

A new polyphysacean alga from the Miocene of Romania and its biomineralization
FILIPPO BARATTOLO, VIOREL IONESI, and PAUL ŢIBULEAC
Barattolo, F., Ionesi, V., and Ţibuleac, P. 2019. A new polyphysacean alga from the Miocene of Romania and its biomineralization. Acta Palaeontologica Polonica 64 (1): 85–100.
Polyphysacean algal fertile caps from the Sarmatian near Suceava (NE Romania) are here described as a new species, Acetabularia moldavica, ascribed to the extant genus Acetabularia due to the presence of both inferior and superior coronae. Gametangia are preserved and closely packed within the gametophore. The gametangium wall is strongly mineralized and SEM observations show acicular aragonite crystals arranged perpendicularly to the wall surface. The occurrence of mineralized cyst walls distinguishes the new species from the other three species ascribed to the genus Acetabularia, namely Acetabularia miocaenica, Acetabularia chiavonica, and Acetabularia transylvana. Cyst wall biomineralization in A. moldavica sp. nov., Chalmasia, and Halicoryne appears to assist in distinguishing taxa at species level but not at genus level. Four types of intracellular biomineralization can be recognized in polyphysaceans according to the mineralization of cyst walls and intergametangial spaces. The attribution of an extant species to a fossil genus and vice-versa can be problematic when the fossil consists of a mineralized spicule (intracellular biomineralization). Two contrasting examples are Acetabularia schenckii (extant species) assigned to the genus Acicularia (fossil), and Halicoryne morelleti (fossil species) referred to an extant genus (Halicoryne). When it is unclear whether fossil taxa possessed one or two coronae, the attribution of Cenozoic species to an extant genus appears speculative. Therefore, we support the view that the genus Acicularia should only be applied to fossil species. In addition, mineralized aggregates of cysts of Halicoryne morelleti should be distinguished from their possible extant counterparts (Chalmasia, Halicoryne, and Acetabularia) when coronal structures are not preserved. In the literature Halicoryne morelleti has been compared with the fossil genera Sedalanella and Ioanella. The first genus must be considered a junior synonym of Oroseina. A new interpretation of the genus Ioanella is supplied. The two layers of cysts of Ioanella conform poorly with the small ellipsoidal calcareous bodies of Halicoryne morelleti. Therefore, we propose the new genus Patruliuspora for Halicoryne morelleti.
Key words: Dasycladales, Polyphysaceae, biomineralization, Miocene, Sarmatian, Romania.
Filippo Barattolo [filippo.barattolo@unina.it], Dipartimento di Scienze della Terra dell’Ambiente e delle Risorse, Università degli Studi di Napoli Federico II, Complesso universitario di Monte Sant’Angelo, via Cintia 21-80126 Napoli, Italy.
Viorel Ionesi [vioion@uaic.ro] and Paul Ţibuleac [paul.tibuleac@uaic.ro], Department of Geology, University Alexandru Ioan Cuza, Bulevardul Carol I, 20 A, 700505 Iaşi, Romania.
Received 25 August 2018, accepted 8 November 2018, available online 18 January 2019.
Copyright © 2019 F. Barattolo et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
During the Sarmatian, a sedimentary succession containing fossils of predominantly euryhaline fauna developed from Central Europe towards Eastern Asia (Fig. 1). Starting from the earliest Oligocene (Studencka and Jasionowski 2011), uplift of the the Alpine Orogenic Belt led to the formation of a distinctive paleoecosystem that evolved during the Neogene. This event modified circum-Mediterranean paleogeography and split the previous (Mesozoic) Neo-Tethys Ocean into the Mediterranean and Paratethys seas (Rögl 1999; Popov et al. 2003). In the Paratethys this segregation led to the evolution of endemic organisms and, consequently, to the need for the establishment of a regional stratigraphic scale. This endemism affected not only the terrestrial flora and fauna but also marine and brackish organisms.
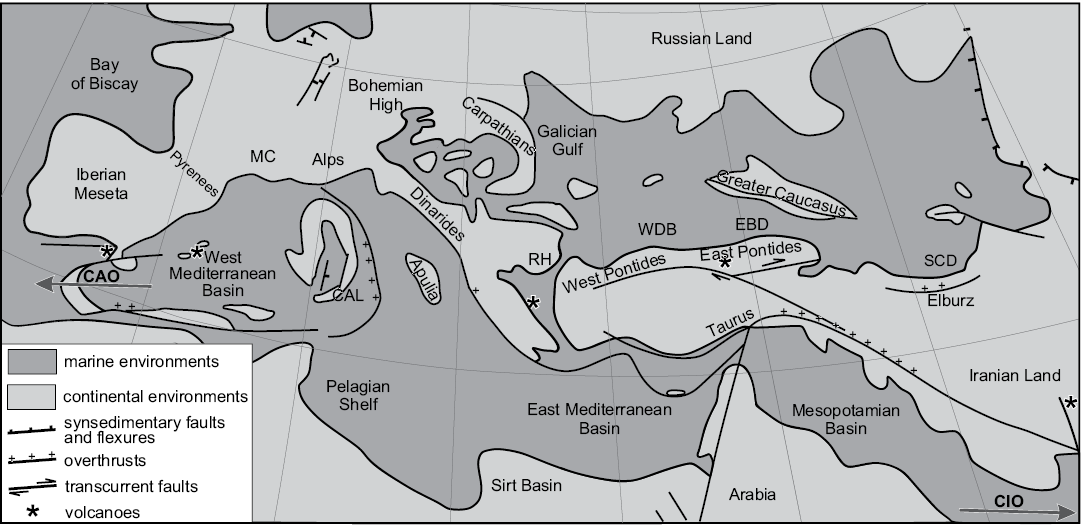
Fig. 1. Paleogeography of the Mediterranean area (Tethys and Paratethys Realms) during the late middle Miocene (12–11 Ma), simplified from Popov et al. (2003). CAO, connection with Atlantic Ocean; CAL, Calabria; CIO, connection with Indian Ocean; EBD, Eastern Black Sea Depression; MC, Massif Central; RH, Rhodope High; SCD, South Caspian Depression; WBD, Western Black Sea Depression.
Dasycladalean algae are typical fossils occurring in Paratethys during the Sarmatian, when brackish water depositional systems were widespread in this aquifer. Many Miocene (mainly Sarmatian) species belonging to extant genera have been recorded in the Paratethyan region ranging from Poland to Iran. A rich literature reports Miocene dasycladaceans (Neomeris and Cymopolia) and polyphysaceans (Acicularia and Halicoryne) from the Paratethys (Reuss 1861; Solms-Laubach 1895; Morellet and Morellet 1927, 1939; Bányai 1929; Bányai and Morellet 1936; Pokorný 1948; Maslov 1955, 1960; Boda 1959; Miletic-Spajic 1961; Małecki 1970, 1974; Stancu and Tautu 1973, 1974; Bucur et al. 1993; Paruch-Kulczycka 1994; Génot et al. 2002, 2008; Barattolo 2006).
Stancu and Tautu (1974) provided first account on Acicularia from the Eastern Carpathians Foreland, recording several species of these algae from Bessarabian strata in the Borehole 3702 Frumușița (Galați County). Later, Ţibuleac (1999, 2009) documented gametophores of Acicularia sp. from the Volhynian sands (Fălticeni-Boroaia Formation) cropping out on the Cetățuii Hill (Rădășeni village, Suceava County). Génot et al. (2002) supplied preliminary data on acetabulariaceans (i.e., polyphysaceans, tentatively assigned to Acicularia or Chalmasia) from lower Volhynian limestone near Iaz village (Suceava County).
Several samples containing polyphysacean algal fertile caps displaying unusually good preservation and the distinctive characters of the biomineralization in the Sarmatian deposits near Suceava (NE Romania) were subject of this collaborative research program.
Institutional abbreviations.—BM (NH), British Museum (Natural History), Department of Palaeontology, London, UK; LPB, Laboratoire de Paléontologie de Brest, Département des Sciences de la Terre (Géologie), Université de Bretagne Occidentale, Brest, France; MP-UAIC, Museum of Palaeontology, Department of Geology, University Alexandru Ioan Cuza, Iaşi, Romania.
Geological setting
Dasycladales were sampled from near-horizontal lower Sarmatian deposits close to Pătrăuţi village, Suceava District (Fig. 2). This area belongs to the Moldavian Platform (Eastern Carpathian Foreland), which represents the south-west extension of the large East-European Platform (Ionesi 1994).
