

New archaeorthopteran insects from the Carboniferous of Poland: Insights into tangled taxonomy
TOMÁŠ DVOŘÁK, MARTINA PECHAROVÁ, WIESŁAW KRZEMIŃSKI, and JAKUB PROKOP
Dvořák, T., Pecharová, M., Krzemiński, W., and Prokop, J. 2019. New archaeorthopteran insects from the Carboniferous of Poland: Insights into tangled taxonomy. Acta Palaeontologica Polonica 64 (4): 787–796.
Archaeorthoptera is a high rank insect taxon comprising Orthoptera as well as the extinct orders Titanoptera and Caloneurodea, and several other late Paleozoic groups formerly assigned to polyphyletic Protorthoptera. Synapomorphies defining Archaeorthoptera and some fossil subordinate taxa are exclusively based on wing venation. This study presents a detailed description of two new archaeorthopteran genera and three new species from the Pennsylvanian of the Upper Silesian Coal Basin in Poland. These new taxa provide new insights into the wing venation disparity of this remarkable and insufficiently studied insect group. Omaliella polonica sp. nov. is based on a well preserved forewing, including the wing base, which allows a thorough discussion and comparison with other archaeorthopterans. Surprisingly, it is the first complete wing for this group of related genera (Omaliella, Omalia, Coselia and Paleomastax). Owadpteron dareki gen. et sp. nov. has an unusual arrangement of cubital veins. The marked resemblance of the venation of Owadpteron to that of some members of the gerarid line, such as Nacekomia, supports its placement within the family Geraridae (stem-group Orthoptera). Finally, the venation of Parapalaeomastax dariuszi gen. et sp. nov. strikingly resembles that of the genus Palaeomastax, differing only in the distally branched media. Discovery of these three new archaeorthopterans from the Upper Silesian Coal Basin fits well with that of closely related taxa known from other deposits in Euramerica, such as Mazon Creek Lagerstätte, Avion in Pas-de-Calais Basin and others. Furthermore, a new re-examination of the earliest archaeorthopteran from the Upper Silesian Coal Basin confirms doubtful assignment of this fragmentary fossil to Archaeorthoptera or even to Pterygota.
Key words: Insecta, Archaeorthoptera, Polyneoptera, wing venation, Pennsylvanian, Poland, Upper Silesia.
Tomáš Dvořák [algalesie@seznam.cz], Martina Pecharová [pecharom@natur.cuni.cz] and Jakub Prokop [jprokop@natur.cuni.cz] Department of Zoology, Faculty of Science, Charles University, Viničná 7, CZ-128 00, Praha 2, Czech Republic.
Wiesław Krzemiński [wieslawk4@gmail.com] Institute of Systematics and Evolution of Animals, Polish Academy of Sciences, ul. Sławkowska 17, 31-016, Kraków, Poland.
Received 5 March 2019, accepted 13 May 2019, available online 9 October 2019.
Copyright © 2019 T. Dvořák et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
The super-order Archaeorthoptera was established on the basis of its wing venation, with main apomorphies in the form of the complex fusions of the veins in medio-cubital area (Béthoux and Nel 2002, 2005). It consists of some taxa previously assigned to the polyphyletic group Protorthoptera listed by Carpenter (1992) and taxa assigned to the orders Orthoptera, Titanoptera, and Caloneurodea. However, the interrelationships within different groups of Archaeorthoptera remain poorly clarified. First, we can clearly separate the large group Panorthoptera (Béthoux and Nel 2002). While the assignments of some taxa to this group are still under debate, Panorthoptera consists of three main orders: Orthoptera, Caloneurodea, and Titanoptera, plus the gerarid line (Béthoux et al. 2012). The crucial diagnostic character of this group is the basal bifurcation of the CuPa vein into CuPaα and CuPaβ (Béthoux and Nel 2002). Aristov (2014) considered the placement of Geraridae among Gryllones and pointed out the resemblance of the venation to the family Cheliphlebiidae. However, the same author is not following the widely accepted concept of Archaeorthoptera and Panorthoptera (Béthoux and Nel 2002). Other established groups of Archaeorthoptera are Protophasmida (which is considered to be the most basal clade), Cnemidolestodea and Lobeattida (Béthoux 2005, Béthoux et al. 2012). However, there are many taxa that clearly belong to Archaeorthoptera, but can not be assigned to any of these high rank groups (Béthoux and Nel 2005, Béthoux 2008). Even the monophyly and validity of the above mentioned taxa is still under debate as well as the homology and nomenclature of the veins of Archaeorthoptera, unlike the situation in other groups of Pterygota (see Rasnitsyn 2007, Desutter-Grandcolas et al. 2017; Li et al. 2018). This complicates the fact that some recent studies of certain authors avoid to use binomen for species in comparative discussion and follow the ICZN rules only formally in supplementary parts of their articles (e.g., Béthoux 2008, Béthoux and Schneider 2010). This practice is confusing and makes future cross generic comparison with studies accepting traditional Linnaean hierarchical classification of organisms more difficult.
Herein, we provide descriptions of two new fossil genera and three new species based on the pattern of their forewing venation. The first new species is based on an exceptionally well preserved holotype, which reveals the fine structural details of this genus. At the same time, this complete fossil demonstrates problems with previously described species based on very fragmentary wings on which sometimes even the crucial characters of higher taxa are missing. All specimens used in this study came from two Pennsylvanian deposits: Leszczyny-Czerwionka nearby Knurów and Sosnowiec-Klimontów, both situated in Upper Silesia, Poland. Moreover, we provide re-evaluation to the earliest archaeorthopteran known from the Upper Silesian Coal Basin.
Institutional abbreviations.—FM, The Field Museum of Natural History, Chicago, USA; MGL, Musée Géologique de Lille, Lille, France; MNHN, Muséum national d’Histoire naturelle, Paris, France; MP ISEA, Natural History Museum of the Institute of Systematics and Evolution of Animals, Polish Academy of Science, Cracow, Poland; NHM, The Natural History Museum, London, UK; RBINS, Royal Belgian Institute of Natural Sciences, Brussels, Belgium.
Other abbreviations.—The wing venation concept and nomenclature follows that of Béthoux and Nel (2002) as established for Archaeorthoptera. 1A/2A, first/second anal vein; CuA/P, cubital anterior/posterior; CuPa/b, anterior/posterior branch of CuP; CuPaα/β cubital anterior/posterior branch of CuPa; M, stem of media; MA/P, medial anterior/posterior; RA/P, radial anterior/posterior; ScA/P, subcostal anterior/posterior.
