elongated, like in Plateosaurus engelhardti (e.g., SMNS 5715). The preacetabular ala is considerably shorter than the postacetabular ala and its cranial tip is set well caudal to the cranialmost edge of the pubic peduncle. Although the preacetabular ala of Pantydraco caducus (Yates 2003) and Leonerasaurus taquetrensis (Pol et al. 2011) extends cranial to the pubic peduncle, that structure is shorter in most early sauropodomorphs (Cooper 1981; Benton et al. 2000; Langer 2003; Rowe et al. 2011), unlike ornithischians and neotheropods. The preacetabular ala and the pubic peduncle are set at a right angle to one another in MCN PV10026. The concave area between them seems to harbor an incipient preacetabular fossa (Fig. 4), but this cannot be confirmed due to breakages. Dorsally, the preacetabular ala shows a slightly bulged lateral rugose muscle scar (Fig. 4), which has been hypothesized to be related to the insertion of M. iliotibialis (Hutchinson 2001b; Langer 2003), which spans caudally along the dorsal edge of the iliac blade. It is caudally connected to a broad muscle attachment area at the caudal portion of the postacetabular ala, which is related to the insertion of M. iliotibialis (Hutchinson 2001b; Langer 2003; Langer et al. 2010).
The iliac blade is slightly dorsoventrally higher than the height from the supracetabular crest to the ventralmost level of the iliac acetabulum. Its lateral surface, caudal to the preacetabular ala, bears two conspicuous depressions separated by a short, elevated area. The dorsal depression (Fig. 4) is rounded and craniocaudally elongated, as well as larger than the lower one. It extends caudally as to almost reach the postacetabular ala, but its deepest point is immediately dorsal to the ventral depression. The latter (Fig. 4) starts caudal to the preacetabular embayment, extending onto its maximal transverse depth right above the supraacetabular crest.
The postacetabular ala is stout and caudodorsally projected, giving a slightly sigmoidal aspect to the dorsal margin of the ilium in lateral/medial views. Its caudoventral portion bears a pair of longitudinal crests restricted to its caudal half. The medial of those (Fig. 4; “posteromedial lamina/shelf” of Ezcurra 2010; “medial lamina/blade” of Martinez and Alcober 2009) extends along the medial surface of the ilium, forming the dorsal margin of the attachment area for the second primordial sacral rib. Its mid-length is right dorsomedial to the caudal tip of the ventral margin of the postacetabular ala (Fig. 4), which extends caudally from the ischiadic peduncle. The medial margin of the brevis fossa (Fig. 4) is formed by these crests; the ventral margin of the postacetabular ala more cranially and the “medial crest” more caudally. Its lateral margin is, on the other hand, formed by the second, more lateral ridge (Fig. 4); i.e., the brevis shelf. The brevis fossa is shallow, transversely and longitudinally broad, but craniocaudally shorter than in neotheropods (e.g., Liliensternus liliensteni, MB. R. 2175), early ornithischians (e.g., Baron et al. 2017c) and early sauropodomorphs like, Saturnalia tupiniquim (Langer 2003), Chromogisaurus novasi (Ezcurra 2010), and Buriolestes scultzi (Cabreira et al. 2016). Such a reduced brevis fossa resembles that of other sauropodomorphs, such as Plateosaurus engelhardti (SMNS 12950, 80664), Massospondylus carinatus (Cooper 1981), and Adeopapposaurus mognai (Martínez 2009).
The area between the supraacetabular crest and the postacetabular ala is concave, unlike that of earlier sauropodomorphs like Chromogisaurus novasi (Ezcurra 2010), neotheropods (e.g., Liliensternus liliensteni, MB. R. 2175), and some ornithischians (e.g., Scelidosaurus harrisonii, NHM R 1111), which bears a ridge connecting the brevis shelf to the supraacetabular crest. The supraacetabular crest extends cranioventrally as a continuous flange from a rugose area at the level of the ischiadic peduncle, along the pubic peduncle, terminating near its articulation area. It is strongly expanded lateroventrally, but not as much as in theropods (e.g., Coelophysis bauri, AMNH FARB 7230, AMNH FARB 7231, AMNH FARB 7233). Its point of maximal transverse breadth is above the center of the acetabulum, equally distant from the distal tips of the ischiadic and pubic peduncles. At this point, the supraacetabular crest is nearly set at the mid dorsoventral height of the ilium, unlike Saturnalia (MCP 3844-PV), Pampadromaeus (ULBRA-PVT016), Panphagia (PVSJ 874), Chromogisaurus (PVSJ 845), and Buriolestes (ULBRA-PVT280), in which the supracetabular crest is positioned far below to this point.
The acetabulum is as craniocaudally expanded as the length between the pre- and postacetabular embayments, and relatively deeper dorsoventrally than in early sauropodomorphs, e.g., Saturnalia tupiniquim, Panphagia protos, and Chromogisaurus novasi (Marsola et al. in press). Its medial wall is ventrally projected, the ventral margin of which levels with the ventral tip of both ischiadic and pubic peduncles. This feature suggests a closed iliac acetabulum, unlike massospondylid and plateosaurid sauropodomorphs, neotheropods, and most ornithischians (see Baron et al. 2017c), but similar to that of some non-dinosaur dinosauromorphs (Langer et al. 2013), and the early sauropodomorphs S. tupiniquim (Langer 2003), P. protos (Martínez and Alcober 2009), and Buriolestes schultzi (Cabreira et al. 2016). There is a shallow ventral notch between the ischiadic peduncle and the ventral margin of the acetabulum, which sets the ventral limit of the ovoid dorsoventrally elongated antitrochanteric area of the acetabulum. The cranioventral portion of the acetabulum has a short vertical ridge forming an angle of 45° to the long axis of the pubic peduncle. As described in other dinosaurs (Langer et al. 2010), this ridge is set medially to the supraacetabular crest, producing a subtriangular ventral fossa (Fig. 4) on the craniolateral corner of the acetabulum.
The pubic peduncle is as long as the extension between the pre- and postacetabular embayments. Its distal portion is dorsoventrally deeper and transversely narrower than the proximal portion. The craniodorsal surface bears a subtle ridge as is also present in Pampadromaeus barberenai (ULBRA-PVT016). Medially the peduncle is mostly flat, with rugose distal areas. Its articulation surface for the pubis faces cranioventrally, is somewhat rounded in outline and craniocaudally elongated, with the medial margin flatter than the lateral. The ischiadic peduncle is short and subtriangular, similar to that of Saturnalia tupiniquim (Langer, 2003). It is laterally bulged, producing a low mound-like process in ventral view that is the acetabular antitrochanter. The articular surface for the ischium is caudoventrally facing, but lacks the caudal heel present in other sauropodomorphs, like P. barberenai (ULBRA-PVT016), Riojasaurus incertus (PULR 56), Coloradisaurus brevis (Apaldetti et al. 2013), and Efraasia minor (SMNS 12354).
The medial surface of the ilium has a complex anatomy, encompassing the concavities and crests that mark the articulation of the sacral vertebrae. Just caudal to the preacetabular embayment, a dorsoventrally elongated concavity (Fig. 4) is present, cranially bounded by a vertical sharp ridge (Fig. 4) formed by the medial edge of the preacetabular fossa. Its dorsal, ventral and caudal margins are formed by a gently elevated, continuous margin. Caudal to that, there is another concavity for attachment of the first primordial sacral vertebra (Fig. 4), which is rounded and craniocaudally longer than the former. The dorsal part of those concavities is formed by a continuous craniocaudally elongated depression that probably received the sacral transverse processes. Its dorsal margin is formed by a longitudinally oriented elevated margin, dorsal to which the medial iliac surface is marked by a spread rugose surface serving for muscle attachment sites, as is also the case of the surface bellow the articulation areas (Fig. 4). More caudally, the medial surface of the postacetabular ala is marked by the “medial crest”, which extends cranially as a subtler ridge. This forms the medial margin of the caudal part of the brevis fossa, which is as medially projected as in Thecodontosaurus antiqus (BRSUG 23613), Efraasia minor (SMNS 12354), and Plateosaurus engelhardti (SMNS 12950), but less markedly so in Massospondylus carinatus (BPI 5238) and Sarahsaurus aurifontanalis (TMM 43646-2). Dorsal to the “medial crest”, there is a longitudinal groove (Fig. 4) that received the transverse process of the second primordial sacral vertebra and merged cranially to the aforementioned “elongated depression”. The attachment site for the corresponding rib is a depressed subtriangular area (Fig. 4) ventral to the “medial crest”. This arrangement matches that of dinosaurs in which two vertebrae form the bulk of the sacrum, as in Saturnalia tupiniquim (Langer 2003) and Staurikosaurus pricei (Bittencourt and Kellner 2009). Yet, it is also possible that the cranial, dorsoventrally elongate depression (Fig. 4B) represents the articulation of the rib of a trunk vertebra incorporated into the sacrum, as seen in one of the paratypes of S. tupiniquim (MCP 3845-PV; Marsola et al. in press).