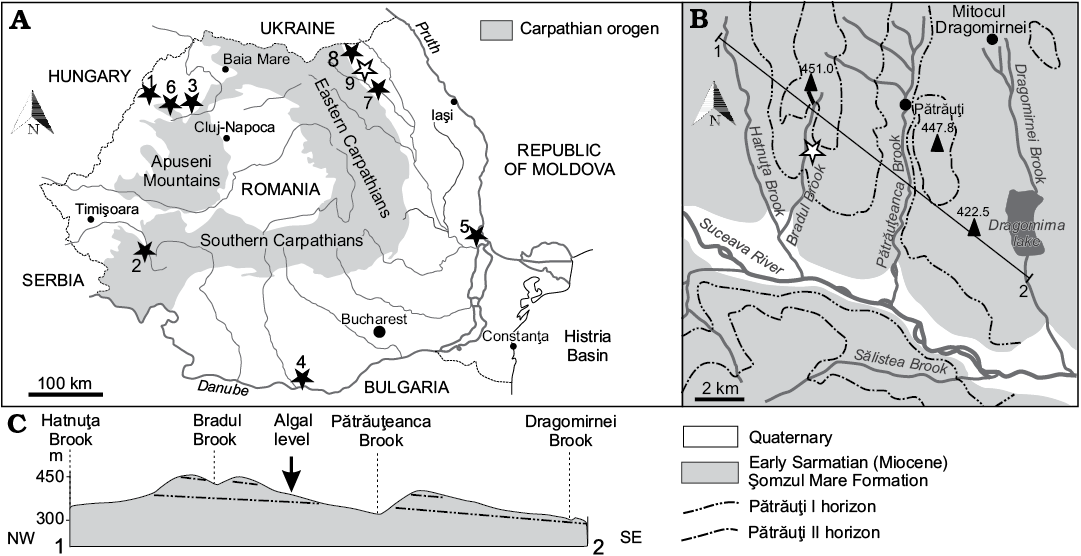
Fig. 2. A. Location of the Sarmatian dasycladalean outcrops in Romania (black stars, white star indicates Acetabularia moldavica type locality). 1, Satu Nou (Bihor County); 2, Soceni (Caraş-Severin County); 3, Copăcel and Chijic (Bihor County); 4, Izlaz (Olt County); 5, Frumuşiţa (Galaţi County); 6, Luncşoara (Bihor County); 7, Rădăşeni (Suceava County); 8, Iaz (Suceava County); 9, Pătrăuţi (Suceava County). B. Geological map of the environs of Pătrăuţi with the site of Acetabularia moldavica sp. nov. C. Profile of the Pătrăuţi outcrop.
Geographically, this is the Dragomirna subunit of Suceava Plateau of the north-western part of the extensive Moldavian Plateau (Băcăuanu et al. 1980). This zone displays the typical depositional pattern that developed along the newly forming Carpathian Orogen Land.
Near the Pătrăuţi village, Ionesi (1968) mapped a 100 m thick succession of clay, sand, and sandstone alternations. Two marker levels, an oolitic limestone and a sandstone, stratigraphically separated by some 80 m of strata, were identified as lithostratigraphic markers, namely Pătrăuţi I and Pătrăuţi II, respectively.
The samples with polyphysaceans were collected from a clay horizon between these two levels that crops out along the slope on the eastern side of the Bradul Stream, a tributary of the Suceava River, approximately 1 km north of European Road E 85. More precisely, the algae were collected from the thin clay level (0.75 m) that crops out for a hundred meters. The algal level overlies a sandstone bed and is covered by up to 4 m of thick sand layer that passes upwards into sandstone with sand intercalations for 1 m (Fig. 3).
The clay deposition can be considered a marine event, unusual because adjacent outcrops along the Bradul and Pătrăuțeanca streams surrounding Pătrăuţi village display a relative abundant brackish fauna of bivalves, gastropods, foraminifers, and ostracods. Ervilia podolica (Eichwald, 1853), E. trigonula Sokolov, 1899, Obsoletiforma lithopodolica (Dubois, 1831), Mactra eichwaldi Laskarev, 1914, Granulalobium bicinctum (Brocchi, 1814) etc. were cited, suggesting the Volhynian, a substage of Sarmatian (Ionesi 1968, 1995, 2006; Ţibuleac 2009). Consequently, the deposit can be ascribed to the Arghira Member of the Șomuz Formation (Ionesi 2006) by correlation with successions in its type-area to the south-east.
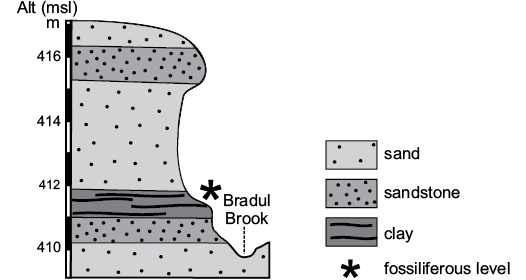
Fig. 3. Section and stratigraphy of the outcrop with the algal level (asterisk).
Material and methods
Several thin slabs of marly clay, 5–8 mm thick, labelled MP-UAIC 8228/PD (Pătrăuti Dasycladales), containing mineralized discs, with fragments and small groups of gametangia on their surfaces were collected. Five slabs were used for this study: MP-UAIC 8228/PD.1 9×6.5 cm, MP-UAIC 8228/PD.2 12×9 cm, MP-UAIC 8228/PD.3 3.5×2 cm, MP-UAIC 8228/PD.4 4×1.5 cm, MP-UAIC 8228/PD.5 1.5×1.5 cm. Material has been observed and photographed under an optical stereo-microscope. Some small pieces were detached from slab MP-UAIC 8228/PD.1 for SEM observations.
Systematic palaeontology
Order Dasycladales Pascher, 1931
Family Polyphysaceae (Kützing, 1843)
Genus Acetabularia Lamouroux, 1812
Type species: Madrepora acetabulum Linnaeus, 1758; Recent, Mediterranean Sea.
Remarks.—The alga consists of a cylindrical stalk terminating, at maturity, with a cap made by a whorl of fertile rays. At the inner end, each fertile ray bears two small convex cushions, one above (corona superior) and one below (corona inferior), and communicates with the stalk by means of a small vestibule.
The presence of both superior and inferior coronae or of the corona superior only (i.e., absence of corona inferior) is a character of generic importance. Among extant genera, Acetabularia possesses both superior and inferior coronae, while Parvocaulis, Chalmasia, and Halicoryne lack the corona inferior (Berger et al. 2003; Berger 2006).
From a structural point of view, coronae have been variously interpreted as (i) expansions of the central stem, (ii) primary laterals together with the basal part, or (iii) as secondary laterals (Dumais and Harrison 2000).
According to Berger et al. (2003), morphological and developmental characters of the cap, and molecular data (18S rDNA), allow Acicularia schenckii to be placed in the Acetabularia that also includes Polyphysa peniculus, a species lacking a corona inferior, the absence of which is supposed to be a derived feature (Berger et al. 2003). These authors concluded that the genus Acetabularia can be split into two subgenera, Acetabularia, containing only A. acetabulum, and Acicularia including all other species. The former subgenus develops cap rings with congenitally fused cap primordia, the latter subgenus has unfused cap primordia.
As discussed below, Acicularia (with type species Acicularia pavantina) should be restricted to fossil aggregates of cysts only. Therefore, extant species of Acetabularia with unfused cap primordia are better referred to the sectio Acetabuloides Solms-Laubach (1895), with type species Acetabularia kilneri, rather than to the fossil genus Acicularia as proposed by Berger et al. 2003, because extant Acetabularia schenckii is not the type species of the genus Acicularia.
Genera included: Several Cenozoic polyphysacean remains showing evidence of coronae (see review in Deloffre and Granier 1992): (i) Andrussow (1887) established A. miocenica from the Miocene of the Kerch Peninsula and near Sevastopol (Crimea). Later Solms-Laubach (1895) moved the taxon to Acicularia and renamed it Acicularia andrussowii Solms-Laubach, 1895 because of the homonymy with Acicularia miocaenica Reuss, 1861. Solms-Laubach (1895: 11) observed Andrussow’s (1887) material and confirmed the occurrence of double coronae. Here the original attribution to Acetabularia is followed: the name A. miocenica Andrussow, 1887 is maintained and a lectotype is designated (Andrussow 1887: text-fig. 1); (ii) Squinabol (1902) described A. chiavonica from Oligocene beds cropping out along the river Chiavon. This is a classical middle Oligocene fossiliferous horizon (with fishes, crustaceans, and palms) near Vicenza (Northern Italy; Giusberti et al. 2014). The thin calcified wall shows evidence of both coronae, and fertile rays display no evidence of intracellular mineralization. The specimen in Squinabol (1902: text-fig. 2) has been selected as lectotype by Deloffre and Granier (1992); (iii) Acicularia transylvana Bányai and Morellet, 1936 from the Sarmatian near Satul Nou (Romania) is preserved with a calcareous sheath and mineralized fertile rays, and structures related to coronae are also present (Génot 1987). The presence of intracellular mineralization allowed Bányai and Morellet (1936) to attribute the taxon to Acicularia. The species resembles Acetabularia miocenica Andrussow, 1887, but according to Bányai and Morellet (1936) mineralization between the fertile rays is much thinner, the distal ends of the fertile rays are more or less rounded, and gametangia are not arranged in a single row on the upper and the lower side of gametophores. The taxon is here moved to the genus Acetabularia and maintained the name A. transylvana (Bányai and Morellet, 1936). The specimen corresponding to the drawings by Bányai and Morellet (1936: fig. 2) is selected as lectotype (Deloffre and Granier 1992); (iv) a fourth new species is described here.
Acetabularia moldavica sp. nov.
Figs. 4–8.
Etymology: From the Moldova Region.
Type material: Holotype: fertile cap (Fig. 4F), MP-UAIC 8228/PD.1. Paratypes: specimens contained on the surfaces of five thin marl layers, MP-UAIC 8228/PD.1–5.
Type locality: The outcrop is located 2.5 km WSW from Pătrăuţi village (Suceava County) along the Bradul stream incision, at 411 m asl, coordinates 47°43’12” N, 26°09’53” E (Fig. 2B).
Type horizon: A clay seam, about 40 cm thick, within a sequence of sand and sandstone beds, twenty metres above the horizon Pătrăuţi I (Fig. 2); Sarmatian (middle Miocene).