Nomenclatural acts.—This published work and the nomenclatural acts it contains, have been registered in ZooBank: urn:lsid:zoobank.org:pub:758133BE-4660-40AF-A939-580B2F1D2E2B
Material and methods
The Carboniferous insects from localities in the Upper Silesian Coal Basin have been studied for more than a century (e.g., Roemer 1883; Handlirsch 1906). Most of the discoveries were linked with the intensive mining of coal in the past and only a few sites are currently accessible as spoil−heaps. One of them situated in the Polish part of the basin is Sosnowiec-Klimontów with material originally excavated from the Porąbka-Klimontów coal mine (e.g., Krawczyński et al. 1997). Fossils are preserved in sphero-sideritic concretions with 3D relief and include various groups of arthropods, molluscs, vertebrates and most commonly plant remains (Krawczyński et al. 2001, Stworzewicz et al. 2009, Pacyna and Zdebská 2010). The entomofauna consists predominantly of paoliids, palaeodictyopteran nymphs and their exuvia, followed by sparsely recorded other groups like Archaeognatha and Archaeorthoptera (Prokop et al. 2012, 2014, 2017, 2019).
The second locality Leszczyny-Czerwionka is situated west of Katowice and is especially well known for its fossil flora with a large accumulation of drifted long logs mainly attributable to Cordaites (Gradziński et al. 1982).
The fossils were studied in a dry state or under a film of ethyl alcohol using Leica MZ12.5 and Nikon SMZ 745 stereomicroscopes. Line drawings of the venation of both specimens were made directly with the aid of a camera lucida. Photographs were taken using a Canon D550 digital camera with EF 50mm and MP-65mm lenses. Original photographs were processed using the image-editing software Adobe Photoshop CS.
Systematic palaeontology
Super-order Archaeorthoptera Béthoux and Nel, 2002
Genus Omaliella Béthoux and Nel, 2005
Type species: Omaliella ramosa Béthoux and Nel, 2005, Pas-de-Calais Basin, France, Duckmantian (Westphalien B), Pennsylvanian.
Omaliella polonica sp. nov.
Fig. 1.
ZooBank LCID: urn:lsid:zoobank.org:pub:D0E08357-DADB-42EA-9E91-A75C0A4DE748
Etymology: From Latin Polonia, Poland.
Holotype: MP ISEA I−F/MP/8/1676/17 (imprint of forewing).
Type locality: Leszczyny-Czerwionka nearby Knurów, Rybnik, and Gliwice in Upper Silesia; found on the slag heaps that dates back to 19th century (Gradziński et al. 1982).
Type horizon: Carboniferous, Pensylvannian, Duckmantian (Westphalian B), Coal-bearing Mudstone Series, Orzesze Beds (Gradziński et al. 1982).
Diagnosis.—Based on fore-wing venation: width of costal area about midwing similar to subcostal area, ScP ending on anterior wing margin two thirds along wing length; RP partially connected with MA1 ending only with a terminal twig, anterior branch of RP distally fused with simple RA; CuA+CuPa extensively developed and posteriorly pectinate with 11 primary branches, basal branch distinctly undulated, CuPb simple.
Measurements.—Wing length 64.5 mm, maximum width 22.3 mm (about the level of division MA and MP).
Description.—Based on fore-wing venation. Nearly complete wing broadest approximately in the middle with originally hyaline membrane and numerous transverse or slightly oblique cross veins; ScA unknown because the basal anterior part of wing is not preserved; proximal part of ScP incomplete due to preservation; ScP reaches anterior wing margin in two thirds along the length of the wing, without discernible anterior branches; space between ScP and costal margin of wing quite narrow, about the same width as space between ScP and RA in the middle of the wing; R strongly convex, division of RA and RP 22 mm from wing base, RA simple, running parallel with ScP, terminally fused with anterior branch of RP very close to anterior wing margin; RP slightly concave and almost simple, partially connected to MA for 1.9 mm, ending with short terminal twig; stem of M shortly behind the wing base connected to CuA for 11.9 mm, stem of M concave divided into MA and MP in about midwing; MA deeply separated into MA1 and MA2 shortly behind the division with MP; MA1 shortly connected to RP; MA1 and MA2 both pectinate ending with 4 main posterior branches; third branch of MA1 secondarily pectinate anteriorly ending with three short branches; MP ending with three main posterior branches, first and third branch with secondary twigs; division of CuA and CuP very close to wing base, CuA+CuPa pectinate with 8 main posterior branches; area between CuPa and CuPb with irregular network of veinlets; CuPb and A1 both simple and closely parallel to one another.
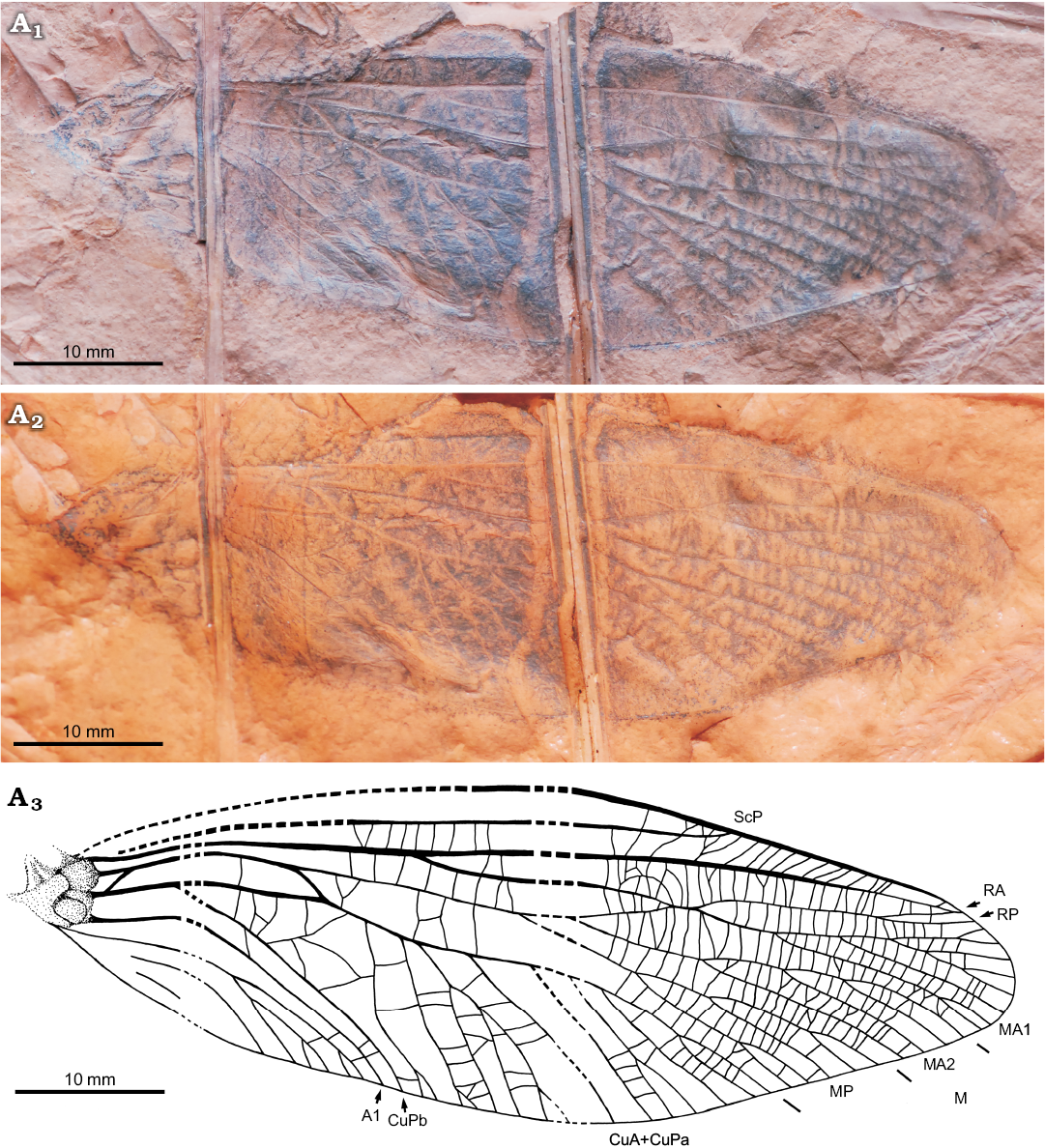