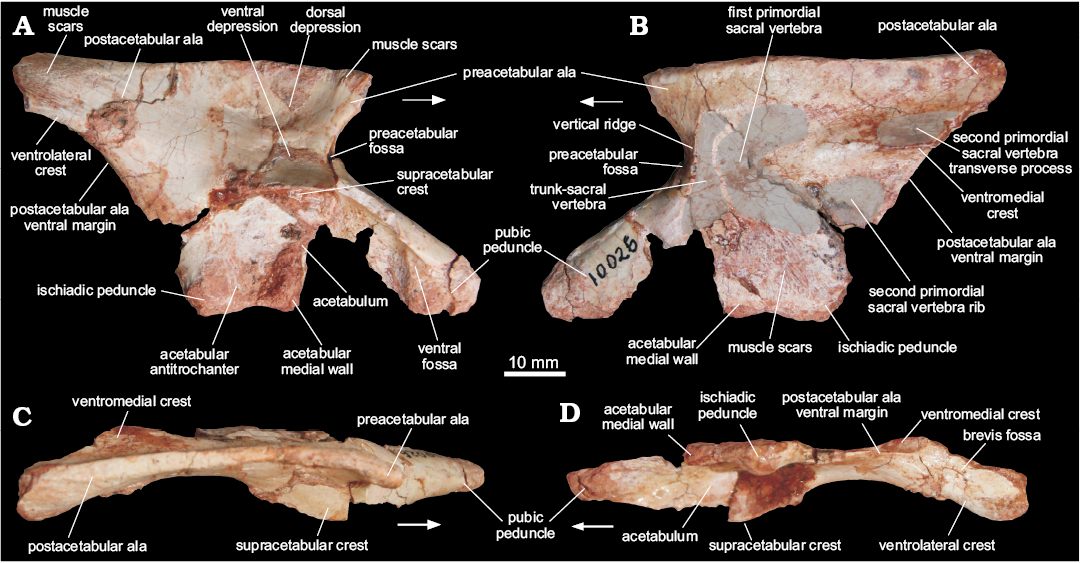
Fig. 4. Right ilium of a sauropodomorph from Sacisaurus site, Late Triassic Caturrita Formation, Agudo, Rio Grande do Sul, Brazil; MCN PV10026 in lateral (A), medial (B), dorsal (C), and ventrocaudal (D) views. Arrows point to cranial.
Femora (MCN PV10007, MCN PV10008).—Even though the femora do not overlap (as they are from different sides) and share a very similar morphology, they probably represent two individuals, as size estimation suggests the right element comes from a considerably larger animal. In both femora, portions of the head and distal condyles are missing. As preserved, the left element (Fig. 5) is 12.25 cm long, whereas the right is 14.75 cm (Fig. 6). If complete, the right femur would be probably nearly 15 cm and the left would measure between 13.5–14 cm. In any case, given their matching morphology, a single description will be provided below for both femora.
The femur has a sinuous shape produced by the craniomedial projection of the head and the cranial and medial bowing of the distal half of the shaft. The head fragment associated with the left femur (Figs. 5E, F) is typically dinosaurian in its transverse expansion, housing a rugose articular surface. Both the ligament sulcus and the lateral tuber are not as pronounced as in other dinosauriforms, such as Sacisaurus agudoensis (Langer and Ferigolo 2013; MCN PV 10014), Saturnalia tupiniquim (MCP 3844-PV), Buriolestes schultzi (ULBRA-PVT280), and Eodromaeus murphi (PVSJ 562). The craniomedial tuber forms a rounded margin as seen perpendicular to the long axis of the head (Fig. 5E, F). The head-shaft transition is rounded and contiguous in cranial and caudal views, its craniolateral surface being excavated by a subtle ventral emargination (Fig. 5). There is no evidence of a groove on the proximal surface of the femur, and the caudal structures of the head, including the caudolateral portion of the femoral head (= “greater trochanter”), have not been preserved.
The rugose craniolateral scar (= “anterolateral scar” of Griffin 2018) is located in the craniolateral surface of the proximal part of the femur, just distal to the articular surface, which extends ventrally as the intertrochanteric area. This attachment site may be related to the insertion of the iliofemoral ligament (Griffin and Nesbitt 2016; but see Griffin 2018 for more details). This area is laterally and medially bound by the S-shaped dorsolateral trochanter and the cranial trochanter, respectively. The cranial trochanter is a cranially bulged, roughly triangular process, the rounded dorsal tip of which is well ventral to the femoral head. On the other hand, the cranial trochanter is not as well-developed as in ornithischians and neotheropods (e.g., Langer and Benton 2006; Nesbitt 2011; Baron et al. 2017c; Griffin 2018), resembling the condition present in early saurischians and sauropodomorphs (Novas 1994; Langer 2003, 2004; Langer and Benton 2006; Nesbitt 2011). From the medial margin of the cranial trochanter, the linea intermuscularis cranialis (Figs. 5 and 6) extends ventrally, bordering the medial and lateral surfaces of the shaft. Between the base of the cranial and the dorsolateral trochanter, there is a rugose and slightly elevated area (Figs. 5 and 6) better seen in the left femur, which is probably the insertion site of M. iliofemoralis externus (Hutchinson 2001a) and homologous to the trochanteric shelf of many early dinosauriforms.
As in herrerasaurids (Novas 1994; Bittencourt and Kellner 2009) and sauropodomorphs (Langer 2004; Langer and Benton 2006), the fourth trochanter of MCN PV10007 and PV10008 forms an asymmetrical flange, located on the caudal surface of the proximal half of the shaft. In lateral/medial views, it has the shape of an obtuse triangle, the longest edge of which projects caudoventrally from the shaft, forming a rounded apex on that same direction. In ornithischians, such as Eocursor parvus (Butler 2010), the apex of the fourth trochanter is straighter and more distally projected, as to acquire a pendant shape. Equally different is the symmetrical and lower fourth trochanter of neotheropods, e.g., Dilophosaurus (Welles 1984; UCMP 37302), and non-dinosaurian dinosauromorphs (Langer et al. 2013). In caudal view, the fourth trochanter has a slightly sinuous aspect and its lateral/medial outline shows that it is more expanded distally than proximally. The distal tip of the fourth trochanter is rounded, and its distal margin forms a nearly right angle to the long axis of the femur. In contrast, the distal margin of the fourth trochanter in ornithischians is strongly concave. A proximodistally elongated and transversely broad fossa (Figs. 5 and 6), medially bound a ridge, is located medial to the fourth trochanter and represents the insertion of M. caudofemoralis longus (Hutchinson 2001a; Langer 2003). The shaft surface proximolateral to the fourth trochanter is also concave, encompassing a diagonally oriented oval rugose area (Figs. 5 and 6) for the origin of M. caudofemoralis brevis (Hutchinson 2001a; Griffin and Nesbitt 2016). From the distal edge of the fourth trochanter, another intermuscular line extends distally along the caudomedial margin of the femoral shaft.
The femoral shaft expands transversely at the distal portion, but the exact morphology of the condyles cannot be evaluated due to its poor preservation. The popliteal fossa is enclosed by broad longitudinal ridges ate the caudal surface of the distal part of the femur. The opposite (cranial) surface of the femur is not depressed, i.e., the flexor fossa seen in some saurischians, like neotheropods, is absent. However, that surface is scarred (Fig. 5) for muscle insertion as in Herrerasaurus (Novas 1994) and Saturnalia (Langer 2003).
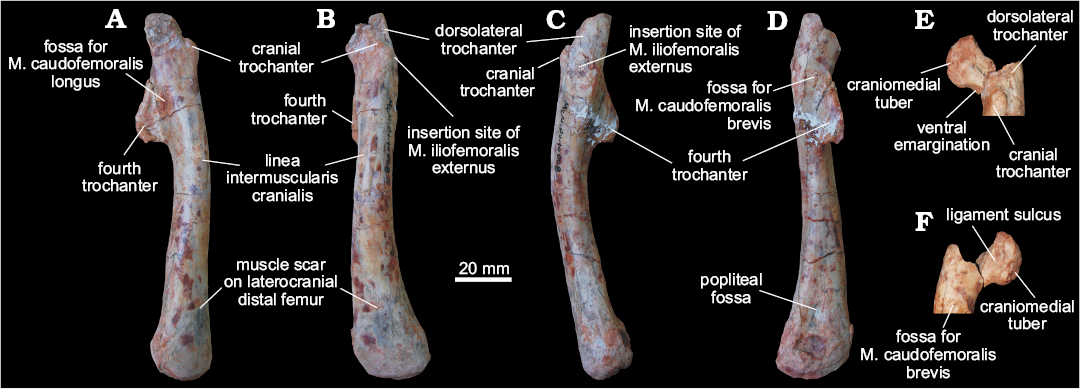
Fig. 5. Left femur of a sauropodomorph from Sacisaurus site, Late Triassic Caturrita Formation, Agudo, Rio Grande do Sul, Brazil; MCN PV10008 in medial (A), cranial (B), lateral (C), and caudal (D) views. The head fragment in articulation with the rest of the bone in craniolateral (E) and caudomedial (F) views. Abbreviation: M., muscle.