Diagnosis.—Discs made of 10–24 mucronate fertile rays with evidence of both corona superior and inferior. Each ray, quite flat in transverse section, contains 14–35 mineralized gametangia (cysts). The mineralized cyst layer, 20–25 μm thick, is made of fibrous aragonite crystals set perpendicular to the wall surface. A delicate extracellular calcareous layer sometimes envelopes the fertile rays (Table 1).
Table 1. Main biometrical parameters (in mm) of Acetabularia moldavica sp. nov.; gametangium (cyst) diameter with mineralized layer included.
|
Specimen number |
Cap diameter |
Fertile ray |
Gametangium (cyst) |
||||
|
outer |
inner |
number in a cap |
distal diameter |
length |
diameter |
number in a ray |
|
|
PD.1/1 |
3.15 |
0.18 |
11 |
0.68 |
1.27 |
0.30 |
15 |
|
PD.1/2 |
6.83 |
|
20 |
1.00 |
3.17 |
0.36 |
30 |
|
PD.1/3 |
4.46 |
0.23 |
17 |
0.75 |
1.92 |
0.31 |
18 |
|
PD.1/6 |
3.83 |
0.29 |
15 |
0.72 |
1.84 |
0.30 |
16 |
|
PD.3/8 |
5.84 |
|
20 |
0.95 |
2.49 |
0.27 |
30 |
|
PD.1/10 |
5.30 |
|
15 |
0.95 |
2.42 |
0.27 |
35 |
|
PD.1/12 |
5.57 |
0.24 |
18 |
0.78 |
2.42 |
0.33 |
28 |
|
PD.2/14 |
5.92 |
|
24 |
0.87 |
2.83 |
0.26 |
35 |
|
PD.1/15 |
2.77 |
|
10 |
0.69 |
1.32 |
0.33 |
14 |
|
PD.1/16 |
4.10 |
0.31 |
15 |
0.67 |
1.65 |
0.30 |
20 |
|
PD.2/17 |
2.84 |
0.18 |
12 |
0.54 |
1.20 |
0.33 |
15 |
|
PD.2/19 |
6.14 |
0.38 |
20 |
0.63 |
2.33 |
0.38 |
28 |
|
PD.3/25 |
3.29 |
|
13 |
0.69 |
1.38 |
0.35 |
17 |
|
Min |
2.77 |
0.18 |
10 |
0.54 |
1.20 |
0.26 |
14 |
|
Max |
6.83 |
0.38 |
24 |
1.00 |
3.17 |
0.38 |
35 |
|
Average |
4.62 |
0.26 |
16.15 |
0.76 |
2.02 |
0.31 |
23.15 |
|
Standard deviation |
1.335278 |
0.067491 |
3.977749 |
0.134014 |
0.615778 |
0.034998 |
7.664565 |
|
Number of specimens |
13 |
7 |
13 |
13 |
13 |
13 |
13 |
Description.—General features of the calcareous skeleton: The alga is known only by its fertile caps (Figs. 4–7). No trace possibly related to the stem has been found on the rock surface. The fertile disks are slightly concave, the central upper part being sunken (Fig. 4A, D, G) while the lower part is protruding (Fig. 4E, F). Some thin extracellular mineralized layers envelope the basal part (primary lateral), superior and inferior coronae, and the space between the fertile rays (Fig. 5D, E). Occasionally a delicate extracellular sheet covers the upper and lower sides of the disk, and a thin radial rib marks the boundary between gametophores (Figs. 5B, 6B).
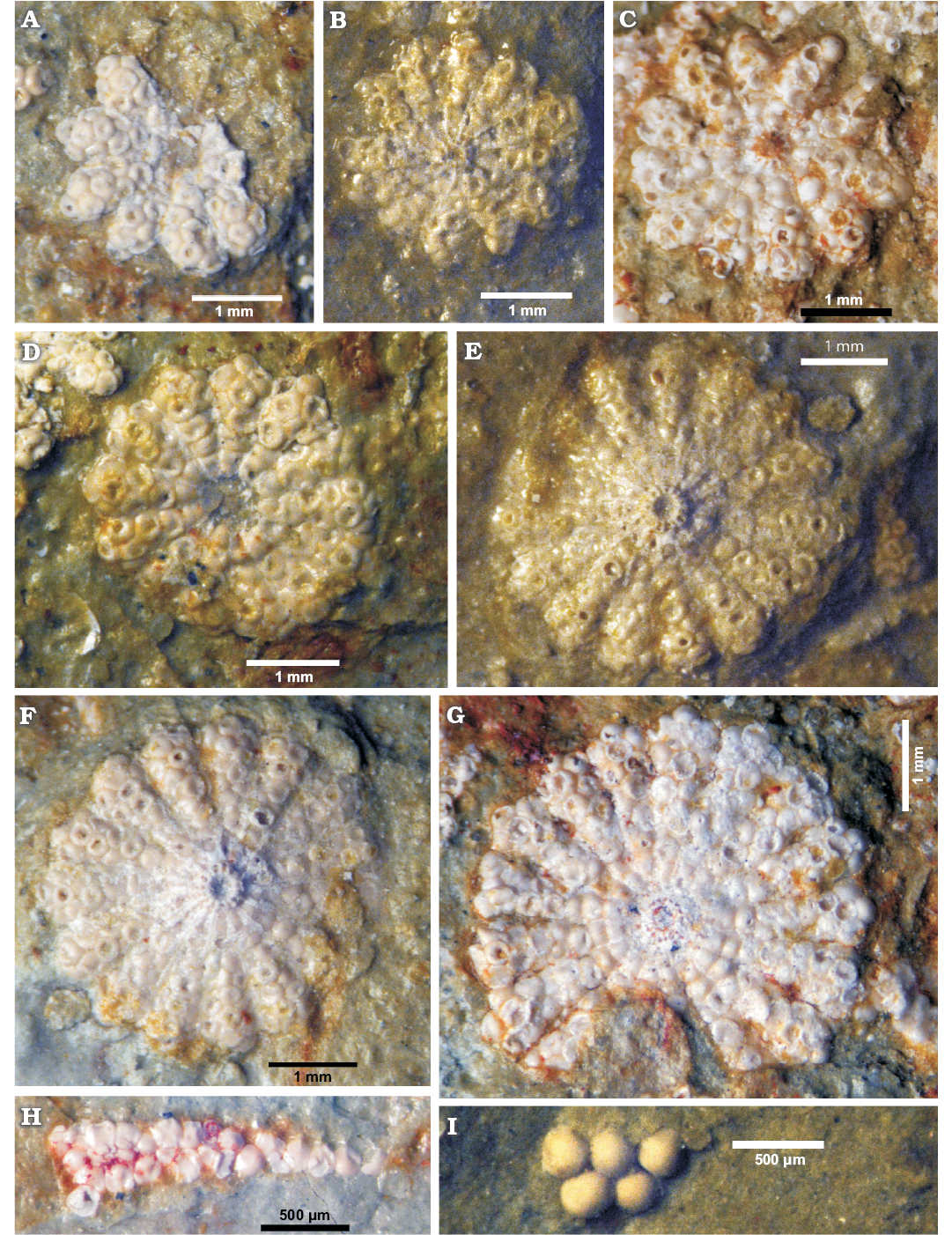
Fig. 4. Polyphysacean alga Acetabularia moldavica sp. nov. (A–H) and Patruliuspora sp. (I), early Sarmatian, Pătrăuți (Suceava County, Romania). A. Paratype, MP-UAIC 8228/PD.1/15, fertile cap in upper view. B. Paratype, MP-UAIC 8228/PD.2/17, fertile cap in lower view. C. Paratype, MP-UAIC 8228/PD.1/1, fertile cap in lower view. D. Paratype, MP-UAIC 8228/PD.2/25, fertile cap in upper view. E. Paratype, MP-UAIC 8228/PD.1/16, fertile cap in lower view. F. Holotype, MP-UAIC 8228/PD.1/6, fertile cap in lower view. G. Paratype, MP-UAIC 8228/PD.1/3, fertile cap in upper view. H. Paratype, MP-UAIC 8228/PD.2/21, an isolated spicule in the matrix. I. Group of five cysts in the matrix, MP-UAIC 8228/PD.3/22.

Fig. 5. Polyphysacean alga Acetabularia moldavica sp. nov. early Sarmatian, Pătrăuți (Suceava County, Romania). A. Paratype, MP-UAIC 8228/PD.1/10, fertile cap in lower view (A1) and detail (lower side) of fertile rays (A2). B. Paratype, MP-UAIC 8228/PD.3/8, fertile cap in upper view with an extracellular mineralized thin layer preserved (lower side). C. Paratype, MP-UAIC 8228/PD.1/3, detail of the fertile cap showing the upper coronae (see also Fig. 4G). D. Holotype, MP-UAIC 8228/PD.1/6, detail of the fertile cap showing lower coronae, notice the extracellular mineralization preserved between spicules (see also Fig. 4F). E. Paratype, MP-UAIC 8228/PD.1/12, detail of a fertile cap in upper view showing the upper coronae and aggregates of compacted mineralized cysts. F. Paratype, MP-UAIC 8228/PD.2/14, detail of two spicules with broken cysts.
Primary laterals, upper and lower corona: According to level at which the structure is broken and the side exposed, it is possible to observe the mineralized walls and cavities belonging to corona superior and inferior (Fig. 5C, D). The coronal structure of Fig. 7B1 (fragment of a cap in upper view), is probably broken at the basal part (primary lateral) level, and therefore prominences (arrows) correspond to mineralized walls among the basal parts (furrows). The pore in each furrow connects the basal part with the corona inferior below.