Fig. 1. Forewing venation of archaeorthopteran insect Omaliella polonica sp. nov., holotype (MP ISEA I−F/MP/8/1676/17), Carboniferous, Pensylvannian, Duckmantian (Westphalian B), Leszczyny-Czerwionka nearby the Knurów, Upper Silesia, Poland. Photograph in dry state (A1), under a film of ethanol (A2), and explanatory drawing (A3). Abbreviations: A1, first anal vein; CuA, cubital anterior; CuPa/b, anterior/posterior branch of cubital posterior; M, media; MA/P, media anterior/posterior; RA/P, radius anterior/posterior; ScP, subcosta posterior.
Remarks.—The present fossil can be assigned to the genus Omaliella based on the following combination of diagnostic forewing characters: RP long before it fuses with MA1, RP fused with MA1, CuA+CuPa posteriorly pectinate ending with about main 11 terminal branches. However, the type species of the genus Omaliella is based on the poorly preserved fossil of O. ramosa Béthoux and Nel, 2005, which does not have even the main apomorphies of the Archaeorthoptera. Therefore, it is necessary to discuss the assignment of Omaliella polonica sp. nov. and convincingly support the placement of Omaliella in the Archaeorthoptera.
We can assign Omaliella polonica sp. nov. to Archaeorthoptera sensu Béthoux and Nel, 2002 on the basis of the following apomorphies: basal division of CuP into CuPa and CuPb, fusion of M and CuA and fusion of distal part of CuA with CuPa. We can exclude its placement in clade Cnemidolestodea due to absence of anteriorly pectinate CuA+CuPa and the presence of a well-developed MP independent of CuA+CuPa (Béthoux 2005). Based on the non-differentiated vein CuPa (branches CuPaα and CuPaβ) the attribution to the clade Panorthoptera is also excluded (Béthoux and Nel 2002). We can also exclude the placement among lobeattid insects because of the more distal bifurcation of R with respect to the end of AA1 on posterior wing margin and quite wide space between RA and RP (Béthoux 2008). The apparently non-developed AP area also prompt us to exclude the assignment of O. polonica sp. nov. to Protophasmida neither (Béthoux 2003). Hence, O. polonica sp. nov. cannot be placed in any of the above mentioned higher clades of Archaeorthoptera.
Currently, there is no phylogenetic analysis of Archaeorthoptera that we could simply follow and therefore we avoid creating a new high level taxon. The placement of O. polonica sp. nov. is based on shared characters that could be indicative of the relationships of the closest taxa. Another problem is fragmentary preservation of some previously described taxa of Omaliella, which make it impossible to compare all the potentially important characters. Generally, we focused on the middle part of the forewing, which is usually well preserved in all the mentioned genera. So, we will compare and discuss these all significant characters of these relevant genera.
In the venation of O. polonica sp. nov. there is a prominent connection between RP and MA1 shared by for instance Geraridae (stem group of Panorthoptera) (Béthoux and Nel 2003) and some species of lobeattids (Béthoux 2005; Béthoux 2008; Béthoux et al. 2012). It also resembles the situation in Gerarus Scudder, 1885, especially Gerarus fisheri (Brongniart, 1885) (MNHN-LP-R.51139), as in both RP and MA1 are partially fused (instead of the much commoner well separated MA), RP, MA and MP have fewer branches and the region of CuA+CuPa is quite well developed. But Gerarus fisheri displays marked differences in venation as RA branched, connection between RP and MA is present only in some specimens, branching pattern of CuA+CuPa differs and furthermore CuPb with branches. Short fusion of RP and MA1 is also shared with Miamia Dana, 1864, of which the species M. maimai Béthoux, Gu, Yue, and Ren, 2012, is assigned to the lobeattids (Béthoux et al. 2012). But in this species there is only a weakly developed CuA+CuPa region and the pattern in the area CuA+M also differs.
Some other lobeattids have fussion of RP and MA instead of RA and MA1. Sinopteron with Sinopteron huangheense Prokop and Ren, 2007 has a posteriorly branched CuA+CuPa vein, and veins RP and MA are shortly connected by a short cross vein (Prokop and Ren 2007). Chenxiella with Chenxiella liuae shares also posteriorly branched CuA+CuPa vein, but MA is shortly connected to RP (Liu et al. 2009). However, the latter genus differs from O. polonica sp. nov. by a more basal division of RA and RP and richly branched RP. Longzhua with Longzhua loculata Gu, Béthoux, and Ren, 2011 has a short fusion of RP and MA and well developed CuA+CuPa region (Gu et al. 2011). On the other hand, R is branched more basally, RP is again richly branched and the forewing is distinctly smaller than that of Omaliella polonica sp. nov.