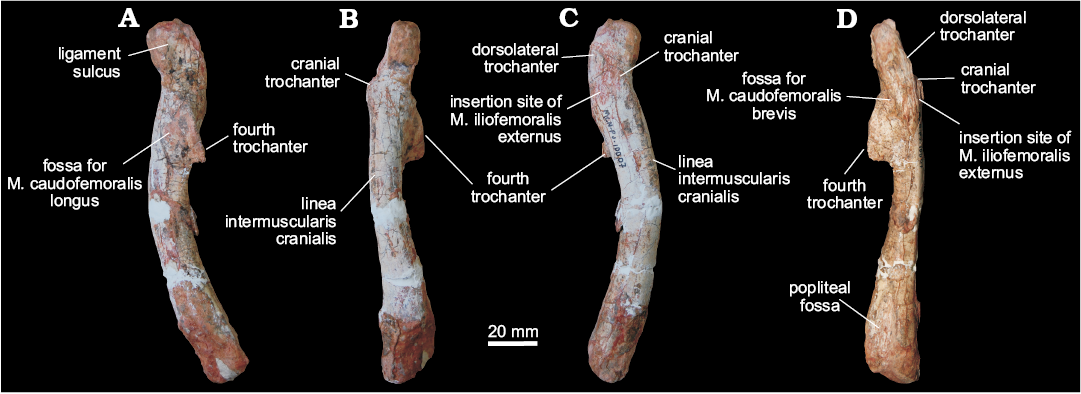
Fig. 6. Right femur of a sauropodomorph from Sacisaurus site, Late Triassic Caturrita Formation, Agudo, Rio Grande do Sul, Brazil; MCN PV10007 in medial (A), cranial (B), lateral (C), and caudal (D) views. Abbreviation: M., muscle.
Metatarsal I (MCN PV 10049).—Only the distal condyles of the left metatarsal I are preserved along with a small portion of the shaft (Fig. 7). The preserved part of the shaft is craniocaudally flattened, with a lateral margin wider than the medial, probably for articulation with metatarsal II, resulting in a subtriangular cross-section. The condyles expand lateromedially compared to the shaft, with the distal articulation being about one third wider than the preserved portion of the latter. The lateral condyle is much larger than the medial, also extending much more distally. It has a roughly triangular distal outline, with rounded corners, formed by lateral, craniomedial, and caudomedial margins. In the same view, the lateral margin of the condyle has a marked concavity, arising from a well-developed ligament pit (Fig. 7), and a laterally projecting caudolateral corner. The medial condyle is small, craniocaudally flattened, and caudomedially directed in distal view, forming a 45° angle to the lateromedial axis of the bone. This shape, along with the distal projection of the lateral condyle, produces a medial displacement of the first digit, as typical of sauropodomorphs as Unaysaurus tolentinoi (UFSM 11069), Efraasia minor (SMNS 12354), Pantydraco caducus (NHMUK RU P77/1), and Leonerasaurus taquetrensis (Pol et al. 2011), but not in Carnian forms like Saturnalia tupiniquim (Langer 2003) and Pampadromaeus barberenai (ULBRA-PVT016). Although the metatarsal I is also known for Guaibasaurus candelariensis, comparisons are hampered by its poor preservation (Langer et al. 2010). In neotheropods, like Coelophysis bauri (e.g., NMMNH P-42200; Rinehart et al. 2009), Dilophosaurus wetherilli (UCMP 37302), and Sinraptor dongi (Currie and Zhao 1993), the medial condyle of metatarsal I is not as medially projected as in sauropodomorphs. This forms a craniocaudally deeper distal articular surface, therefore differing from MCN PV 10049. A condition like that of neotheropods is found in early ornithischians as Abrictosaurus concors (NHMUK RU B.54), Lesothosaurus diagnosticus (NHMUK RU B.17), and Heterodontosaurus tucki (SAM-PK K 1332). The craniomedial surface of the medial condyle bears a shallow ligament pit (Fig. 7), the craniolateral rim of which borders the lateral condyle. The caudolateral margin of the medial condyle forms the medial margin of a lateromedially wider than deep flexor fossa (Fig. 7). Unlike that of Plateosaurus engelhardti (SMNS 13200 Z), and the above cited neotheropods and ornithischians, an equally wider than deep flexor fossa is present in the sauropodomorphs Unaysaurus tolentinoi (UFSM 11069), Efraasia minor (SMNS 12354), Pantydraco caducus (NHMUK RU P77/1), and Leonerasaurus taquetrensis (Pol et al. 2011). Both medial and lateral ligament pits are heavily scarred for ligament insertion.
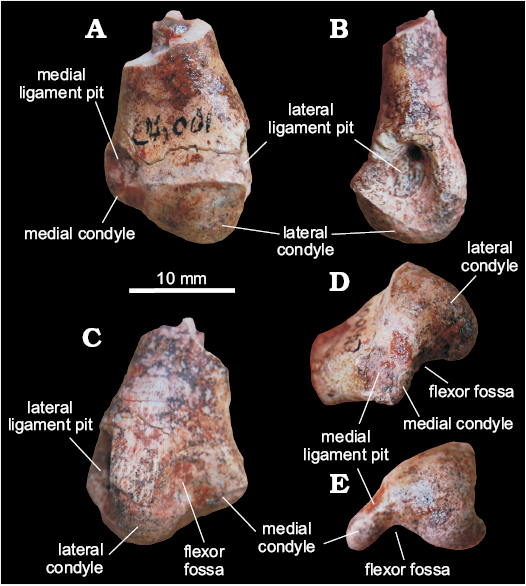
Fig. 7. Distal end of metatarsal I of a sauropodomorph from Sacisaurus site, Late Triassic Caturrita Formation, Agudo, Rio Grande do Sul, Brazil; MCN PV 10049 in cranial (A), lateral (B), caudal (C), mediodistal (D), and distal (E) views.
Cynodont teeth.—The specimen previously referred to Riograndia guaibensis (Ribeiro et al. 2011) corresponds to an isolated postcanine (MCN PV 10204) here interpreted as a left lower element (Fig. 8A). It has a leaf-shaped crown with eight small cusps in mesiodistal line, which are the main features it shares with R. guaibensis (Bonaparte et al. 2001; Soares et al. 2011). The eight cusps have an asymmetrical distribution on the crown; the tallest cusp is preceded by three mesial cups and followed by four distal cusps. The cusps are separated by shallow but long and curved (mesially convex) longitudinal (inter-cusp) grooves located on both lingual and labial sides. Following the curvature of the grooves, the main cusp and the four distal ones are slightly inclined backward. The cusps, especially the main and the mesial ones, have a marked ridge on their lingual surface. The distalmost cusp is located below the level of the mesialmost cusp and is slightly displaced lingually. The labial surface of the crown is convex and the lingual is concave, with the mesiolingual edge of the first cusp forming an elevated ridge. There is no conspicuous crown-root constriction, but the distal border is more concave than the mesial in this area. The latter is almost straight in labial/lingual views. The root is apically open and hollow, with a circular cross-section along all its extension.
In fact, MCN PV 10204 resembles postcanine teeth of Riograndia guaibensis (Fig. 8B, C) in general aspect, but it is also reminiscent of the leaf-shaped crown morphology seen in several Triassic archosauromorph taxa, such as Azendohsaurus, Revueltosaurus, ornithischians, and sauropodomorphs (e.g., Flynn et al. 1999; Barret 2000; Parker et al. 2005; Irmis et al. 2007). Yet, none of the above archosauromorphs have teeth with the unique set of features of MCN PV 10204, e.g., long and mesially convex inter-cusp grooves (= interdenticular sulci of Hendrickx et al. 2014) and crown with a concave lingual surface. Although, the postcanine teeth of Riograndia guaibensis (e.g., UFRGS-PV-833-T, UFRGS-PV-1319-T) typically have more transversely narrow crowns, less developed (i.e., small) cusps and inter-cusp grooves, a considerably less concave lingual surface, and a more transversely flattened root with an incipient longitudinal groove, more conspicuously in the labial side (Fig. 8B, C). Thus, the morphology of MCN-PV 10204 could fit to a more rostral R. guaibensis postcanine tooth, as more caudal teeth increase the cusp number up to 11 (e.g., UFRGS-PV-1319-T). However, a new Riograndia-like cynodont, sister-taxon to R. guaibensis, has been recently reported from Janner site (Martinelli et al. 2016), showing the occurrence of leaf-shape toothed Riograndia-like cynodonts (as that described here) in faunas older than those yielding R. guaibensis.