Fertile rays (gametophores): The gametophore develops beyond the basal part, and is about 1.2–3.2 mm long and 0.5–1.0 mm wide distally. The outer end is pointed (mucronate; Fig. 4A–G), but this character decreases as size increases (Fig. 5A1) and is not evident in Figs. 5B, 6B. The transverse distal sections are elliptical in small sized specimens and rectangular in large ones, both are arranged horizontally. A cap contains 10–24 gametophores.

Fig. 6. Polyphysacean alga Acetabularia moldavica sp. nov., early Sarmatian, Pătrăuți (Suceava County, Romania). A. Paratype, MP-UAIC 8228/PD.2/19, large fertile cap in lower view. B. Paratype, MP-UAIC 8228/PD.2/20, fragment of a fertile cap in upper view showing the extracellular mineralization. C. MP-UAIC 8228/PD.1, assemblage of fertile caps on the rock surface, disks are deposited with the upper side (white arrow) or the lower side (black arrow) exposed.
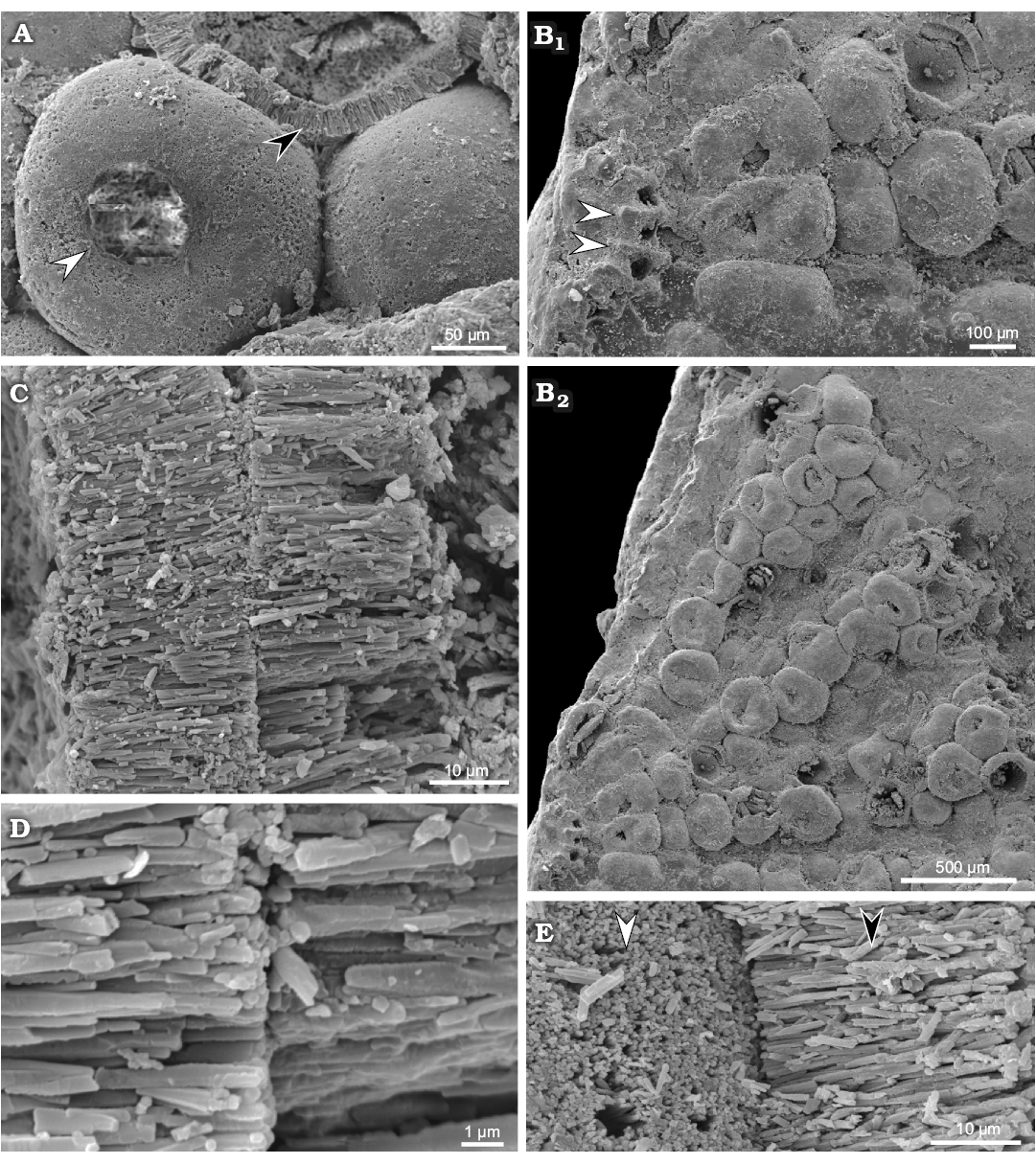
Fig. 7. Polyphysacean alga Acetabularia moldavica sp. nov., early Sarmatian, Pătrăuți (Suceava County, Romania). A. MP-UAIC 8228/PD.1, three mineralized gametangia (cysts), one shows the opening (white arrow head), another shows the fibrous layer (black arrow head); notice the empty space (not filled by calcareous matrix) between gametangia (cysts). B. MP-UAIC 8228/PD.1, part of a fertile disc, the coronal structure with three spicules, prominences (white arrow heads) correspond to mineralized walls among the basal parts (furrows) (B1), pores probably represent the communication to the lower coronae; general view of spicules with mineralized cysts (B2). C. MP-UAIC 8228/PD.1, contact between fibrous aragonitic layers of two gametangia (cysts). D. MP-UAIC 8228/PD.1, sharp contact between two fibrous layers. E. MP-UAIC 8228/PD.1, contact between two gametangia (cysts), outer surface of the mineralized layer (white arrow head) and aragonitic fibrous structure of a broken cyst layer (black arrow head).
Gametangia (cysts): Fertile rays are estimated to contain 14–35 mineralized gametangia (cysts), closely packed so that their outlines are roughly polygonal to quadrangular. Cysts are usually bound together at the cyst-cyst contact (Fig. 7C–E). The spaces between cysts appear to be uncalcified (Fig. 7A), but very occasionally micro-spherulitic crystal aggregates have been observed under SEM (Fig. 8). Mineralized cysts are 0.26–0.38 mm in diameter. The wall is 20–25 μm thick, fibrous, made by acicular crystals arranged orthogonal to the wall surface (seemingly original aragonite). Each gametangium has a circular opening, 50 μm wide, closed by a mineralized lid, that is commonly lost.
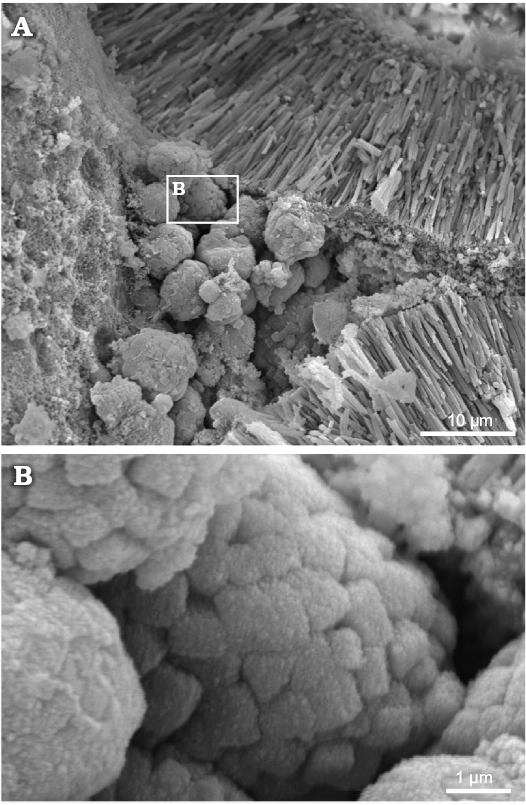
Fig. 8. Polyphysacean alga Acetabularia moldavica sp. nov., MP-UAIC 8228/PD.1a, early Sarmatian, Pătrăuți (Suceava County, Romania). A. Space among three mineralized gametangia (cysts), partially filled by calcareous micro-spherules. B. Detail of micro-spherules, showing the equant crystals.
Reconstruction of the thallus: Figure 9 depicts the possible appearance of the alga in axial (Fig. 9A, B) and apical view (Fig. 9C). Extracellular mineralization is drawn in black, and the intracellular mineralized cyst wall is dark gray (Fig. 9A, B). Cyst structure is shown in Fig. 10A.
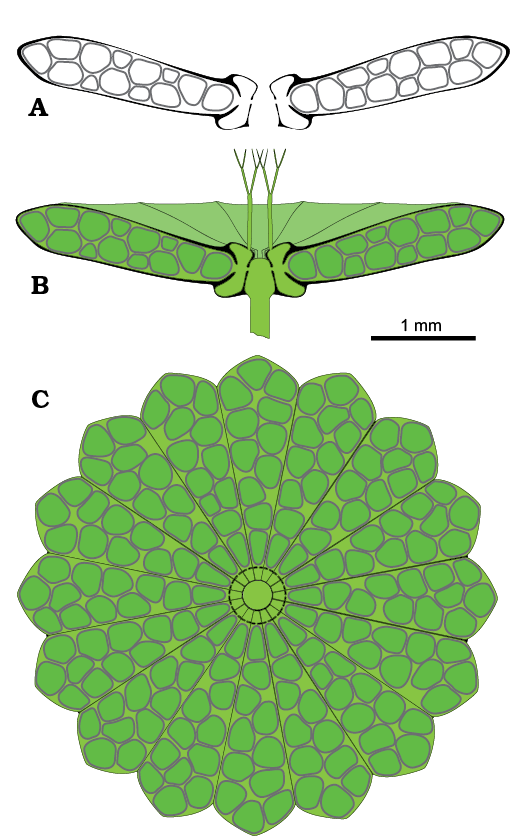
Fig. 9. Reconstruction of polyphysacean alga Acetabularia moldavica sp. nov. A. Mineralized fertile cap in axial section. B. Soft parts (green) and calcareous skeleton (black and dark gray) in axial view. C. Fertile cap in upper view.