However, Omaliella polonica sp. nov. shares most traits with the following unassigned genera: Omalia Van Beneden and Coemans, 1867, Coselia Bolton, 1922, Paleomastax, and Omalliela. The main common difference is the connection of RP with MA instead MA1. Another difference in Omalia macroptera (RBINS a7687) is the non-regular branching pattern of CuA+CuPa. Omalia sp. (specimen MGL 4217) has a CuA+CuPa with a regular pattern of 5–6 main posterior branches. In this specimen we can also see a regular network of veinlets between CuA+CuPa and CuPb, which differs from that in O. polonica sp. nov. in which the irregular network of veinlets and most basal posterior branch of CuA+CuPa undulate. They are unfortunately not preserved in this specimen.
Due to incomplete preservation of the type specimens of Coselia and Paleomastax we do not know if MA is connected to RP. However, the general pattern of wing venation corresponds to that of Omalia and Omaliella polonica sp. nov. The preserved part of the wing of Coselia palmiformes (NHM I.15893) has a different branching pattern of CuA+CuPa and regular network of veinlets between CuA+CuPa. Palaeomastax carbonis Handlirsch, 1904 (RBINS a7700) has a similar pattern of regular posteriorly branching of veins CuA+CuPa, but the number of branches is unknown due to poor preservation.
The venation of Omalliela polonica sp. nov. shares numerous characteristics in the venation with the type species Omalliela ramosa Béthoux and Nel, 2005 known from Westphalian B of Pas-de-Calais Basin in France (Béthoux and Nel 2005). The vein RP is connected with MA1, area CuA+CuPa broadly developed and posteriorly pectinate with 10 main branches, unlike 11 branches found in O. polonica sp. nov., and simple CuPb. Unfortunately, we do not have any information on the branching patterns of R and M, but MP seems to be only weakly branched, as in O. polonica. Unfortunately, the basal part of forewing in O. ramosa is unknown. But the general course of the main veins in the type species strongly resembles that in O. polonica sp. nov., with the exception of the wider costal area, more basal connection of RP and MA1 and slightly different branching pattern of CuA+CuPa. On the basis of shared characters in their venations and general correspondence, we assign our new species to the genus Omalliela as O. polonica.
Stratigraphic and geographic range.—S Poland: Upper Silesia, Leszczyny-Czerwionka nearby Knurów; Pensylvannian, Duckmantian (Westphalian B).
Order Panorthoptera Crampton, 1928 (sensu Béthoux and Nel, 2002)
Family ?Geraridae Scudder, 1885
Genus Owadpteron nov.
ZooBank LCID: urn:lsid:zoobank.org:pub:38710148-8A61-4200-B0FC-42E0A6FAD893
Type species: Owadpteron dareki sp. nov.; by monotypy, see below.
Etymology: From Polish owad, insect and Greek pteron, wing; neuter in gender.
Diagnosis.—As for the type species by monotypy.
Owadpteron dareki sp. nov.
Fig. 2.
ZooBank LCID: urn:lsid:zoobank.org:pub:3BEC8E31-32AD-4601-8799-256FD9102B23
Etymology: In honour of our colleague and collector of Carboniferous fossils Dariusz Wojciechowski (Darek diminutive of Dariusz).
Holotype: MP ISEA I−F/MP/1488/26a,b/08 (print and counterimprint of forewing).
Type locality: Sosnowiec-Klimontów, originally Porąbka-Klimontów Mine, Upper Silesian Coal Basin, Poland (Krawczyński et al. 1997, 2001).
Type horizon: Langsettian (Westphalian A), Pensylvannian, Mudstone series (Załęże beds), Coal-bearing Mudstone Series (Pacyna and Zdebska 2012).
Diagnosis.—Based on fore-wing venation: broad costal area, RP ending with at least three branches, long stem of M+CuA; MP pectinate, each branch with secondary level branching; area between MP and CuA+CuPaα very broad at widest part and comparable to costal area; point connection of free part of CuA and CuPa; CuPa divides into branch CuPaα continuously attached to CuA and well separated from branch CuPaβ; CuPb anteriorly branched and ending in at least three branches.
Measurements.—Forewing fragment length 36.4 mm long, estimated forewing length 55 mm, minimum width at widest part 21.7 mm (about the level of contact between CuA and CuPa).
Description.—Based on fore-wing venation. Broad wing incompletely preserved and lacking basal part and apex. Wing membrane originally rather thick with two types of cross veins present. Regular transverse cross veins broadly spaced between main veins occur together with a dense network of fine veinlets. ScA unknown; ScP ending on RA in distal part of wing; broad costal space between anterior margin of wing and ScP markedly narrowing posteriorly, subcostal field regularly narrow; R divides into RA and RP slightly behind the division of M and CuA, RA nearly straight and probably simple, RP distally pectinate ending with at least three branches; common stem of M+CuA present, free part of MP anteriorly richly branched ending with three main branches all secondarily bifurcated, broad space between veins MP and CuA with several straight cross veins, in widest part as wide as costal space; convex CuA diverging from stem of M+CuA proximally from division of RA and RP, one terminal twig of CuA apparent; concave CuPa divides into branch CuPaα continuously attached to CuA and well separated from branch CuPaβ; CuPb anteriorly branched and ending with three branches; A1 simple.
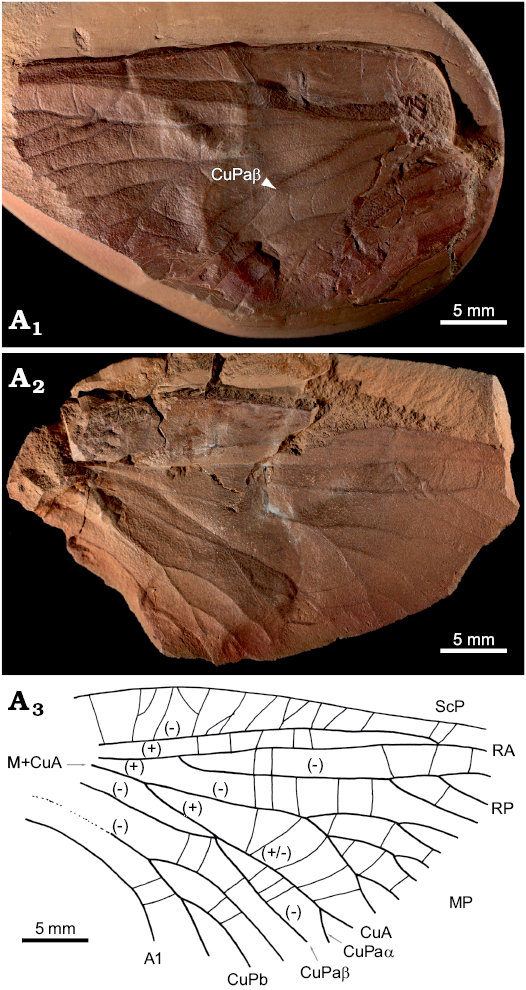
Fig. 2. Forewing venation of archaeorthopteran insect Owadpteron dareki sp. nov., holotype (MP ISEA I−F/MP/1488/26a,b/08), Carboniferous, Pensylvannian, Langsettian (Westphalian A), Sosnowiec-Klimontów, Upper Silesian Coal Basin, Poland. Photograph of imprint (A1) and counterimprint (A2), and explanatory drawing (A3). Abbreviations: (+), convex vein; (-), concave vein; A1, first anal vein; CuA, cubital anterior; CuPa/b, anterior/posterior branch of cubital posterior; CuPaα/β, cubital anterior/posterior branch of CuPa; M, media; MP, media posterior; RA/P, radius anterior/posterior; ScP, subcosta posterior.