The specimens originally referred to Brasilitherium riograndensis (Ribeiro et al. 2011) include two isolated right lower postcanines (MCN PV 10202, MCN PV 10203). Their crown morphology is similar, but MCN PV 10202 (Fig. 8D) is larger than MCN PV 10203 (Fig. 8E). Given the crown complexity, they seem to be from the middle to caudal portions of the dental series, and MCN-PV 10202 is possibly from a more posterior position in the series than MCN PV 10203. They are asymmetrical with a main cusp “a” followed by a large cusp “c” and a small cusp “d”. Cusp “b” is smaller than cusp “d” and placed lower in the crown than the remaining cusps. Cusps “a” to “d” are aligned, forming a sectorial crest, with conspicuous mesial and distal cutting edges connecting cusps, whereas cusp “b” is separated from cusp “a” by a concave notch. Both teeth have one mesiolabial and one mesiolingual accessory cusps (the latter possibly corresponding to cusp “e” of Crompton 1974), located below the notch between cusps “a” and “b”. Consequently, in mesial view, cusp “b” is flanked by these two accessory cusps, as not seen in any Brasilodon–Brasilitherium specimens. A distolingual cingulum bears three cusps in MCN-PV 10102 and two discrete cusps in MCN PV 10103. In MCN PV 10102, the most distal cusp is broken at its base, but seems to be larger than the remaining elements. The more mesial cusp, near which the cingulum ends, could correspond to cusp “g”. In MCN PV 10103, the distolingual cusps are considerably larger. Particularly, the more mesially placed (just below the notch between cusps “a” and “c”) is acute and conspicuous, corresponding to cusp “g”. The root grooves are deep both lingually and labially. Particularly in MCN PV 10103, it divides the root in two portions, with the distal one considerably larger mesiodistally and labiolingually, with a strongly convex distal wall.
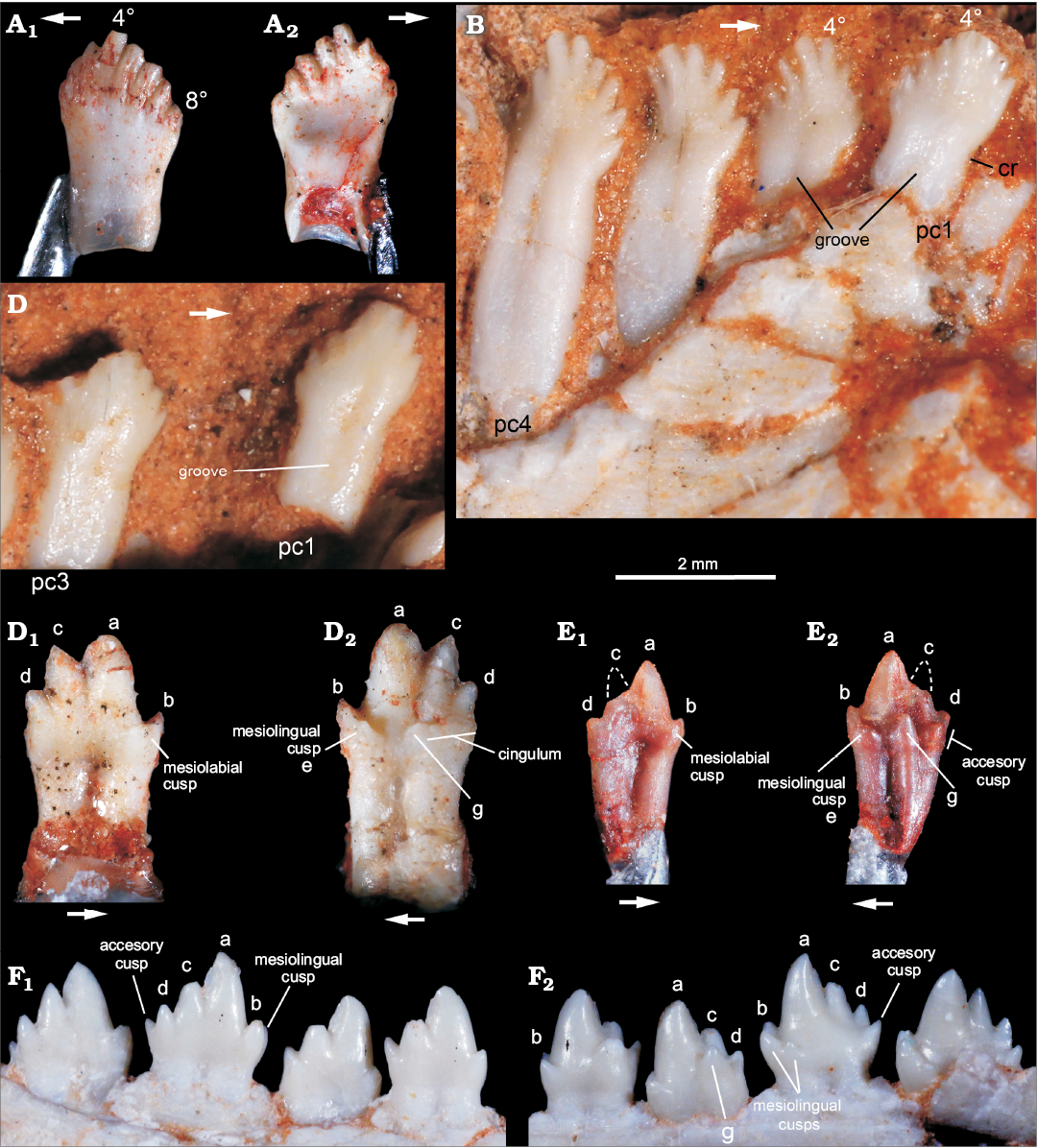
Fig. 8. A. Probainognathian cynodonts from Sacisaurus site, Late Triassic Caturrita Formation, Agudo, Rio grande do Sul, Brazil; isolated left lower postcanine tooth (MCN-PV 10204) originally interpreted as belonging to Riograndia, in labial (A1) and lingual (A2) views. B, C. Riograndia guaibensis Bonaparte, Ferigolo, and Ribeiro, 2001 from the Riograndia Assemblage Zone, Linha São Luiz site, Late Triassic Caturrita Formation, Faxinal do Soturno, Rio grande do Sul, Brazil; anterior postcanines of UFRGS-PV-833-T in lingual view (B) and UFRGS-PV-1319-T in labial view (C). D, E. Riograndia guaibensis from Sacisaurus site, Late Triassic Caturrita Formation, Agudo, Rio grande do Sul, Brazil, originally referred to Brasilitherium riograndensis; isolated right lower postcanines of MCN-PV 10102 (D) and MCN-PV 10103 (E), in labial (D1, E1) and lingual (D2, E2) views. F. Brasilodon quadrangularis Bonaparte, Martinelli, Schultz, and Rubert, 2003 from the Riograndia Assemblage Zone, Late Triassic Caturrita Formation, Faxinal do Soturno, Rio grande do Sul, Brazil, UFRGS-PV-603-T (inverted), detail of middle and posterior left lower postcanines, in labial (F1) and lingual (F2) views. Abbreviations: 4º, fourth cusp; 8º,eighth cusp; a–d, names of lower cusps; (e), cusp e; g, lower cusp g; cr, constriction; pc1, pc4, postcanine tooth 1 and 4.
In general aspect, MCN PV 10202 and MCN PV 10203 resemble more Brasilitherium riograndensis and Brasilodon quadrangularis than any other known probainognathians from South America (e.g., Bonaparte and Barberena 2001; Martinelli et al. 2017a, b). They differ from the postcanine teeth of Alemoatherium huebneri because these lack a mesiolabial accessory cusp, their lingual cusps and cingulum are much less developed and cusp “d” is slightly smaller and lower positioned than cusp “b” (Martinelli et al. 2017b). Besides, Prozostrodon brasiliensis and Botucaraitherium belarminoi (Soares et al. 2014; Pacheco et al. 2018) postcanine teeth have a more continuous lingual cingulum, with a higher number of accessory cusps than MCN PV 10202 and MCN PV 10203. There are also subtle differences between these teeth and those of Brasilitherium riograndensis and Brasilodon quadrangularis. The caudal lower postcanine teeth of those two forms have an accessory distal cusp (distal to cusp “d”) not seen in MCN PV 10202 and MCN PV 10203. The distolingual accessory cusps of MCN PV 10202 (at least three, including a larger distal cusp) and MCN-PV 10203 (two large cusps, the mesial corresponding to cusp “g”) are usually absent in the more caudal teeth of Brasilitherium riograndensis and Brasilodon quadrangularis and at least two cusps (“g” and accessory cusp) are present in their middle postcanine teeth (e.g., UFRGS-PV-603-T, UFRGS-PV-1043-T). Further, a mesiolabial cusp (in addition to mesial cusp “b” and the accessory mesiolingual cusp = cusp “e”) is not seen in any specimen of Brasilitherium riograndensis and Brasilodon quadrangularis, regardless the ontogenetic stage. Such differences do not seem to represent intra-specific variations, as they are unknown in the large available sample of both Brasilitherium riograndensis and Brasilodon quadrangularis (Bonaparte et al. 2003, 2005; Martinelli et al. 2017a, b). Accordingly, although MCN PV 10202 and MCN PV 10203 clearly represent derived prozostrodontians closely related to the “brasilodontids”, the differences in their dentition better indicate a new, still poorly sampled taxon.