Fig. 10. Scheme of gametangium and mineralization of Acetabularia moldavica sp. nov. (A) and extant polyphysaceans (B). A. Gametangium in axial section (A1), stratified wall (gray), and mineralization (sector); detail of the cyst wall with acicular crystals of aragonite (A2). B. Extant polyphysaceans (B1), cyst wall and detached lid (gray), intergametangial space (wavy lines), not to scale. Acetabularia schenckii (B2), arrangement of aragonite crystals (data from Bailey et al. 1976). Chalmasia antillana from Florida Bay (1, Blackwater Sound; 2, Rodrigues Key) (B3), arrangement of aragonite crystals (data from Bailey et al. 1976; Marszalek 1975).
Comparisons.—Acetabularia moldavica sp. nov. differs from all other fossil Acetabularia species by the presence of mineralized gametangia. Moreover, the number of fertile rays in a cap is very low (10–24), whereas it is much higher in A. miocenica (90–100), A. chiavonica (78), and A. transylvana (90–100).
Remarks.—The sediments of Moldavian Platform represent lacustrine environments evolving into peat bog facies (observed to alternate cyclically), while being connected to the Carpathian Orogen by temporary land bridges (Ionesi et al. 1993; Ţibuleac 1999; Ionesi 2006).
On the contrary, there is no consensus regarding reverse brackish-marine evolution, despite its being reported within the Moldavian Platform (Ţibuleac 1999; Ionesi 2006).
Dasycladaceans are euryhaline organisms and prefer shallow water conditions ranging from mangrove swamps to coral reefs environments (Valet 1979). The pelitic facies containing A. moldavica sp. nov. suggests a soft substrate and in-situ fossilisation. The alga probably populated environments equivalent to a mangrove swamp. Acetabulariaceans quoted by Génot et al. (2002) probably required marine conditions.
Stratigraphic and geographic range.—Type locality and horizon only.
Genus Patruliuspora nov.
Etymology: In honour of Dan Patrulius (1921–1982), pioneer of dasycladalean studies in Romania.
Type species: Chalmasia morelleti Pokorný, 1948; middle Miocene, Podivin, Czech Republic.
Species included: Halicoryne carpatica Mišík, 1987, Upper Triassic (Carnian–Rhaetian) of the Western Carpathians; Acicularia valeti Segonzac, 1970 (Segonzac 1970, 1976) from the Paleocene of the French Pyrenees. It is possible that other species referred to the genus Acicularia but displaying Type D biomineralization (Fig. 11) could belong to Patruliuspora.
Diagnosis.—Group of mineralized gametangia (cysts), immersed or not in a calcified mass to form ellipsoidal, irregular to variously elongated club shaped aggregates of cysts.
Remarks.—Patruliuspora is restricted to fossil taxa falling in types C and D of Fig. 11. As discussed above, they may be the fossil remains of polyphysacean gametophores, presently belonging to Chalmasia, Halicoryne, or Acetabularia. In any case, we cannot exclude the possibility that in the past Parvocaulis or other extinct genera of Polyphysaceae, if not Triploporellaceae, might have produced equivalent mineralized aggregates of cysts.
Numerous genera are known to be mineralized parts of reproductive organs of dasycladaleans. Some of these are thought to be by-products of extracellular biomineralization of single gametophores (ampullae), Dameryella Bignot and Génot, 1998 (Génot 2009), or tufts of gametophores Carpenterella Morellet and Morellet, 1922 (Génot 2009). Small bodies formed by intracellular biomineralization include Acicularia Archiac, 1843, Terquemella Morellet and Morellet, 1922, Frederica Barta-Calmus, 1966 and Russoella Barattolo, 1983. From a morphological point of view, Terquemella and Russoella are discoidal and not ellipsoidal, and Acicularia and Frederica are elongate club-shaped and ovoidal, respectively (Génot 2009). However, all those genera show biomineralization of type B (Fig. 11), rather than type C or D as in Patruliuspora.
Stratigraphic and geographic range.—Species of Patruliuspora ranges from Late Triassic to Miocene and includes at least three species: Patruliuspora morelleti (Pokorný, 1948) comb. nov., P. carpatica (Mišík, 1987) comb. nov., and P. valeti (Segonzac, 1970) comb. nov.
The Jurassic–Cretaceous gap can be interpreted in different ways, for example: (i) because the taxon is a form-genus, it may assemble fossils from different contexts, but with similar morphologies (e.g., cladosporous, choristosporous, or goniosporous type of reproduction); (ii) the ellipsoidal aggregates may display different type of mineralization (e.g., type B and C of Fig. 11). Viewed in this light, Upper Jurassic–Lower Cretaceous ellipsoidal aggregates such as Acicularia elongata Carozzi, 1955, and Acicularia endoi Praturlon, 1964 could be accommodated within the genus Patruliuspora.
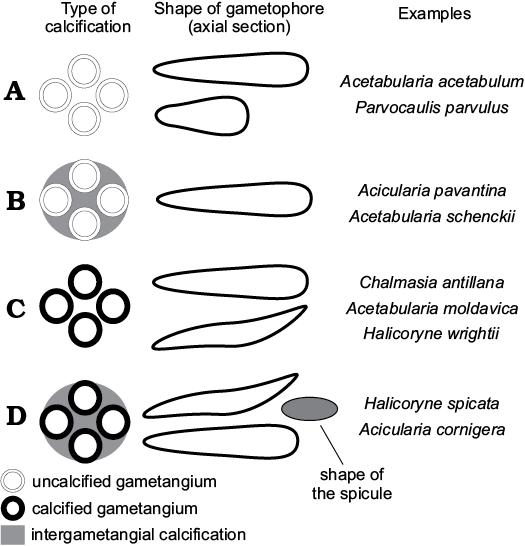
Fig. 11. Scheme of intracellular mineralization in polyphysaceans (gametophores are sketched in cross section). A. Gametangia and intergametangial space not mineralized. B. Gametangia unmineralized, intergametangial space mineralized. C. Gametangia mineralized, intergametangial space not mineralized. D. Gametangia and intergametangial space mineralized.
Discussion
Intracellular mineralization of gametophores.—Gametangia of most extant species of acetabulariaceans are uncalcified, and the unmineralized cyst wall of Acetabularia has been shown to be cellulose (Nisizawa et al. 1974; Herth et al. 1975). Only four out of eighteen species, i.e., A. schenckii, Chalmasia antillana, Halicoryne wrightii, and H. spicata, display variously mineralized gametangia (Berger and Kaever 1992).
Solms-Laubach (1895) widely addressed biomineralization inside gametophores and around the cysts. The cyst membrane of Halicoryne wrightii is particularly thick and he recognized a three-layered wall (Solms-Laubach 1895: 14–15), the middle one being very thick and the seat of the biomineralization in the form of aragonite. The scheme is sketched in Fig. 10B1. The lime mass binding cysts together, observed in Acicularia and Halicoryne spicata, was interpreted to be the result of a biomineralization process external to the cyst wall (Solms-Laubach 1895: 15). The ultrastructure in Acetabularia schenckii revealed by SEM shows that the calcareous matrix consists of a layer of parallel aragonite crystals adjacent and normal to the inside cyst membrane, whereas outwards they are more irregularly arranged (Bailey et al. 1976). A structural scheme based on Bailey et al. (1976) is shown in Fig. 10B2.
In Chalmasia antillana the layer of parallel aragonite crystals is set along the outer surface of the thick mineralized wall (Marszalek 1975; Bailey et al. 1976). More inward, acicular crystals, 7 μm long and 0.4 μm thick, are arranged irregularly. The inner surface is formed by flat lying acicular crystals (Marszalek 1975), which show mineralized cysts of the same species from Florida Bay composed of granular to acicular crystals ranging in size from less than one micron to 7 μm in length. The two schemes described by Marszalek (1975) are drawn in Fig. 10B3.
Extant and fossil polyphysaceans display different degrees of intracellular biomineralization that can be grouped in four classes. More than 80% of extant polyphysacean species exhibit fertile rays free of intracellular mineralization (type A of Fig. 11). Acicularia pavantina Archiac, 1843, a middle Eocene species from the Paris Basin, which is the type species of the genus Acicularia, looks like small mineralized rods pierced by rounded cavities set peripherally. Cavities correspond to cysts surrounded by lime (Génot 2009). The cyst wall was not mineralized, as in Acetabularia schenckii, and therefore the cavity matches its outer mould (type B of Fig. 11). In Halicoryne wrightii the thick membrane is mineralized, and cysts are free (intergametangial biomineralization is absent; Berger and Kaever 1992). In fossils, cavities correspond to their inner mould (type C of Fig. 11). Type C occurs also in Chalmasia antillana (Berger and Kaever 1992) and Acetabularia moldavica sp. nov. In Halicoryne spicata mineralized cysts are grouped together by intergametangial biomineralization (type D of Fig. 11), thus forming ellipsoidal aggregates of cysts (Berger and Kaever 1992). Rare species of Acicularia exhibit such a structure, for example Acicularia cornigera Morellet and Morellet, 1922 from the Lutetian of Brittany (Génot 2009), and perhaps Acicularia miocaenica Reuss, 1861 from the Miocene of Romania (Reuss 1861; Valet and Segonzac 1969) and Acicularia valeti Segonzac, 1970 from the upper Paleocene of the French Pyrenees.