Remarks.—It is not easy to classify this wing due to incomplete preservation and especially the missing wing base. Presence of prominent convex veinal stem located posterior of the apparent strongly convex R, which is further divided into a weakly concave MP and convex CuA, which we consider to be the common stem of M+CuA. The presence of M+CuA is considered as one of the key apomorphies of the Archaeorhoptera (Béthoux and Nel 2002). Another important Archaeorthoptera apomorphy is the basal division of CuP into the branches CuPa and CuPb, which is also present on this wing. However, the free part of CuA does not fuse with CuPa and there is only one point connection between these two veins. This raises the question: was this situation an ancestral state for Archaeorthoptera before the complete fusion of these two veins occurred?
Due to the more distal bifurcation of R with respect to the end of AA1 on posterior wing margin and the presence wide space between branches of RA and RP, the placement to lobeattid taxa is unlikely (Béthoux 2008). The wing venation similarly lacks the typical character of Cnemidolestodea, in which there is an anteriorly pectinate CuA+CuPa (Béthoux 2005). We have no information on the area of AP, which is well developed in Protophasmida (Bethoux 2003), and the general venation in the preserved part differs considerably. So it cannot be placed in Protophasmida. There is also a weak correspondence of the venation with that of the genera Omalia, Omaliella, Palomastax and Coselia. Potential attribution to the clade Panorthoptera is discussed bellow.
However, the pattern in the venation of our wing resembles that in the monotypic Ampeliptera, with A. limburgica described from Namurian A in The Netherlands (Pruvost 1927). On both wings there is a dense network of veinlets, posteriorly pectinate RP and anteriorly pectinate MP with bifurcated branches. Even the short fusion of the veins CuA and CuPa is present in both taxa. However, this fusion in Ampeliptera is a bit longer. Branched CuPb is also present in both species. Main differences are a narrower space between anterior margin of wing and ScP, and wider space between RA and RP in Ampeliptera. The branching pattern of MP is also slightly different. But the missing distal part of wing poses a problem, we do not know the exact branching pattern. Same problem concerns the branching of CuA, but there are presumably fewer branches in Owadpteron. Systematic position of Ampeliptera limburgica has been debated for long time. Béthoux and Nel (2002), after examining of the holotype, placed A. limburgica into the Archaeorthoptera and considered it to be the oldest known member of this clade at that time. This supports our idea about the antiquity of Owadpteron based on an incompletely fused CuA and CuPa.
The venation does resemble that of Nacekomia rossae described from the Pennsylvanian strata at Mazon Creek, Ilinois (Richardson 1956), which is considered to be a member of the family Geraridae (Kukalová-Peck and Brauckmann 1992; Béthoux and Nel 2002). Both taxa share the same organisation of the cubital area, with CuA ending in only a few branches and a similar shaped CuPb with two anterior branches. The vein CuPa is fused with a free CuA over a short distance in Nacekomia (see Fig. 3), whereas in Owadpteron it is a point fusion. Nevertheless, this course of the CuPa vein is generally rare in Archaeorthoptera, so we can consider this character to be relatively stable in both taxa. Another resemblance between the venation of both taxa is the wide costal field and sparse but quite regular network of simple thin cross veins. On the other hand, there are some differences, such as ScP ends on RA instead of on the anterior margin of the wing in N. rossae and there is a more distal division of the veins RA and RP. The apparent resemblance of the venation to that of Nacekomia and the presence of several differences, lead us to assign our fossil to a new genus placed presumably in Geraridae (Panorthoptera). However, the exact taxonomic placement of N. rossae is under discussion because of the indistinct differentiation of CuPa into CuPaα and CuPaβ, which is a key apomorphy of Panorthoptera comprised of Geraridae (Béthoux and Nel 2002). Our re-examination of the type of N. rossae confirmed that the branches of CuPaα and CuPaβ are markedly concave, which justifies its placement in Panorthoptera (see Fig. 3). The bifurcation of CuPa is not clearly discernible in Owadpteron. However, the markedly different polarity of CuA from convex to neutral at its point of contact with CuPa support our notion that CuA continues as an anterior branch of CuPaα, while CuPaβ diverges separately. An interpretation that would confirm the placement of Owadpteron in Panorthoptera and most probably close to Nacekomia (Geraridae). Nevertheless, we need a more complete specimen of Owadpteron in order to clarify its taxonomic status.
Stratigraphic and geographic range.—S Poland: Upper Silesia, Sosnowiec-Klimontów; Pensylvannian, Langsettian (Westphalian A).

Fig. 3. Forewing venation of archaeorthopteran insect Nacekomia rossae Richardson, 1956, holotype (FM PE791), Pensylvannian, Moscovian (Westphalian D), Mazon Creek Lagerstätte, Ilinois, USA.
Genus Parapalaeomastax nov.
ZooBank LCID: urn:lsid:zoobank.org:pub:13BC1408-34E6-4553-8714-B7D64A718552
Type species: Parapalaeomastax dariuszi sp. nov.; by monotypy, see below.
Etymology: Named after its similarities in venation to the extinct Palaeomastax Handlirsch, 1904; feminine in gender.
Diagnosis.—As for the type species by monotypy.
Parapalaeomastax dariuszi sp. nov.
Fig. 4.
ZooBank LCID: urn:lsid:zoobank.org:pub:08E34AA1-C66D-4FAF-A4C1-49F61D87A63B
Etymology: In honour of our colleague and collector of Carboniferous fossils Dariusz Wojciechowski.
Holotype: MP ISEA I−F/MP/1540/20/09 (imprint of forewing).
Type locality: Sosnowiec-Klimontów, originally Porąbka-Klimontów Mine, Upper Silesian Coal Basin, Poland (Krawczyński et al. 1997, 2001).
Type horizon: Pensylvannian, Langsettian (Westphalian A), Coal-bearing Mudstone Series, Mudstone series (Załęże beds) (Pacyna and Zdebska 2012).