The specimen MCN PV 10205 includes an isolated left lower incisor 1 (Fig. 9) similar to those of Exaeretodon spp. (e.g., Abdala et al. 2002). The preserved portion is 28 mm tall, including most of the crown and a small portion of the root. It has an oval cross section at the base, longer labiolingually than mesiodistally. The labial surface is apicobasally convex with a relatively thick layer of enamel that defines mesial and distal cutting edges. The lingual surface is apicobasally concave, so that the tooth is overall slightly curved lingually. That surface is also transversally convex, forming a bulged central area along its entire length, which is separated from the distal cutting edge by an apicobasally oriented groove. An enamel layer is not seen in the lingual surface and the whole element exhibits evident postmortem weathering, with small, irregular pits. Although, the referral of an isolated incisor to any specific taxon is hardly feasible, the morphology of MCN PV 10205 matches of that of gomphodontosuchine traversodontid incisor, in particular the genus Exaeretodon, which is typical of the Hyperodapedon Assemblage Zone (Abdala et al. 2002; Soares et al. 2014) and very common at the Janner site.

Fig. 9. Traversodontidae indet. (cf. Exaeretodon sp.) from Sacisaurus site, Late Triassic Caturrita Formation, Agudo, Rio Grande do Sul Brazil; MCN-PV 10205, isolated left lower incisor 1 in lingual (A) and distal (B) views.
Discussion
The geological review provided here indicates that all fossil-bearing levels of the Sacisaurus site match the usual sandstone upward increase that regionally marks the Caturrita Formation (sensu Andreis et al. 1980) and that the entire site is above the neighbouring mudstone of Janner site (Fig. 1). In palaeontological terms, although the lower levels of the Sacisaurus site, which record Exaeretodon-like cynodonts (Ribeiro et al. 2011), have been roughly correlated to the fossil-bearing mudstone of the Janner site (Langer et al. 2007; Langer and Ferigolo 2013), a younger age has been tentatively assigned to the type-stratum of Sacisaurus agudoensis, based on the presence of small, isolated postcanine teeth referred to the probainognathians Riograndia guaibensis and Brasilitherium riograndensis (Langer et al. 2007; Langer and Ferigolo 2013). Indeed, their presence implies a correlation to the “Riograndia Assemblage Zone” of Soares et al. (2011; see also Bonaparte et al. 2010), which is younger than the “Hyperodapedon Assemblage Zone” sampled from the Janner site. A 206Pb/238U date of 225.42±0.37Ma (Norian) has been recently (Langer et al. 2018) recovered based on detrital zircon from Caturrita Formation deposits at a different site (Linha São Luiz) with a typical Riograndia Assemblage Zone fauna, so that a similar age could be tentatively inferred for the type-stratum of Sacisaurus agudoensis.
Sacisaurus agudoensis is the only silesaurid from the Santa Maria Supersequence. Although known from a significant amount of material, its uniqueness does not help to correlate its type locality to other Triassic sites in Rio Grande do Sul. As for the sauropodomorph remains described here, the anatomy of the recovered bones (especially the neck vertebra, ilium, and metatarsal) is more reminiscent of that seen in putatively younger (Norian) members of the group, such as Pantydraco caducus and Unaysaurus tolentinoi, than in Carnian sauropodomorphs such as Saturnalia tupiniquim and Panphagia protos.
To evaluate the phylogenetic relationships of the sauropodomorph remains, all materials were considered as a single Operational Taxonomic Unit, although this assignment is uncertain, based on their similar phylogenetic signal and topotypic principles (see Material). For the analysis, this OTU was scored (see SOM, Supplementary Online Material available at http://app.pan.pl/SOM/app63-Marsola_etal_SOM.pdf) in the Bronzati et al. (2018) dataset to assess its affinities with other Triassic sauropodomorphs. We used the software TNT 1.5 beta (Goloboff and Catalano 2016) for the analysis. The heuristic search was performed with: 10 000 replications of Wagner Trees (with random addition sequence), TBR (tree bi-section and reconnection) for branch swapping; hold = 20 (trees saved per replicate); and collapse of zero length branches according to “rule 1” in TNT (Coddington and Scharff 1994; Goloboff et al. 2008). Then, the resultant MPTs were subject to a second round of TBR to ensure all MPTs are found. We used the TNT command-line best to exclude suboptimal trees. Following Bronzati et al. (2018), we also set characters 86 and 365 as inactive.
The phylogenetic analysis resulted in 2.052 MPTs of 1250 steps. The strict consensus of which (Fig. 10) shows the sauropodomorph from the Sacisaurus site (labelled “MCN PV Sauropodomorph”) nested among Norian and Rhaetian taxa, more related to Thecodontosaurus and Efraasia than to Carnian representatives of the lineage. Some of the characters that support these relationships include: the inferred presence of one trunk-sacral vertebrae; the length of the post acetabular ala between 40–100% of that between the pubic and ischiatic peduncles; the brevis fossa well-developed and ventrally facing; and the proximomedially angled transverse axis of the distal end of the metatarsal I.
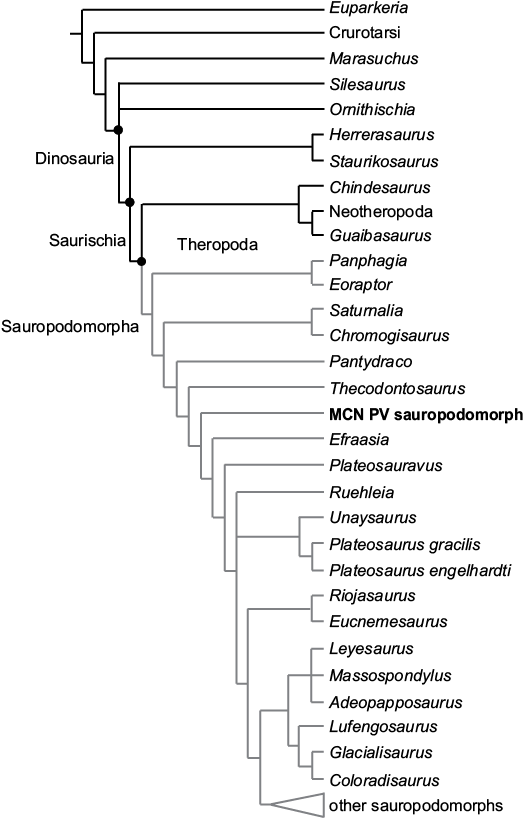
Fig. 10. Strict consensus tree of 2052 MPTs found in the analysis of the data matrix of Bronzati et al. (2018), showing the phylogenetic relationships of MCN PV sauropodomorph among Triassic sauropodomorphs and other dinosauromorphs.
The isolated Riograndia- and Exaeretodon-like cynodont teeth do not help correlating the Sacisaurus site to either the top of the Alemoa Member of the Santa Maria Formation or the base of the Caturrita Formation, as similar forms are found in both geological settings (Abdala et al. 2002; Ribeiro et al. 2011; Bittencourt et al. 2012; Martinelli et al. 2016). However, the record of “brasilodontid” teeth seem to match that of the sauropodomorphs described here, as these cynodonts are until now restricted to the younger (Norian) “Riograndia Assemblage Zone”. Yet, the resemblance of those same teeth to those of Alemoatherium huebneri, and their differences relative to those of Brasilitherium riograndensis and Brasilodon quadrangularis, precludes strong biostratigraphic inferences.
Taken together, geological and palaeontological data indicate that the strata identified in the Sacisaurus site, with its discovered fauna, including the Exaeretodon-like gomphodontosuchinae traversodontids found in the sandstones below the type-stratum of S. agudoensis, are likely younger than the fossil-bearing beds of the Janner site. In lithostratigraphical terms, the mudstones of the latter site are typical of the Alemoa Member, matching those from other sites of the Santa Maria Supersequence that typically yield “Hyperodapedon Assemblage Zone” faunas. In particular, the abundance of Exaeretodon riograndensis in the Janner site suggests that it belongs to younger faunas within the “Hyperodapedon Assemblage Zone” (Langer et al. 2007; Pretto et al. 2015). On the contrary, the rocks exposed at the Sacisaurus site belong to the Caturrita Formation, which regionally overlies the Alemoa Member, matching the signal provided by the sauropodomorph and “brasilodontid” fossils from the S. agudoensis type-stratum. Accordingly, the available data indicates a younger age for the Sacisaurus site relative of the entire “Hyperodapedon Assemblage Zone”, so that the Riograndia-like teeth from the site could actually belong to R. guaibensis, which characterizes the younger “Riograndia Assemblage Zone”. On the other hand, the Exaeretodon-like cynodonts found both at and below the S. agudoensis type-stratum indicates that traversodontids similar to that genus have extended their occurrence to strata younger than the “Hyperodapedon Assemblage Zone”, as previously suggested by the presence of a small-sized form in “Riograndia Assemblage Zone” deposits at the Poste site (Ribeiro et al. 2011), in the area of Candelaria (Bittencourt et al. 2012).
Conclusions
The specimens described here reveal the presence of a larger dinosauromorph in the type-stratum of Sacisaurus agudoensis, most probably corresponding to a sauropodomorph dinosaur. Its anatomy resembles more that of Norian representatives of the group, such as Pantydraco caducus and Unaysaurus tolentinoi than that of Carnian taxa such as Saturnalia tupiniquim and Pampadromaeus barberenai. Together with local stratigraphic correlations and the presence of brasilodontid teeth in the fossil assemblage, this indicates a higher stratigraphic position and younger age for the Sacisaurus site relative to the better sampled fauna of the neighbouring Janner site. The fossiliferous layers of the latter correspond to the typical mudstones of the Santa Maria Formation, which yield the Carnian “Hyperodapedon Assemblage Zone”. These are regionally overlapped by the sandstone of the Caturrita Formation, which yield the Norian “Riograndia Assemblage Zone” and correspond to the entire sequence exposed at the Sacisaurus site. Hence, a Norian age is, based on the available evidence, the best estimate for Sacisaurus agudoensis.