Biomineralization of the cyst wall displayed in Acetabularia moldavica sp. nov., Chalmasia antillana, Halicoryne wrightii, and H. spicata demonstrates that such kind of biomineralization may occur in different genera. Therefore, this character seems to be a specific rather than generic attribute.
Living fossil and fossil living taxa: a problem.—Two examples are emblematic: Acetabularia schenckii Möbius, 1889 and Halicoryne morelleti (Pokorny, 1948): (i) Acetabularia schenckii, an extant species referred to a fossil genus (Acicularia), was first described by Moebius (1889) and assigned to Acetabularia. Later the species was transferred to the genus Acicularia by Solms-Laubach (1895) because the cysts were surrounded by calcium carbonate forming a mineralized spicule as in Acicularia pavantina Archiac, 1843 (Génot 2009), type species of the genus Acicularia (type B of Fig. 11). Solms-Laubach (1895) considered the intracellular biomineralization (mineralized spicule) so distinct as to assume generic status and regarded Acicularia schenckii as a living representative of the genus Acicularia. By extension, the characters of the soft parts of Acicularia schenckii were considered to reflect those of fossil examples, Acicularia schenckii therefore implicitly became for phycologists the reference species of the genus Acicularia and was assumed to be an extant genus. The significance of the intracellular biomineralization at genus or species level has been widely debated, without attaining a general consensus (Howe 1901; Egerod 1952; Valet 1969; Bailey et al. 1976; Berger and Kaever 1992); (ii) Halicoryne morelleti (Pokorný, 1948) is an example running in the opposite direction, a fossil species assigned to an extant genus. The species consists of mineralized cysts, either isolated or grouped in ellipsoidal aggregates due to intergametangial biomineralization (type D of Fig. 11). Pokorný (1948) originally referred the species to the extant genus Chalmasia. Then, Miletic-Spajic (1961) attributed it to Acicularia and finally Valet and Segonzac (1969) moved it to Halicoryne. A more complete discussion on this taxon and its ultrastructure is provided by Bucur et al. (1993) and Bucur and Şuraru (1994). Isolated cysts were also figured by Paruch-Kulczycka (1994).
The fossilization of individually mineralized aggregates of cysts (intracellular biomineralization with no extracellular biomineralization), present in most Acicularia species and in Halicoryne morelleti, has the effect that fundamental characters such as coronae are lost, because they are only preserved by extracellular biomineralization in the fossils. Thus, even if the resemblance to extant counterparts is evident, it is not possible to argue whether these fossil taxa would have had one or two coronae, and the attribution of Cenozoic species to Acetabularia, Parvocaulis, Chalmasia, or Halicoryne therefore appears speculative. The attribution seems even more hypothetical if not misleading for Mesozoic taxa (e.g., Mišík 1987). It is a matter of fact that palaeophycologists handle dasycladalean mineralized aggregates of cysts (Acicularia, Terquemella, etc.) as form-taxa (Barattolo 1983). Therefore, we agree with Bailey et al. (1976) to limit the genus Acicularia to the sole fossil species.
From a paleontological point of view, fossil mineralized aggregates of cysts like Halicoryne morelleti should be distinguished from their possible extant counterparts (Chalmasia, Halicoryne, and Acetabularia) when coronal structures are not preserved. As already proposed by Granier and Berthou (2002), such fossil species should be ascribed to a form-genus.
Sedalanella Elliott, 1986 and Ioanella Granier and Berthou, 2002.—Halicoryne morelleti has been compared by Elliott (1986) and Granier and Berthou (2002) with the fossil genera Sedalanella and Ioanella respectively. The former genus was defined by the following diagnosis “Calcareous, thin-walled, near-spherical hollow body, slightly flattened on one hemisphere” (Elliott 1986: 741). The author supplied two photographs, the one contained the holotype (Elliott 1986: text-fig. 1A) and part of two other specimens. Further paratypes were photographed by FB more than 30 years ago and are here reported in Fig. 12A. The fossil was interpreted as reproductive cysts from an extinct, largely uncalcified, dasycladalean alga possibly related to Halicoryne (Elliott 1986: 740). The recrystallized spherical bodies were supposed to have a very thin mineralized layer (6 μm), but Fig. 12A1, A2, in addition to the outer micritic film common to the all specimens, shows an inner micritic film. The spherical cavity limited by the inner dark film should represent the real volume of the reproductive organ. If so, then the distance between the two dark layers corresponds to the mineralized wall thickness (80–90 μm). Radial short pores filled by micrite occur all around the outer layer, piercing it (Fig. 12A). In Fig. 12A1 two radial pores reach the inner surface. In summary, the fossil represents spherical bodies with a central, spherical hollow cavity, the moderately thick mineralized wall is pierced by numerous thin, tubular radial pores, and most of the elements have been obliterated by recrystallization. As stated by Barattolo (2002: 25–26) the general feature of Sedalanella Elliott, 1986 corresponds to the genus Oroseina Dieni, Massari, and Radoičić, 1985, a fossil related to neomerid mineralized gametangia (Barattolo 1998: 89). Therefore, Seladanella has been considered a junior synonym of Oroseina (Barattolo 2002). Moreover, because of the biometrical correspondence, the species Sedalanella segonzacae should also be considered a junior synonym of Oroseina solaris.
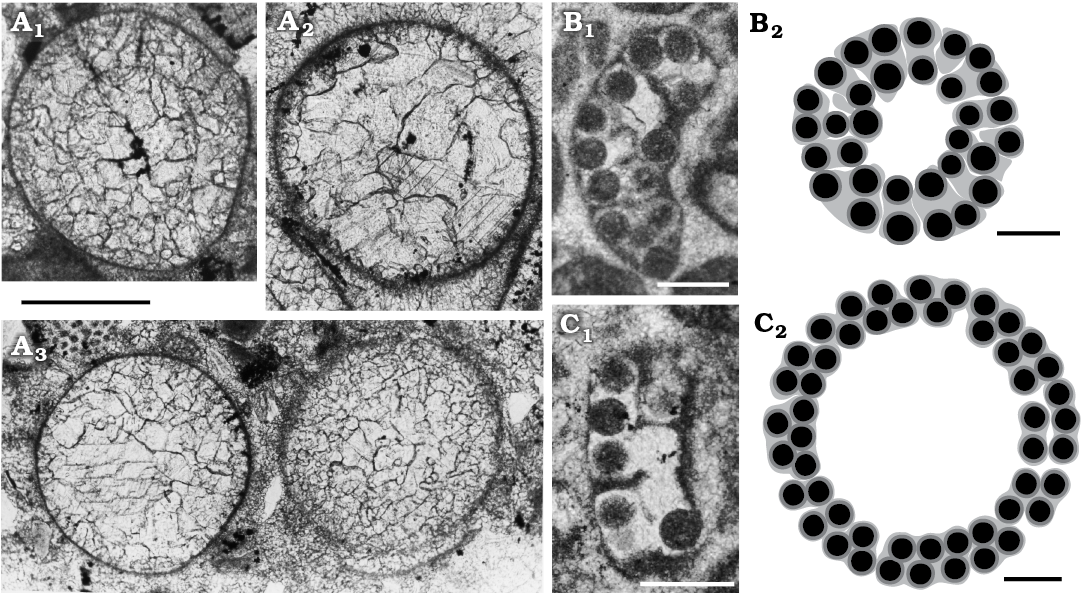
Fig. 12. Mineralized reproductive organs of dasycladalean alga Sedalanella segonzacae Elliott, 1986 from Palaeocene–lower Eocene, Sedalan (Kurdistan, Iraq) (A), and Ioanella lusitanica Granier and Berthou, 2002 from Ferreiras (Central Algarve, Portugal) (B, C). A. BM (NH) V. 41606, paratypes; A1, specimen displaying vestige of internal cavity (= Elliott 1986: fig. 1A, right specimen); A2, specimen with the internal cavity outline as in A, the micritic outer layer showing evidence of radial pores; A3, two specimens with different degrees of preservation, both show no trace of an internal cavity. B. LPB-29900(A)/B, holotype (1st specimen in http://paleopolis.rediris.es/IFAA/jpg/Granier/Cluj_02_02.html). C. LPB-29900(A)/A (3rd specimen in http://paleopolis.rediris.es/IFAA/jpg/Granier/Cluj_02_02.html). Photographs (B1, C1), interpretative drawings (B2, C2). Scale bars 200 µm.