Diagnosis.—Vein M distally divided into MA and MP, MA shortly braced to RP. Free part of M before bifurcation four times or less longer than width of costal field where CuA+M divides.
Measurements.—Forewing fragment length 34.4 mm long, estimated forewing length 55 mm, maximum width in widest preserved part 14 mm (estimated maximum width is probably only slightly larger).
Description.—Based on fore-wing venation. Wing generally incompletely preserved, cubital area partly distorted, apex missing. Wing membrane hyaline, numerous transverse or slightly oblique crossveins regularly arranged. ScA unknown, ScP ends on costal margin in distal third of wing. Costal area broad with regular pattern of numerous oblique veinlets. R almost straight, bifurcated 6.18 mm distal from divergence of M and CuA. RA probably simple. RP posteriorly pectinate, ending with at least three terminal branches. Short stout brace present between RP and MA. Long basal fusion between M+CuA. M diverging from the common stem well before the middle of the wing. M divided into MA and MP beyond the middle of the wing. Short brace between MA and RP 2.3 mm from M bifurcation. MA and MP run parallel to one another over a long distance. Distal bifurcation of both M veins present close to posterior margin of wing. CuA fused with CuPa shortly after its emergence from the M+CuA vein. CuPb probably simple. Details of cubital area indistinct, but posterior pectination of CuA+CuPa ending with at least 4 primary branches, the first of which is secondarily bifurcated. Anal area formed by at least two anal veins, second anal vein distally bifurcated.
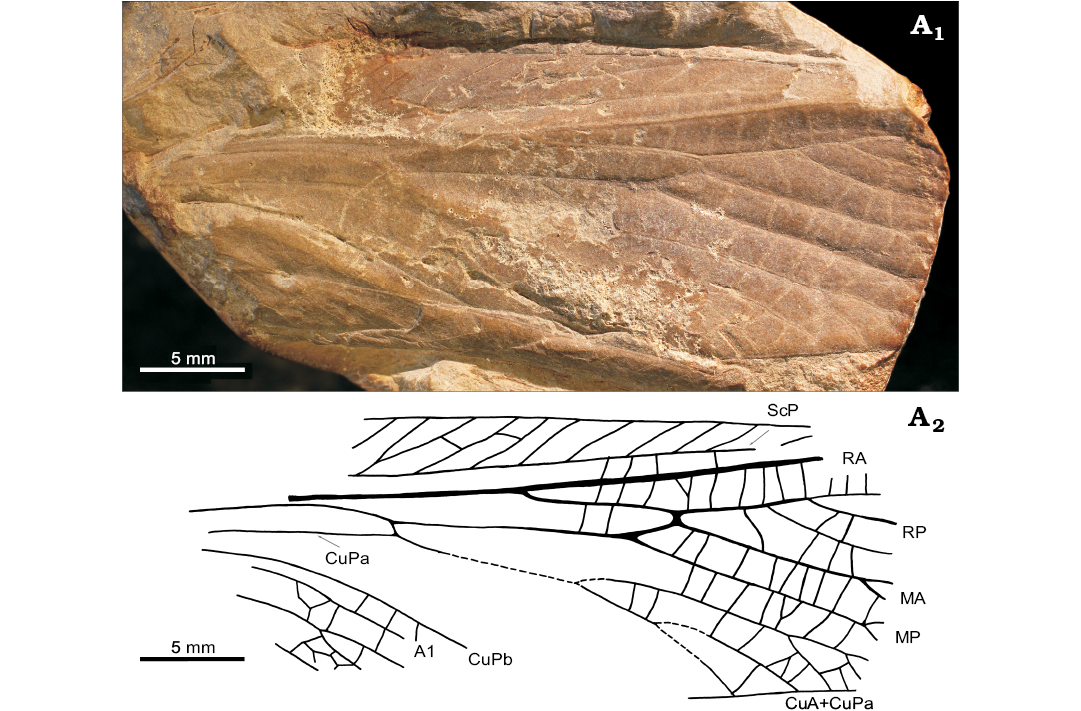
Fig. 4. Forewing venation of archaeorthopteran insect Parapalaeomastax dariuszi gen. et sp. nov., holotype (MP ISEA ISEA I−F/MP/1540/20/09), Carboniferous, Pensylvannian, Langsettian (Westphalian A), Sosnowiec-Klimontów, Upper Silesian Coal Basin, Poland. Photograph (A1) and explanatory drawing (A2). Abbreviations: A1, first anal vein; CuA, cubital anterior; CuPa/b, anterior/posterior branch of cubital posterior; MA/P, media anterior/posterior; RA/P, radius anterior/posterior; ScP, subcosta posterior.
Remarks.—This forewing corresponds to that of Archaeorthoptera sensu Béthoux and Nel (2002) because of the following synapomorphies: CuP differentiated into two branches (CuPa and CuPb), M and CuA fused over long distance and distal part of CuA fused with CuPa. Despite a poorly preserved cubital area, there is no sign of a branched CuPa vein, so it is unlikely that it can be placed in Panorthoptera. Wide space between RA and RP lead us to exclude this fossil from Lobeattida. Due to posteriorly pectinate CuA+CuPa vein, we can also exclude the placement within Cnemidolestodea (Béthoux 2005). AP area is indistinct, but is probably not well developed, and therefore our fossil cannot be placed in Protophasmida. Nevertheless, the general pattern of the venation of this fossil resembles that of the group of genera consisting of Coselia, Omaliella, Paralongzhua, Omalia, and especially Palaeomastax (Béthoux and Nel 2005; Prokop et al. 2015).
Parapalaeomastax gen. nov. differs from Omalia with Omalia macroptera, especially by its more distal division of MA and MP and connection of MA by only a short brace to RP, instead of a partial fusion (Béthoux and Nel 2005). There is also a more complicated pattern of cross veins in the costal area in O. macroptera and the same is true for Omalia sp. (specimen MGL 4217), where in addition, the bifurcation of MP is closer to the division of MA and MP (Béthoux and Nel 2005). Coselia mainly differs from Parapalaeomastax in having a markedly wider costal area with a prominent meshwork of cross veins, but the type species of C. palmiformis is based only on the basal part of a wing. In Omaliella with Omaliella ramosa the branching pattern of M is different and vein RP is connected to the anterior branch MA1. The comparison of Parapalaeomastax with early Permian Paralongzhua based on Paralongzhua elongata from Lodève (Hérault, France) reveals it has a much narrower costal area, the division of MA and MP located more basally and MA is deeply bifurcated (Prokop et al. 2015). Palaeomastax Handlirsch, 1904 based on Palaeomastax carbonis from Frameries in Belgium has the same general pattern of venation, with the exception of a long and simple M unlike our fossil with deeply branched M and MA shortly braced to R (Béthoux and Nel 2005). But the fossil of P. carbonis lacks distal part of wing and even the distal end of the preserved part is damaged. It is quite plausible, that a bifurcation of M and a connection of MA with RP can occur immediately distal to the preserved area. However, we have to stick to the facts. So, the long simple M is a crucial diagnostic character of the genus Palaeomastax and therefore provides sufficient differentiation of Parapalaeomastax, but we need to search for more complete fossils of Palaeomastax in order to accurately compare their wing apices.