Acknowledgements
JCAM is grateful to the following collection managers who provided access to the specimens under their care: Alan Turner (Stony Brook University, USA), Alejandro Kramarz (Museo Argentino de Ciencias Naturales Bernardino Rivadavia, Buenos Aires, Argentina), Carl Mehling (American Museum of Natural History, New York, USA), Caroline Buttler (National Museum of Wales, Cardiff, UK), César Schultz (UFRGS), Claudia Hildebrandt (University of Bristol, UK), Deborah Hutchinson (Bristol Museum and Art Gallery, UK), Gabriela Cisterna (PURL), Ingmar Werneburg (Eberhard Karls Universität Tübingen, Germany), Jaime Powell (Fundación Miguel Lillo, Tucumán, Argentina), Jessica Cundiff (Museum of Comparative Zoology, Cambridge, USA), Marco Brandalise de Andrade (MCP), Oliver Rauhut (Ludwig-Maximilians-Universität, Munich, Germany), Rainer Schoch (SMNS), Ricardo Martínez (PVSJ), Sandra Chapman (NHM), Sérgio Cabreira (ULBRA), Sifelani Jirah (BPI), Thomaz Schossleitner (MB. R.), Tomasz Sulej (Institute of Paleobiology, Polish Academy of Sciences, Warsaw, Poland) and Mateusz Tałanda (University of Warsaw, Poland), and Zaituna Erasmus (SAM-PK). The authors also thank Adam Marsh and William Parker (both Division of Resource Management, Petrified Forest National Park, Petrified Forest, Arizona, USA) for sharing valuable photos for comparisons. The following grants supported this research: FAPESP 2013/23114-1 and 2016/02473-1 to JCAM; 2014/03825-3 to MCL; and FAPEMIG APQ-01110-15 to JSB. The authors thank the APP editor Stephen Brusatte, and the reviewers Christopher Griffin (Department of Geosciences, Virginia Tech, Blacksburg, VA, USA) and Brandon Peecook (Integrative Research Center, Field Museum of Natural History, Chicago, Illinois, USA) for their valuable comments which improved quality to the manuscript. TNT 1.5 is a free program made available by the Willi Hennig Society, which is thanked.
References
Abdala, F., Barberena, M.C., and Dornelles, J. 2002. A new species of the traversodontid cynodont Exaeretodon from the Santa Maria Formation (Middle/Late Triassic) of southern Brazil. Journal of Vertebrate Paleontology 22: 313–325. Crossref
Agnolín, F.F., and Rozadilla, S. 2017. Phylogenetic reassessment of Pisanosaurus mertii Casamiquela, 1967, a basal dinosauriform from the Late Triassic of Argentina. Journal of Systematic Palaeontology 16: 853–879. Crossref
Andreis, R.R., Bossi, G.E., and Montardo, D.K. 1980. O Grupo Rosário do Sul (Triássico) no Rio Grande do Sul. Congresso Brasileiro de Geologia 31: 659–673.
Apaldetti, C., Martínez, R.N., Cerda, I.A., Pol, D., and Alcober, O. 2018. An early trend towards gigantism in Triassic sauropodomorph dinosaurs. Nature Ecology & Evolution 2: 1–6. Crossref
Apaldetti, C., Pol, D., and Yates, A. 2013. The postcranial anatomy of Coloradisaurus brevis (Dinosauria: Sauropodomorpha) from the Late Triassic of Argentina and its phylogenetic implications. Palaeontology 56: 277–301. Crossref
Baron, M.G. 2017. Pisanosaurus mertii and the Triassic ornithischian crisis: could phylogeny offer a solution? Historical Biology: 1–15. Crossref
Baron, M.G., Norman, D.B., and Barrett, P.M. 2017a. A new hypothesis of dinosaur relationships and early dinosaur evolution. Nature 543: 501–506. Crossref
Baron, M.G., Norman, D.B., and Barrett, P.M. 2017b. Baron et al. reply. Nature 551: E4–E5. Crossref
Baron, M.G., Norman, D.B., and Barrett, P.M. 2017c. Postcranial anatomy of Lesothosaurus diagnosticus (Dinosauria: Ornithischia) from the Lower Jurassic of southern Africa: implications for basal ornithischian taxonomy and systematics. Zoological Journal of the Linnean Society 179: 125–168.
Barret, P.M. 2000. Prosauropod dinosaurs and iguanas: speculations on the diets of extinct reptiles. In: H.-D. Sues (ed.), Evolution of Herbivory in Terrestrial Vertebrates. Perspectives from the Fossil Record, 42–78. Cambridge University Press, London. Crossref
Benson, R.B., Butler, R.J., Carrano, M.T., and O’Connor, P.M. 2012. Air-filled postcranial bones in theropod dinosaurs: physiological implications and the “reptile”-bird transition. Biological Reviews 87: 168–193. Crossref
Benton, M.J., Forth, J., and Langer, M.C. 2014. Models for the rise of the dinosaurs. Current Biology 24: R87–R95. Crossref
Benton, M.J., Juul, L., Storrs, G.W., and Galton, P.M. 2000. Anatomy and systematics of the prosauropod dinosaur Thecodontosaurus antiquus from the Upper Triassic of southwest England. Journal of Vertebrate Paleontology 20: 77–108. Crossref
Bernardi, M., Gianolla, P., Petti, F.M., Mietto, P., and Benton, M.J. 2018. Dinosaur diversification linked with the Carnian Pluvial Episode. Nature Communications 9: 1–10. Crossref
Bittencourt, J.S., and Kellner, A.W.A. 2009. The anatomy and phylogenetic position of the Triassic dinosaur Staurikosaurus pricei Colbert, 1970. Zootaxa 2079: e56.
Bittencourt, J.S., Leal, L.A., Langer, M.C., and Azevedo, S.A. 2012. An additional basal sauropodomorph specimen from the Upper Triassic Caturrita Formation, southern Brazil, with comments on the biogeography of plateosaurids. Alcheringa 36: 269–278. Crossref
Bonaparte, J.F., and Barberena, M.C. 2001. On two advanced carnivorous cynodonts from the Late Triassic of Southern Brazil. Bulletin of the Museum of Comparative Zoology 156: 59–80.
Bonaparte, J.F., Ferigolo, J., and Ribeiro, A.M. 2001. A primitive Late Triassic “ictidosaur” from Rio Grande do Sul, Brazil. Palaeontology 44: 623–635. Crossref
Bonaparte, J.F., Martinelli, A.G., and Schultz, C.L. 2005. New information on Brasilodon and Brasilitherium (Cynodontia, Probainognathia) from the Late Triassic of southern Brazil. Revista Brasileira de Paleontologia 8: 25–46. Crossref
Bonaparte, J.F., Martinelli, A.G., Schultz, C.L., and Rubert. R. 2003. The sister group of mammals: small cynodonts from the Late Triassic of southern Brazil. Revista Brasileira de Paleontologia 5: 5–27.
Bonaparte, J.F., Schultz, C.L., Soares M.B., and Martinelli, A. 2010. La fauna local de Faxinal do Soturno, Triásico Tardío de Rio Grande do Sul, Brazil. Revista Brasileira de Paleontologia 13: 1–14. Crossref
Britt, B.B. 1993. Pneumatic Postcranial Bones in Dinosaurs and Other Archosaurs. 383 pp. Ph.D. Dissertation, University of Calgary, Calgary.
Bronzati, M., Benson, R.B., and Rauhut, O.W. 2018. Rapid transformation in the braincase of sauropod dinosaurs: integrated evolution of the braincase and neck in early sauropods? Palaeontology 61: 289–302. Crossref
Brusatte, S.L., Benton, M.J., Ruta, M., and Lloyd, G.T. 2008a. Superiority, competition, and opportunism in the evolutionary radiation of dinosaurs. Science 321: 1485–1488. Crossref
Brusatte, S.L., Benton, M.J., Ruta, M., and Lloyd, G.T. 2008b. The first 50 Myr of dinosaur evolution: macroevolutionary pattern and morphological disparity. Biology Letters 23: 733–736. Crossref
Butler, R.J. 2010. The anatomy of the basal ornithischian dinosaur Eocursor parvus from the lower Elliot Formation (Late Triassic) of South Africa. Zoological Journal of the Linnean Society 160: 648–684. Crossref
Cabreira, S.F., Kellner, A.W.A., Dias-da-Silva, S., da Silva, L.R., Bronzati, M., Marsola, J.C.A., Müller, R.T., Bittencourt, J.S., Batista, B.J., Raugust, T., Carrilho, R., Brodt, A., and Langer, M.C. 2016. A unique Late Triassic dinosauromorph assemblage reveals dinosaur ancestral anatomy and diet. Current Biology 26: 3090–3095. Crossref
Coddington, J. and Scharff, N. 1994. Problems with zero-length branches. Cladistics 10: 415–423. Crossref
Cooper, M.R. 1981. The prosauropod Massospondylus carinatus Owen from Zimbabwe: its biology, mode of life and phylogenetic significance. Occasional Papers of the National Museums and Monuments 6: 689–840.