The genus Ioanella has been established to group mineralized aggregates of cysts formerly assigned to the genus Halicoryne (H. carpatica Mišík, 1987 and H. morelleti [Pokorny, 1948] Valet and Segonzac, 1969) with the following diagnosis: “Aggregates of randomly arranged small cysts” (Granier and Berthou 2002: 120). The genus Ioanella is based on a third species, Ioanella lusitanica Granier and Berthou, 2002, from the Berriasian of the Central Algarve (Portugal). Only two specimens were illustrated in the original publication (Granier and Berthou 2002: pl. 2: 5, 6) but further photographs of the type material were later uploaded by Bruno Granier to the internet (http://deptsec.ku.edu/~ifaaku/jpg/Granier/Cluj_02_02.html; accessed on 15 October 2018). The holotype as well as several other sections (e.g., the 3rd, 6th, 16th, and 19th specimens, from left to right and from top to bottom) appear as an arched wall of loosely cemented cysts mainly arranged in two rows truncated at both ends. Tangential sections display more or less rounded contours (15th, 18th, 20th specimens). These two kinds of sections are compatible with a spherical cap. According to this view, sections with an irregular shape could be interpreted as various oblique sections. Moreover, the fact that the calcareous wall is crossed by a fissuration, radial and parallel to the surface, makes the structure rather delicate. Therefore, the little aggregate of cysts could be the result of fragmentation of a wider, rounded, calcified body. A possible reconstruction of two entire specimens (the holotype and 3rd specimen) is given in Fig. 12B2, C2. Fig. 12B2 compared with Fig. 12C2, displays a narrower central cavity and a thicker wall with more than two layers of cysts. The incongruity can be explained by a nearly tangential cut (Fig. 12B2) versus a transversal one (Fig. 12C2).
The plausibly large size of the complete fossil (more than 1 mm), and the two layers of gametangia (cysts) with a large empty internal space, make it unlikely to be a mineralized product within an ampulla or a fertile lateral. More likely, Ioanella lusitanica corresponds to the aggregate of cysts within the central stem (endosporous reproduction) of an uncalcified club-shaped to spherical dasycladalean. Thus, Ioanella should be closer to Atractyliopsis and Aciculella than to Acicularia or Frederica.
The nature of Ioanella lusitanica needs to be verified by more abundant material, but in any case, the spherical caps of two layers of cysts in Ioanella lusitanica conform poorly with the small ellipsoidal calcareous bodies of Halicoryne morelleti. On this basis, the latter species is here separated from the genus Ioanella and the new genus Patruliuspora is proposed for it.
Conclusions
Fossil remains of polyphysacean fertile caps have been found in Sarmatian rocks near Suceava (NNE Romania). The new taxon, Acetabularia moldavica sp. nov., is attributed to the extant genus Acetabularia because it exhibits structural elements referable to inferior and superior coronae. Gametangia are preserved and closely packed within the gametophore. The gametangium wall is strongly mineralized and, as shown in SEM, the thick layer is made by acicular aragonite crystals arranged perpendicularly to the wall surface. The crystal arrangement differs from extant counterparts (Chalmasia antillana and Acetabularia schenckii), as shown by Marszalek (1975) and Bailey et al. (1976).
A critical review of the paleontological literature allows us to confirm one species and to ascribe an additional two species to the genus Acetabularia: A. miocaenica Andrussow, 1887, A. chiavonica Squinabol, 1902 and A. transylvana (Bányai and Morellet, 1936). The occurrence of mineralized cysts distinguishes the new species from all other known fossil and extant species of Acetabularia. Apart from its ultrastructure, A. moldavica sp. nov. shares cyst biomineralization with Chalmasia antillana, Halicoryne wrightii, and H. spicata, a character that appears to be typical at species rather than genus level.
Within the polyphysaceans at least four types of intracellular biomineralization can be identified (Fig. 11). Type A, cysts and intergametangial space not mineralized; more than 80% of extant species exhibit this type. Type B, cyst wall not mineralized, intergametangial space mineralized; this occurs in Acetabularia schenckii and in most Acicularia. Type C, cyst wall mineralized, intergametangial space not mineralized; this type is observed in Halicoryne wrightii, Chalmasia antillana, and Acetabularia moldavica sp. nov. Finally type D, cyst wall and intergametangial space mineralized, as in Halicoryne spicata.
Assignment of an extant species to a fossil genus, and vice-versa, can be problematic when a fossil consists of a mineralized spicule (intracellular biomineralization).
Two contrasting examples are discussed: (i) Acetabularia schenckii (extant species) assigned to the genus Acicularia (fossil), and (ii) Halicoryne morelleti (fossil species) referred to an extant genus (Halicoryne). Even if the resemblance to extant counterparts is evident, it is not possible to argue confidently whether fossil taxa would have had one or two coronae. Consequently, ascribing Cenozoic species to an extant genus appears speculative. We therefore support restricting the genus Acicularia to the sole fossil species.
From a paleontological point of view fossil mineralized aggregates of cysts such as Halicoryne morelleti should be set apart from their possible extant counterparts (Chalmasia, Halicoryne, and Acetabularia) when coronal structures are not preserved. For these fossils, we propose the new genus Patruliuspora. Moreover, a new interpretation of the genus Ioanella Granier and Berthou, 2002 is supplied.
Acknowledgements
The dasycladalean algae were collected by VI on 30.08.2002 togetheter with Loredana Marlen Cuțic-Costaș (Rădăuți, România) during a fieldtrip devoted to sampling of Sarmatian rocks for her licenciate thesis. We thank her for her field support. We thank Bruno Granier (Université de Bretagne Occidentale, Brest, France), Robert Riding (University of Tennessee, Knoxville, USA), and Nicola Carras (Institute of Geology & Mineral Exploration, Acharnes, Greece) for their very helpful reviews of the manuscript, and Roberto De Gennaro (Università di Napoli Federico II, Naples, Italy) for his expert technical support in taking the SEM photos. This work was supported by the University of Naples Federico II under the bilateral accord with the University Alexandu Ioan Cuza of Iaşi, Romania.
References
Andrussow, N. 1887. Eine fossile Acetabularia als gesteinbildender Organismus. Annalen des K.K. Naturhisthistorischen. Hofmuseums Wien 2: 77–80.
Archiac, E.J. d’ 1843. Description géologique du département de l’Aisne. Mémoires de la Société géologique de France 1: 129–418.
Băcăuanu, V., Barbu, N., Pantazică, M., Ungureanu, A., and Chiriac, D. 1980. Podişul Moldovei. 345 pp. Editura Ştiinţifică şi Enciclopedică, Bucureşti.
Bailey, G.P., Rezak, R., and Cox, E.R. 1976. A revision of generic concepts of living members in the subfamily Acetabularieae (Dasycladaceae, Dasycladales) based on scanning electron microscopy. Phycologia 15: 7–18. Crossref
Bányai, J. 1929. Sarmatische Acicularia und Neritina im Tale von Kis-Homoród (Szeklerland). Erdélyi Irodalmi Szemle 6: 320–327.
Bányai, J. and Morellet, L. 1936. Deux Acétabulariacées nouvelles du Sarmatien de Transylvanie. Bulletin de la Section scientifique de l’Académie de Roumanie 17: 168–172.
Barattolo, F. 1983. Su alcuni organi riproduttori isolati di Dasicladacee nel Cretacico dell’Appennino. Bollettino della Società dei Naturalisti in Napoli 92: 17–49
Barattolo, F. 1998. Dasycladacean green algae and microproblematica of the uppermost Cretaceous–Paleocene in the Karst area (NE Italy and Slovenia). In: L. Hottinger and K. Drobne (eds.), Paleogene Shallow Benthos of the Tethys. Opera Dela Slovenska Akademija Znanosti in Umetnosti 34 (2): 65–128.
Barattolo, F. 2002. Late Cretaceous–Paleogene dasycladaleans and the K/T Boundary problem. In: I. Bucur and S. Filipescu (eds.), Research Advances in Calcareous Algae and Microbial Carbonates, 19–40. Cluj University Press, Cluj-Napoca.
Barattolo, F. 2006. Re-description of Cymopolia miocenica (Karrer) (Chlorophyta, Dasycladales) from the Late Miocene of Sicily (Italy) and the Middle Miocene of Banat (Romania). Bollettino della Società Paleontologica Italiana 45: 15–28.
Berger, S. 2006. Photo-atlas of living Dasycladales. Carnets de Géologie 2006/02: CG2006_BOOK_02, http://hdl.handle.net/2042/5831.
Berger, S. and Kaever, M. 1992. Dasycladales, an Illustrated Monograph of a Fascinating Algal Order. vii + 247 pp. Georg ThiemeVerlag, Stuttgart.
Berger, S., Fettweiss, U., Gleissberg, S., Liddle, L.B., Richter, U., Sawitsky, H., and Zuccarello, G.C. 2003. 18S rDNA phylogeny and evaluation of cap development in Polyphysaceae (formerly Acetabulariaceae; Dasyladales, Chlorophyta). Phycologia 42: 506–561. Crossref
Boda, J. 1959. Algae. In: J. Boda (ed.), Das Sarmat in Ungarn und seine Invertebraten-Fauna. Annales Instituti Geologici Publici Hungarici 47: 749–752.
Bucur, I.I., Nicorici, E., and Şuraru, N. 1993. Sarmatian calcareous algae from Romania. In: F. Barattolo, P. De Castro, and M. Parente (eds.), Studies on Fossil Benthic Algae. Bollettino della Società Paleontologica Italiana, Special Volume 1: 81–91.
Bucur, I.I. and Şuraru, N. 1994. Halicoryne morelleti (Pokorny) from the lower Sarmatian deposits of the Borod basin (Romania). In: E. Nicorici (ed.), The Miocene from the Transylvanian Basin, Romania, 41–46. Editura Carpatica, Cluj.
Deloffre, R. and Granier, B. 1992. Inventaire critique des algues Dasycladales fossiles. Revue de Paléobiologie 11: 331–356.
Dumais, J. and Harrison, L.G. 2000. Whorl morphogenesis in the dasycladalean algae: the pattern formation viewpoint. Philosophical Transactions Royal Society of London, Biological Sciences 355: 281–305. Crossref
Egerod, L.E. 1952. An analysis of the siphonous Chlorophycophyta with special reference to the Siphonocladales, Siphonales and Dasycladales of Hawaii. University of California Publications in Botany 25: 325–453.