Stratigraphic and geographic range.—S Poland: Upper Silesia, Sosnowiec-Klimontów; Pensylvannian, Langsettian (Westphalian A).
Re-evaluation of the earliest archaeorthopteran from the Upper Silesian Coal Basin
Prokop et al. (2005) reported the anterobasal part of wing attributable to Archaeorthoptera from the lowermost Namurian A/E1 (ca. 324 Ma) of Ostrava Formation in Upper Silesian Coal Basin (Czech Republic). Due to a poor and fragmentary preservation of this fossil the authors were unable to provide any formal attribution to a precise group or closer comparison at that time. However, this fossil potentially represents an important calibration point as it is at the same time the oldest Pterygota. Wolfe et al. (2016) regarded this fragmentary fossil as insufficiently characterized for dating. Our re-examination of this fossil specimen (No. B13711) reveals several obstacles as supposed longitudinal veins are interrupted and distinctly stronger in distal part, branching pattern of the main veins near the supposed wing base seems to be very unusual without clearly discernable veins ScP and R (including different polarity), and thus support uncertainties of the assignment. Therefore, we considered the placement of this fossil among Archaeorthoptera or even Pterygota as doubtful. Our new observation reveals that it could rather belong to another group of organisms (presumably a fragment of fish fin).
Conclusions
The Carboniferous insects from the Upper Silesian Coal Basin have been well studied for more than a century by the classic authors Ferdinand von Roemer, Anton Handlirsch and Pierre Pruvost. Recently discovered localities in the Polish part of the Basin at Czerwionka and particularly Sosnowiec with exceptionally well preserved specimens in sideritic nodules reveal a diverse entomofauna with a high potential for future studies.
This study focused on members of Archaeorthoptera and includes descriptions of two new genera and three new species based on fore-wing venation. A remarkably well preserved forewing of Omaliella polonica sp. nov. reveals structural details of the venation, which are unknown for the type species Omaliella ramosa due to its fragmentary preservation. In particular, this species allows us to study the venation near the wing base and apex for the first time.
Owadpteron dareki gen. et sp. nov. has an unusual trait, CuPaα is partially fused with CuA. Our hypothesis is supported by the examination of Nacekomia rossae, which has a similar venation, with the free part of CuPaα distinctly emerging from CuA. This pattern supports the assignment of Owadpteron to the Geraridae and Panorthoptera. However, the bifurcation of CuPa is an essential apomorphy of Panorthoptera and its absence in O. dareki would greatly weaken our taxonomic placement, or challenge the significance of this character. Parapaleomastax dariuszi gen. et sp. nov. is poorly preserved, but important apomorphies are visible and allow the specific assignment within Archaeorthoptera. The comparison with the genus Palaeomastax is complicated by the lack of a preserved wing apex with the crucial diagnostic characters.
All three new species of Archaeorthoptera differ greatly morphologically and in their wing venation, which could help in a future phylogenetic analysis of this stunning group.
Acknowledgements
We cordially thank Dariusz Wojciechowski (Katowice, Poland) for his effort and help with the fieldwork at Sosnowiec. The authors are grateful to Paul Mayer (The Field Museum, Chicago, USA), who kindly provided access to the collection under his care. We thank to André Nel (Muséum national d’Histoire naturelle, Paris, France) and anonymous reviewer for their insightful comments to early version of the manuscript. Authors are grateful Anthony F.G. Dixon (University of East Anglia, Norwich, United Kingdom) for improving the English and Tomáš Přikryl (Institute of Geology of the Czech Academy of Sciences, Praha) for valuable discussion on taphonomy. JP and MP were supported by a research project of the GAČR (No. 18-03118S).
References
Aristov, D.S. 2014. Classification of the order Cnemidolestida (Insecta: Perlidea) with descriptions of new taxa. Far Eastern Entomologist 277: 1–46.
Béthoux, O. 2003. Protophasma dumasii, a link between Orthoptera and the dictyopterid orders? Journal of Orthoptera Research 12: 57–62. Crossref
Béthoux, O. 2005. Cnemidolestodea (Insecta): an ancient order reinstated. Journal of Systematic Palaeontology 3: 403–408. Crossref
Béthoux, O. 2006. Revision of Cacurgus Handlirsch, 1911, a basal Pennsylvanian Archaeorthoptera (Insecta: Neoptera). Bulletin of the Peabody Museum of Natural History 47: 29–35. Crossref
Béthoux, O. 2008. Revision and phylogenetic affinities of the lobeattid species bronsoni Dana, 1864 and silvatica Laurentiaux & Laurentiaux-Vieira, 1980 (Pennsylvanian, Archaeorthoptera). Arthropods Systematics & Phylogeny 66: 145–163.
Béthoux, O. and Nel, A. 2002. Venation pattern and revision of Orthoptera sensu nov. and sister groups. Phylogeny of Palaeozoic and Mesozoic Orthoptera sensu nov. Zootaxa 96: 1–88. Crossref
Béthoux, O. and Nel, A. 2003. Wing venation morphology and variability of Gerarus fischeri (Brongniart, 1885) sensu Burnham (Panorthoptera; Upper Carboniferous, Commentry, France), with inferences on flight performance. Organisms Diversity and Evolution 3: 173–183. Crossref
Béthoux, O. and Nel, A. 2005. Some Palaeozoic Protorthoptera are ancestral Orthopteroids: major wing braces as clues to a new split among the Protorthoptera (Insecta). Journal of Systematic Palaeontology 2: 285–309. Crossref
Béthoux, O. and Schneider, J.W. 2010. Description of a hind wing of a new basal Archaeorthoptera (Mazon Creek, IL; Pennsylvanian). Alavesia 3: 81–85.
Béthoux, O., Gu, J.-J., and Ren, D. 2012. A new Upper Carboniferous stem-orthopteran (Insecta) from Ningxia (China). Insect Science 19: 153–158. Crossref
Béthoux, O., Gu, J.-J., Yue, Y., and Ren, D. 2012. Miamia maimai n. sp., a new Pennsylvanian stem-orthopteran insect, and a case study on the application of cladotypic nomenclature. Fossil Record 15: 103–113. Crossref
Brauckmann, C. 1984. Weitere neue Insekten (Paleodictyoptera: Protoorthoptera) aus Namurium B von HagenVorhalle. Jahresberichte des Naturwissenschaftlichen Vereins in Wuppertal 37: 108–115.