Crompton, A.W. 1974. The dentitions and relationships of the southern African Triassic mammals, Erythrotherium parringtoni and Megazostrodon rudnerae. Bulletin of the British Museum (Natural History), Geology 24: 397–437.
Currie, P.J., and Zhao, X.J. 1993. A new carnosaur (Dinosauria, Theropoda) from the Jurassic of Xinjiang, People’s Republic of China. Canadian Journal of Earth Sciences 30: 2037–2081. Crossref
Da Rosa, Á.A.S. 2005. Paleoalterações em depósitos sedimentares de planícies aluviais do Triássico Médio a Superior do sul do Brasil: caracterização, análise estratigráfica e preservação fossilífera. 211 pp. Ph.D. Dissertation, Universidade do Vale dos Sinos, São Leopoldo.
Da Rosa, Á. A. 2015. Geological context of the dinosauriform-bearing outcrops from the Triassic of Southern Brazil. Journal of South American Earth Sciences 61: 108–119. Crossref
Dzik, J. 2003. A beaked herbivorous archosaur with dinosaur affinities from the early Late Triassic of Poland. Journal of Vertebrate Paleontology 23: 556–574. Crossref
Ezcurra, M.D. 2010. A new early dinosaur (Saurischia: Sauropodomorpha) from the Late Triassic of Argentina: a reassessment of dinosaur origin and phylogeny. Journal of Systematic Palaeontology 8: 371–425. Crossref
Ezcurra, M.D. 2016. The phylogenetic relationships of basal archosauromorphs, with an emphasis on the systematics of proterosuchian archosauriforms. PeerJ 4: e1778. Crossref
Ferigolo, J. and Langer, M.C. 2007. A Late Triassic dinosauriform from south Brazil and the origin of the ornithischian predentary bone. Historical Biology 19: 23–33. Crossref
Flynn, J.J., Parrish, M., Rakotosamimanana, B., Simpson, W.F., Whatley, R.L., and Wyss, A.R. 1999. A Triassic fauna from Madagascar, including early dinosaurs. Science 286: 763–765. Crossref
Galton, P.M. and Kermack, D. 2010. The anatomy of Pantydraco caducus, a very basal sauropodomorph dinosaur from the Rhaetian (Upper Triassic) of South Wales, UK. Revue de Paléobiologie 29: 341–404.
Goloboff, P.A. and Catalano, S.A. 2016. TNT version 1.5, including a full implementation of phylogenetic morphometrics. Cladistics 32: 221–238. Crossref
Goloboff, P.A., Farris, J.S., and Nixon, K.C. 2008. TNT, a free program for phylogenetic analysis. Cladistics 24: 774–786. Crossref
Griffin, C.T. 2018. Developmental patterns and variation among early theropods. Journal of Anatomy 232: 604–640. Crossref
Griffin, C.T. and Nesbitt, S.J. 2016. The femoral ontogeny and long bone histology of the Middle Triassic (? late Anisian) dinosauriform Asilisaurus kongwe and implications for the growth of early dinosaurs. Journal of Vertebrate Paleontology 36: e1111224. Crossref
Hendrickx, C. and Mateus, O. 2014. Abelisauridae (Dinosauria: Theropoda) from the Late Jurassic of Portugal and dentition-based phylogeny as a contribution for the identification of isolated theropod teeth. Zootaxa 3759: 1–74. Crossref
Horn, B.L.D., Melo, T.M., Schultz, C.L., Philipp, R.P., Kloss, H.P., and Goldberg, K. 2014. A new third-order sequence stratigraphic framework applied to the Triassic of the Paraná Basin, Rio Grande do Sul, Brazil, based on structural, stratigraphic and paleontological data. Journal of South American Earth Sciences 55: 123–132. Crossref
Hutchinson, J.R. 2001a. The evolution of femoral osteology and soft tissues on the line to extant birds (Neornithes). Zoological Journal of Linnean Society 131: 169–197. Crossref
Hutchinson, J.R. 2001b. The evolution of pelvic osteology and soft tissues on the line to extant birds (Neornithes). Zoological Journal of the Linnean Society 131: 123–168. Crossref
Irmis, R.B. 2007. Axial skeleton ontogeny in the Parasuchia (Archosauria: Pseudosuchia) and its implications for the ontogenetic determination in archosaurs. Journal of Vertebrate Paleontology 27:350–361. Crossref
Irmis, R.B., Parker, W.G., Nesbitt, S.J., and Liu, J. 2007. Early ornithischian dinosaurs: the Triassic record. Historical Biology 19: 3–22. Crossref
Langer, M.C. 2003. The pelvic and hind limb anatomy of the stem-sauropodomorph Saturnalia tupiniquim (Late Triassic, Brazil). PaleoBios 23: 1–40.
Langer, M.C. 2004. Basal saurischia. The Dinosauria 2: 25–46. Crossref
Langer, M.C. and Benton, M.J. 2006. Early dinosaurs: a phylogenetic study. Journal of Systematic Palaeontology 4: 309–358. Crossref
Langer, M.C. and Ferigolo, J. 2013. The Late Triassic dinosauromorph Sacisaurus agudoensis (Caturrita Formation; Rio Grande do Sul, Brazil): anatomy and affinities. Geological Society, London, Special Publications 379: 353–392. Crossref
Langer, M.C., Bittencourt, J.S., and Schultz, C.L. 2010. A reassessment of the basal dinosaur Guaibasaurus candelariensis, from the Late Triassic Caturrita Formation of south Brazil. Earth and Environmental Science Transactions of the Royal Society of Edinburgh 101: 301–332. Crossref
Langer, M.C., Ezcurra, M.D., Rauhut, O.W.M., Benton, M.J., Knoll, F., McPhee, B.W., Novas, F.E., Pol, D., and Brusatte, S. 2017. Untangling the dinosaur family tree. Nature 551: E1–E5. Crossref
Langer, M.C., Nesbitt, S.J., Bittencourt, J.S., and Irmis, R.B. 2013. Non-dinosaurian Dinosauromorpha. Geological Society, London, Special Publications 379: 157–186. Crossref
Langer, M.C., Ramezani, J., and Da Rosa, Á.A.S. 2018. U-Pb age constraints on dinosaur rise from south Brazil. Gondwana Research 57: 133–140. Crossref
Langer, M.C., Ribeiro, A.M., Schultz, C.L., and Ferigolo, J. 2007. The continental tetrapod-bearing Triassic of south Brazil. New Mexico Museum of Natural History and Science Bulletin 41: 201–218.
Marsicano, C.A., Irmis, R.B., Mancuso, A.C., Mundil, R., and Chemale, F. 2016. The precise temporal calibration of dinosaur origins. Proceedings of the National Academy of Sciences 113: 509–513. Crossref
Marsola, J.C.A., Bittencourt, J.S., Butler, R.J., Da Rosa, Á.A.S., Sayão, J.M., and Langer, M.C. (in press). A new dinosaur with theropod affinities from the Late Triassic Santa Maria Formation, South Brazil. Journal of Vertebrate Paleontology.
Martínez, R.N. 2009. Adeopapposaurus mognai, gen. et sp. nov. (Dinosauria: Sauropodomorpha), with comments on adaptations of basal Sauropodomorpha. Journal of Vertebrate Paleontology 29: 142–164. Crossref
Martínez, R.N. and Alcober, O.A. 2009. A basal sauropodomorph (Dinosauria: Saurischia) from the Ischigualasto Formation (Triassic, Carnian) and the early evolution of Sauropodomorpha. PLoS One 4: e4397. Crossref
Martínez, R.N., Sereno, P.C., Alcober, O.A., Colombi, C.E., Renne, P.R., Montañez, I.P., and Currie, B.S. 2011. A basal dinosaur from the dawn of the dinosaur era in southwestern Pangaea. Science 331: 206–210. Crossref
Martinelli, A.G., Eltink, E., Da-Rosa, Á.A.S., and Langer, M.C. 2017a. A new cynodont (Therapsida) from the Hyperodapedon Assemblage Zone (upper Carnian–Norian) of southern Brazil improves the Late Triassic probainognathian diversity. Papers in Palaeontology 3: 401–423. Crossref
Martinelli, A.G., Soares, M.B., Oliveira, T.V., Rodrigues, P.G., and Schultz. C.L. 2017b. The Triassic eucynodont Candelariodon barberenai revisited and the early diversity of stem prozostrodontians. Acta Palaeontologica Polonica 62: 527–542. Crossref
Martinelli, A.G., Soares, M.B., Rodrigues, P., and Schultz, C.L. 2016. The oldest ictidosaur cynodont (Therapsida) from the late Carnian of southern Brazil and its implication in probainognathian evolution. In: O.N. Grillo, P.S.R. Romano, and G.R. Oliveira (eds.), X Simpósio Brasileiro de Paleontologia de Vertebrados, Rio de Janeiro. Boletim de Resumos, 111. Sociedade Brasileira de Paleontologia, Rio de Janeiro.