Elliott, G.F. 1986. Isolated algal cysts in the Palaeocene–Lower Eocene of Kurdistan. Palaeontology 29: 739–741.
Génot, P. 1987. Les Chlorophycées calcaires du Paléogène d’Europe nord-occidentale – Bassin de Paris, Bretagne, Cotentin, Bassin de Mons. 500 pp. Ph.D. Thesis, Sciences de la Terre, Université de Nantes, Nantes.
Génot, P. 2009. Cenozoic Dasycladales. A photo-atlas of Lutetian species from French Cenozoic basins. Carnets de Géologie 2009/01: CG2009_SP01, http://hdl.handle.net/2042/21981.
Génot, P., Ionesi, V., and Bucur, I.I. 2002. Preliminary results from the study of Acetabulariaceae fertile caps discovered in Sarmatian sediments from Moldavia (Romania). In: I.I. Bucur and S. Filipescu (eds.), Research Advances in Calcareous Algae and Microbial Carbonates, 95–101. Cluj University Press, Bolzano.
Génot, P., Saint-Martin, J.-P., and Bucur, I.I. 2008. Polyphysaceae fertile caps in Hungarian Sarmatian sediments. Geologia Croatica 61: 341–344.
Giusberti, L., Roghi, G., Martinetto, E., Fornasiero, M., and Simonetto, L. 2014. The Palaeogene flora of northern Italy. In: E. Kustatscher, G. Roghi, A. Bertini, and A. Miola (eds.), Palaeobotany of Italy, 206–231. Naturmuseum Sudtirol, Bolzano.
Granier, B. and Berthou, P.Y. 2002. Algues calcaires fossiles, nouvelles ou peu connues du Portugal. 1ère partie. In: I.I. Bucur and S. Filipescu (eds.), Research Advances in Calcareous Algae and Microbial Carbonates, 117–126. Cluj University Press, Cluj-Napoca.
Herth, W., Kuppel, A., and Franke, W.W. 1975. Cellulose in Acetabularia cyst walls. Journal of Ultrastructural Research 50: 289–292. Crossref
Howe, M.A. 1901. Observations on the algal genera Acicularia and Acetabulum. Bulletin of the Torrey Botanical Club 28: 321–334. Crossref
Ionesi, B. 1968. Stratigrafia depozitelor miocene de platformă dintre Valea Siretului şi Valea Sucevei (Platforma Moldovenească). 395 pp. Academy Press, Bucuresti.
Ionesi, L. 1994. Geologia unităţilor de platformă şi a Orogenului Nord-Dobrogean. 280 pp. Tehnique Press, Bucuresti.
Ionesi, L. 1995. Sarmatian of the Moldavian Platform. General data. In: L. Ionesi, B. Ionesi, M. Brânzilă, N. Trelea, P. Ștefan, and V. Ionesi (eds.), Guide to Excursion A2-Sarmatian of the Moldavian Platform. Romanian Journal of Stratigraphy 76 (2): 1–8.
Ionesi, V. 2006. Sarmaţianul dintre Valea Siretului şi Valea Şomuzului Mare. 238 pp. Alexandru Ioan Cuza Univeristy of Iași Press, Iași.
Ionesi, L., Ionesi, B., and Ţibuleac, P. 1993. Observaţii asupra depozitelor cu cărbuni din zona Fălticeni-Boroaia (Platforma Moldovenească). Bulletin de la Societé Geologique Series 4 14: 89–95.
Małecki, J. 1970. Chlorophyta from Miocene sediments of Poland. Rocznik Polskiego Towarzystwa Geologicznego 40: 167–176.
Małecki, J. 1974. Grünalgen Chlorophyta aus dem sarmatischen Ablagerungen von Gliwice Stare (Polen). In: J. Seneš and E. Brestenská (eds.), Chronostratigraphie und Neostratotypen, Miozan M5 Sarmatien, 598–605. VEDA, Verlag der Slowakischen Akademie der Wissenschaften, Bratislava.
Marszalek, D.S. 1975. Calcisphere ultrastructure and skeletal aragonite from the alga Acetabularia antillana. Journal of Sedimentary Research 45: 266–271. Crossref
Maslov, V.P. 1955. New forms of Tertiary algae [in Russian]. Doklady Akademii Nauk SSSR 103: 145–148.
Maslov, V.P. 1960. Aciculariae and their importance for stratigraphy of the USSR [in Russian]. Paleontologičeskij žhurnal 3: 115–122.
Miletic-Spajic, O. 1961. Beleska o nalaski Krecnjackih algi u sarmatien. Vesnik Zavoda za geološka i geofizička istraživanja, Serija A 19: 598–605.
Mišík, M. 1987. The alga Halicoryne carpatica n. sp. from Upper Triassic of western Carpathians. Geologický sborník – Geologica Carpathica 38: 285–293.
Moebius, M. 1889. Bearbeitung der von H. Schenck in Brasilien gesammelten Algen. Hedwigia 28: 309–347.
Morellet, L. and Morellet, J. 1927. Les Dasycladacées du Néogène de Kostej (Banat) et de Lapugy (Transylvanie). Bulletin de la Société géologique de France 36: 223–228.
Morellet, L. and Morellet, J. 1939. Sur une nouvelle espece d’Acicularia du Sarmatien moyen d’Azam, Iran septentrional. Eclogae Geologicae Helvetiae 32: 31–32.
Nisizawa, K., Kuroda, K., Tomita, Y., and Shimahara, H. 1974. Main cell wall constituents of the cysts of Acetabularia. Botanica Marina 17: 16–19. Crossref
Paruch-Kulczycka, J. 1994. Algae in the Sarmatian deposits from the Machów outcrop and from the boreholes Jamnica M-83 and S-119 (Carpathian Foredeep). Geological Quarterly 38: 571–576.
Pokorný, V. 1948. Chalmasia morelleti n. sp. (Dasycladacées) du Sarmatien de Tchéchoslovaquie. Bulletin international de l’Académie tchèque des Sciences 49: 13–17.
Popov, S.V., Rögl, F., Rozanov, A.Y., Steininger, F.F., Shcherba, I.G., and Kováč, M. 2003. Lithological-paleogeographic Maps of Paratethys. 10. Maps Late Eocene to Pliocene. Paleontological Institute RAS, Moscow, Forschungsinstitute and Naturmuseum Senckenberg, Frankfurt am Main.
Reuss, A.E. 1861. Über die fossile Gattung Acicularia d’Arch. Sitzungsberichte Akademie Wissenschaften Wien, Mathematische-Naturwissenshaften Klasse (I) 43: 7–10.
Rögl, F. 1999. Mediterranean and Paratethys. Facts and hypotheses of an Oligocene to Miocene. Paleogeography (Short Overview). Geologica Carpathica 50: 339–349.
Segonzac, G. 1970. Essai de classement de quelques Acétabulariacées tertiaires (algues calcaires). Bulletin de la Société d’Histoire Naturelle de Toulouse 106: 333–340.
Segonzac, G. 1976. Dasycladacées nouvelles ou peu connues du Thanétien des Pyrénées. Bulletin de la Société d’Histoire Naturelle de Toulouse 112: 123–136.
Solms-Laubach, H. graf zu 1895. Monograph of the Acetabularieae. Transactions of the Linnean Society of London, 2nd Series. Botany 5 (1): 1–39.
Squinabol, S. 1902. Di una specie fossile di Acetabularia. Atti e Memorie della R. Accademia di Scienze Lettere ed Arti di Padova 18: 151–155.
Stancu, J. and Tautu, E. 1973. Dasycladacele sarmatiene din Romania. Dări de Seamă ale Şedinţelor 60: 237–247.
Stancu, J. and Tautu, E. 1974. Dasycladacees der Sarmatischen ablagerungen. In: J. Seneš and E. Brestenská (eds.), Chronostratigraphie und Neostratotypen-Miozän der Zentralen Paratethys: M5 Sarmatien, 606–623. VEDA, Verlag der Slowakischen Akademie der Wissenschaften, Bratislava.
Studencka, B. and Jasionowski, M. 2011. Bivalves from the Middle Miocene reefs of Poland and Ukraine: A new approach to Badenian/Sarmatian boundary in the Paratethys. Acta Geologica Polonica 61: 79–114.
Țibuleac, P. 1999. Studiul geologic al Sarmațianului din zona Fălticeni-Baia-Boroaia (Platforma Moldovenească), cu referire specială asupra stratelor de cărbuni. 360 pp. Ph.D. Thesis, University “Alexandru Ioan Cuza”, Iași.
Ţibuleac, P. 2009. Sarmaţianul cu strate de cărbuni din zona Fălticeni-Boroaia (Platforma Moldovenească). 313 pp. Tehnopress, Iaşi.
Valet, G. 1969. Contribution à l’étude des Dasycladales. 2 Cytologie et reproduction. 3 Révision systématique. Nova Hedwigia 17: 551–644.
Valet, G. 1979. Approche paléoécologique du monde des Dasycladales a partir de l’écologie des formes actuelles. Bulletin du Centre de Recherches Exploration-Production elf-Aquitaine 3/2: 859–866.
Valet, G. and Segonzac, G. 1969. Les genres Chalmasia et Halicoryne (Algues Acétabulariacées). Bulletin de la Société géologique de France 11: 124–127. Crossref
Acta Palaeontol. Pol. 64 (1): 85–100, 2019
https://doi.org/10.4202/app.00537.2018