Carpenter, F.M. 1992. Superclass Hexapoda. In: R.C. Moore and R.L. Kaesler (eds.), Treatise on Invertebrate Paleontology, Part R, Arthropoda 4, 3/4, 1–655. The Geological Society of America, Boulder and the University of Kansas, Lawerence.
Desutter-Grandcolas, L., Jacquelin, L., Hugel, S., Boistel, R., Garrouste, R., Henrotay, M., Warren, B.H., Chintauan-Marquier, I.C., Nel, P., Grandcolas, P., and Nel, A. 2017. 3-D imaging reveals four extraordinary cases of convergent evolution of acoustic communication in crickets and allies (Insecta). Scientific Reports 7: 7099. Crossref
Gradziński, R., Doktor, M., and Brzyski B. 1982. Accumulation of drifted logs and other large plant debris in a Carboniferous fluvial channel at Czerwionka, Upper Silesia. Acta Geologica Polonica 32: 69–81.
Gu, J.-J., Béthoux, O., and Ren, D. 2011. Longzhua loculata n. gen. n. sp., one of the most completely documented Pennsylvanian Archaeorthoptera (Insecta; Ningxia, China). Journal of Palaeontology 85: 303–314. Crossref
Handlirsch, A. 1906–1908. Die fossilen Insekten und die Phylogenie der rezenten Formen. Ein Handbuch für Paläontologen und Zoologen. 1430 pp. V.W. Engelman, Leipzig.
Krawczyński, W., Filipiak, P., and Gwoździewicz, M. 1997. Zespoł skamieniałości z karbońskich sferosyderitów (westfal A) NE części Górno−śląskiego Zagłębia Węglowego. Przegląd Geologiczny 45: 1271–1274.
Krawczyński,W., Filipiak, P., and Wojciechowski, D. 2001. Owady z warstw załęskich (westfal A) NE części Górnośląskiego Zagłębia Węglowego. In: XXI Terenowa Szkoła Geologów Uniwersytetu Śląskiego, 25–28. Uniwersytet Śląski, Sosnowiec.
Kukalová-Peck, J. and Brauckmann, C. 1992. Most Paleozoic Protorthoptera are ancestral hemipteroids: major wing braces as clues to a new phylogeny of Neoptera (Insecta). Canadian Journal of Zoology 70: 2452–2473. Crossref
Li, X.R., Zheng, Y.H., Wang, C.C., and Wang, Z.Q. 2018. Old method not old-fashioned: parallelism between wing venation and wing-pad tracheation of cockroaches and a revision of terminology. Zoomorphology 137: 519–533. Crossref
Liu, Yu., Ren, D., and Prokop, J. 2009. Discovery of a new Namurian archaeorthopterid from Ningxia, China (Insecta: Archaeorthoptera). Zootaxa 2032: 63–68. Crossref
Pacyna, G. and Zdebska, D. 2010. Upper Carboniferous seed fern (Pteridospermophyta) pollen organs from Silesia (Poland) and related evolution considerations. Monographiae Botanicae 100: 1–81. Crossref
Prokop, J. and Ren, D. 2007. New significant fossil insects from the Upper Carboniferous of Ningxia in northern China (Palaeodictyoptera, Archaeoptera). European Journal of Entomology 104: 267–275. Crossref
Prokop, J., Krzemińska, E., Krzemiński, W., Rosová, K., Pecharová, M., Nel, A., and Engel, M.S. 2019. Ecomorphological diversification of the Late Palaeozoic Palaeodictyopterida reveals different larval strategies and amphibious lifestyle in adults. Royal Society Open Science 6: 190460. Crossref
Prokop, J., Krzemiński, W., Krzemińska, E., and Wojciechowski, D. 2012. Paoliida, a putative stem-group of winged insects: Morphology of new taxa from the Upper Carboniferous of Poland. Acta Palaeontologica Polonica 57: 161–173. Crossref
Prokop, J., Krzemiński, W., Krzemińska, E., Hörnschemeyer, T., Ilger, J.M., Brauckmann C., Grandcolas, P., and Nel, A. 2014. Late Palaeozoic Paoliida is the sister group of Dictyoptera (Insecta: Neoptera). Journal of Systematic Palaeontology 12: 601–622. Crossref
Prokop, J., Nel, A., and Hoch, I. 2005. Discovery of the oldest known Pterygota in the Lower Carboniferous of the Upper Silesian Basin in the Czech Republic (Insecta: Archaeorthoptera). Geobios 38: 383–387. Crossref
Prokop, J., Pecharová, M., Nel, A., Hörnschemeyer, T., Krzemińska, E., Krzemiński, W., and Engel, M.S. 2017. Paleozoic nymphal wing pads support dual model of insect wing origins. Current Biology 27: 263–269. Crossref
Prokop, J., Szwedo, J., Lapeyrie, J., Garrouste, R., and Nel, A. 2015. New middle Permian insects from Salagou Formation of the Lodève Basin in southern France (Insecta: Pterygota). Annales de la Société Entomologique de France 51: 14–51. Crossref
Pruvost, P. 1927. Sur une aile d’insecte trouvée au sondage de Gulpen. Jaarvestag van voor het Nederlandsche Mijngebied te Heerlen 1926: 76–77.
Rasnitsyn, A.P. 2007. On the discussion of the wing venation of (Archae) Orthoptera (Insecta). Paleontological Journal 41: 341–344. Crossref
Richardson, E.S. Jr. 1956. Pennsylvanian invertebrates of the Mazon Creek area, Illinois. Insects. Fieldiana, Geology 12: 15–56. Crossref
Roemer, F. 1883. Über einen im Schieferthon der zwischen Königshütte und Laurahütte gelegenen Alfredgrube, 10 m im Liegenden des Carolinen-flözes, gefundenen Insectenflügel. Jahresbericht der Schlesischen Gesell-schaft für Vaterländische Kultur 62: 226.
Stworzewicz, E., Szulc, J., and Pokryszko, B.M. 2009. Late Paleozoic continental gastropods from Poland: Systematic, evolutionary and paleoecological approach. Journal of Paleontology 83: 938–945. Crossref
Wolfe, J.M., Daley, A.C., Legg, D.A., and Edgecombe, G.D. 2016. Fossil calibrations for the arthropod tree of life. Earth Science Reviews 160: 43–110. Crossref
Acta Palaeontol. Pol. 64 (4): 787–796, 2019
https://doi.org/10.4202/app.00614.2019