Müller, R.T., Langer, M.C., Bronzati, M., Pacheco, C.P., Cabreira, S.F., and Dias-Da-Silva, S. 2018. Early evolution of sauropodomorphs: anatomy and phylogenetic relationships of a remarkably well-preserved dinosaur from the Upper Triassic of southern Brazil. Zoological Journal of the Linnean Society: 1–62. Crossref
Nesbitt, S.J. 2005. Osteology of the Middle Triassic pseudosuchian archosaur Arizonasaurus babbitti. Historical Biology 17: 19–47. Crossref
Nesbitt, S.J. 2007. The anatomy of Effigia okeeffeae (Archosauria, Suchia), theropod-like convergence, and the distribution of related taxa. Bulletin of the American Museum of Natural History 302: 1–84. Crossref
Nesbitt, S J. 2011. The early evolution of archosaurs: relationships and the origin of major clades. Bulletin of the American Museum of Natural History 352: 1–292. Crossref
Nesbitt, S.J., Sidor, C.A., Irmis, R.B., Angielczyk, K.D., Smith, R.M.H., and Tsuji, L.A. 2010. Ecologically distinct dinosaurian sister-group shows early diversification of Ornithodira. Nature 464: 95–98. Crossref
Novas, F.E. 1994. New information on the systematics and postcranial skeleton of Herrerasaurus ischigualastensis (Theropoda: Herrerasauridae) from the Ischigualasto Formation (Upper Triassic) of Argentina. Journal of Vertebrate Paleontology 13: 400–423. Crossref
Pacheco, C.P., Martinelli, A.G., Pavanatto, A.E.B., Soares, M.B, and Dias-da-Silva, S. 2018. Prozostrodon brasiliensis, a probainognathian cynodont from the Late Triassic of Brazil: second record and improvements on its dental anatomy. Historical Biology 30: 475–485. Crossref
O’Connor, P.M. 2006. Postcranial pneumaticity: An evaluation of soft-tissue influences on the postcranial skeleton and the reconstruction of pulmonary anatomy in archosaurs. Journal of Morphology 267: 1199–1226. Crossref
O’Connor, P.M. 2007. The postcranial axial skeleton of Majungasaurus crenatissimus (Theropoda: Abelisauridae) from the Late Cretaceous of Madagascar. Journal of Vertebrate Paleontology 27: 127–162. Crossref
Parker, W.G., R.B. Irmis, S.J. Nesbitt, J.W. Martz, and L.S. Browne. 2005. The Late Triassic pseudosuchian Revueltosaurus callenderi and its implications for the diversity of early ornithischian dinosaurs. Proceedings of the Royal Society of London B 272: 963–969. Crossref
Pol, D., Garrido, A., and Cerda, I.A. 2011. A new sauropodomorph dinosaur from the Early Jurassic of Patagonia and the origin and evolution of the sauropod-type sacrum. PLoS ONE 6: e14572. Crossref
Porro, L.B., Witmer, L.M., and Barrett, P.M. 2015. Digital preparation and osteology of the skull of Lesothosaurus diagnosticus (Ornithischia: Dinosauria). PeerJ 3: e1494. Crossref
Pretto, F.A., Schultz, C.L., and Langer, M.C. 2015. New dinosaur remains from the Late Triassic of southern Brazil (Candelária Sequence, Hyperodapedon Assemblage Zone). Alcheringa 39: 264–273.
Pretto, F.A., Langer, M.C., and Schultz, C.L. 2018. A new dinosaur (Saurischia: Sauropodomorpha) from the Late Triassic of Brazil provides insights on the evolution of sauropodomorph body plan. Zoological Journal of the Linnean Society: 1–29. Crossref
Prieto-Márquez, A. and Norell, M.A. 2011. Redescription of a nearly complete skull of Plateosaurus (Dinosauria: Sauropodomorpha) from the Late Triassic of Trossingen (Germany). American Museum Novitates 3727: 58. Crossref
Ribeiro, A.M., Abdala, F., and Bertoni, R.S. 2011. Traversodontid cynodonts (Therapsida–Eucynodontia) from two Upper Triassic localities of the Paraná Basin, southern Brazil. Ameghiniana 48 (Supplement): R111.
Raath, M.A. 1977. The Anatomy of the Triassic theropod Syntarsus rhodesiensis (Saurischia: Podokesauridae) and Considerations of Its Biology. 233 pp. Ph.D. Dissertation, Rhodes University, Salisbury.
Rauhut, O.W.M. 2003. The interrelationships and evolution of basal theropod dinosaurs. Special Papers in Palaeontology 69: 1–215.
Rauhut, O.W.M. and Carrano, M.T. 2016. The theropod dinosaur Elaphrosaurus bambergi, from the Late Jurassic of Tendaguru, Tanzania. Zoological Journal of the Linnean Society 178: 546–610. Crossref
Rinehart, L.F., Lucas, S.G., Heckert, A.B., Spielmann, J.A., and Celeskey, M.D. 2009. The Paleobiology of Coelophysis bauri (Cope) from the Upper Triassic (Apachean) Whitaker quarry, New Mexico, with detailed analysis of a single quarry block. New Mexico Museum of Natural History and Science Bulletin 45: 1–260.
Rowe, T.B., Sues, H.D., and Reisz, R.R. 2011. Dispersal and diversity in the earliest North American sauropodomorph dinosaurs, with a description of a new taxon. Proceedings of the Royal Society of London B 278: 1044–1053. Crossref
Sereno, P.C. and Novas, F.E. 1993. The skull and neck of the basal theropod Herrerasaurus ischigualastensis. Journal of Vertebrate Paleontology 13: 451–476. Crossref
Sereno, P.C., Forster, C.A., Rogers, R.R., and Monetta, A.M. 1993. Primitive dinosaur skeleton from Argentina and the early evolution of Dinosauria. Nature 361: 64–66. Crossref
Sereno, P.C., Martínez, R.N., and Alcober, O.A. 2012. Osteology of Eoraptor lunensis (Dinosauria, Sauropodomorpha). Journal of Vertebrate Paleontology 32: 83–179. Crossref
Soares, M.B., Martinelli, A.G., and Oliveira, T.V. 2014. A new prozostrodontian cynodont (Therapsida) from the Late Triassic Riograndia Assemblage Zone (Santa Maria Supersequence) of Southern Brazil. Anais da Academia Brasileira de Ciências 86: 1673–1691. Crossref
Soares, M.B., Schultz, C.L., and Horn, B.L.D. 2011. New information on Riograndia guaibensis Bonaparte, Ferigolo and Ribeiro, 2001 (Eucynodontia, Tritheledontidae) from the Late Triassic of southern Brazil: anatomical and biostratigraphic implications. Anais da Academia Brasileira de Ciências 83: 329–354. Crossref
Sookias, R.B., Butler, R.J., and Benson, R.B.J. 2012. Rise of dinosaurs reveals major body-size transitions are driven by passive processes of trait evolution. Proceedings of the Royal Society of London B 279: 2180–2187. Crossref
Tykoski, R.S. 2005. Anatomy, Ontogeny, and Phylogeny of Coelophysoid Theropods. 553 pp. Ph.D. Dissertation, The University of Texas at Austin, Austin.
Wedel, M.J. 2007. What pneumaticity tells us about “prosauropods”, and vice versa. Special Papers in Palaeontology 77: 207–222.
Welles, S.P. 1984. Dilophosaurus wetherilli (Dinosauria, Theropoda). Osteology and comparisons. Palaeontographica Abteilung A 4–6: 85–180.
Whiteside, J.H., Lindström, S., Irmis, R.B., Glasspool, I.J., Schaller, M.F., Dunlavey, M., Nesbitt, S.J., Smith, N.D., and Turner, A.H. 2015. Extreme ecosystem instability suppressed tropical dinosaur dominance for 30 million years. Proceedings of the National Academy of Sciences 112: 7909–7913. Crossref
Wilson, J.A. 1999. A nomenclature for vertebral laminae in sauropods and other saurischian dinosaurs. Journal of Vertebrate Paleontology 19: 639–653. Crossref
Wilson, J.A. 2012. New vertebral laminae and patterns of serial variation in vertebral laminae of sauropod dinosaurs. Contributions from the Museum of Paleontology, University of Michigan 32: 91–110.
Wilson, J.A., Michael, D.D., Ikejiri, T., Moacdieh, E.M., and Whitlock, J.A. 2011. A nomenclature for vertebral fossae in sauropods and other saurischian dinosaurs. PLoS One 6: e17114.
Yates, A.M. 2003. A new species of the primitive dinosaur Thecodontosaurus (Saurischia: Sauropodomorpha) and its implications for the systematics of early dinosaurs. Journal of Systematic Palaeontology 1: 1–42. Crossref
Acta Palaeontol. Pol. 63 (4): 653–669, 2018
https://doi.org/10.4202/app.00492.2018