

Morphological homology, evolution, and proposed nomenclature for bear dentition
QIGAO JIANGZUO, JINYI LIU, and JIN CHEN
Jiangzuo, Q., Liu, J., and Chen, J. 2019. Morphological homology, evolution, and proposed nomenclature for bear dentition. Acta Palaeontologica Polonica 64 (4): 693–710.
Bears are a group of carnivores with diverse diets and complicated dental structure. Several large rearrangements of dental structures are known in different lineages of bears, making the homology of dental structures between the different bears difficult to evaluate. By tracing the evolutionary processes of the two lineages of bears with the most complicated dental structure, i.e., the giant panda lineage (Ailuropodinae) and cave bear lineage (Ursinae), we were able to clarify the homology of dental structures of the two subfamilies. We define a new assemblage of dental nomenclature (based mainly on the homology to the giant panda) that can be very useful to infer the evolution of fossil bears. The evolutionary positions of some fossil bears are reviewed based on our results.
Key words: Mammalia, Ailuropodinae, Ursinae, dentition, homology, Pleistocene, China.
Qigao Jiangzuo [jiangzuo@ivpp.ac.cn] (corresponding author), Key Laboratory of Vertebrate Evolution and Human Origins of Chinese Academy of Sciences; Institute of Vertebrate Paleontology and Paleoanthropology, Chinese Academy of Sciences and Center for Excellence in Life and Paleoenvironment, Beijing, 100044, China; University of Chinese Academy of Sciences, Beijing, 100049, China; Division of Paleontology, American Museum of Natural History, New York, 10024, USA.
Jinyi Liu [liujinyi@ivpp.ac.cn] (corresponding author), Key Laboratory of Vertebrate Evolution and Human Origins of Chinese Academy of Sciences; Institute of Vertebrate Paleontology and Paleoanthropology, Chinese Academy of Sciences and Center for Excellence in Life and Paleoenvironment, Beijing, 100044, China.
Jin Chen [chenjin@ivpp.ac.cn], Key Laboratory of Vertebrate Evolution and Human Origins of Chinese Academy of Sciences; Institute of Vertebrate Paleontology and Paleoanthropology, Chinese Academy of Sciences, Beijing, 100044, China.
Received 28 April 2019, accepted 26 June 2019, available online 30 October 2019.
Copyright © 2019 Q. Jiangzuo et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
Bears are a group of carnivores with diverse diets, in which several lineages evolved towards the herbivorous adaptation; among those the extremes are the giant panda and the cave bear (Erdbrink 1953; Bocherens 2015). The giant panda (Ailuropoda melanoleuca) is known for peculiar morphological characters adapted to the bamboo diet, e.g., the heavy skull, strong sagittal crest and the greatly enlarged paranasal sinuses for large bite force (Sicher 1944; Davis 1964), an enlarged radial sesamoid functioning as a false thumb for grasping (Davis 1964; Endo et al. 1999; Endo 2001) and most importantly, the enlarged and widened teeth with strong tubercle-like cusps and deep valleys in the occlusal surface for grinding (Gregory 1936; Davis 1964). These dental adaptations appeared early in Ailuropodini (Thenius 1979; Qiu and Qi 1989; Abella et al. 2011, 2012), but how these complicated dental characters evolved gradually from the early representatives (Kretzoiarctos, Agriarctos, and Ailurarctos) to the living Ailuropoda has never been studied in detail. Therefore, the origin and homology of the additional cusps or ridges in Ailuropoda remain unclear. Some attempts have been made to interpret these cusps/tubercles, e.g., Huang (1993), but none of them analyzed the fine dental structures from the evolutionary aspect. Until now, there is still no consensus about the terminology of cusps and ridges in Ailuropoda teeth.
In this work, we compare the general dental morphology and trace every stable cusp and ridge of molars and P4/p4 of giant panda and their counterparts (if present) in cave bears (Ursus spelaeus, Ursus deningeri, and related species, with a special focus on U. deningeri in this study, since a large number of specimens are available to authors, and there is not much innovation from Ursus deningeri to the slightly more derived cave bears such as Ursus spelaeus) (Fig. 1). Dental characters of Ailuropoda are redefined. Cave bears are also well known for their complicated dental occlusal surface (Torress Perezhidalgo 1992; Baryshnikov 1998; Rabeder 1999; Wagner and Čermák 2012), and this has been interpreted as an adaptation to plant diet, as confirmed by analysis of carbon and nitrogen stable isotopes (Bocherens et al. 2011; Bocherens 2015) as well as cranium morphology (Van Heteren et al. 2018). Whether these shared characters are convergent or homologous is evaluated by tracing these characters through the evolution of both lineages.
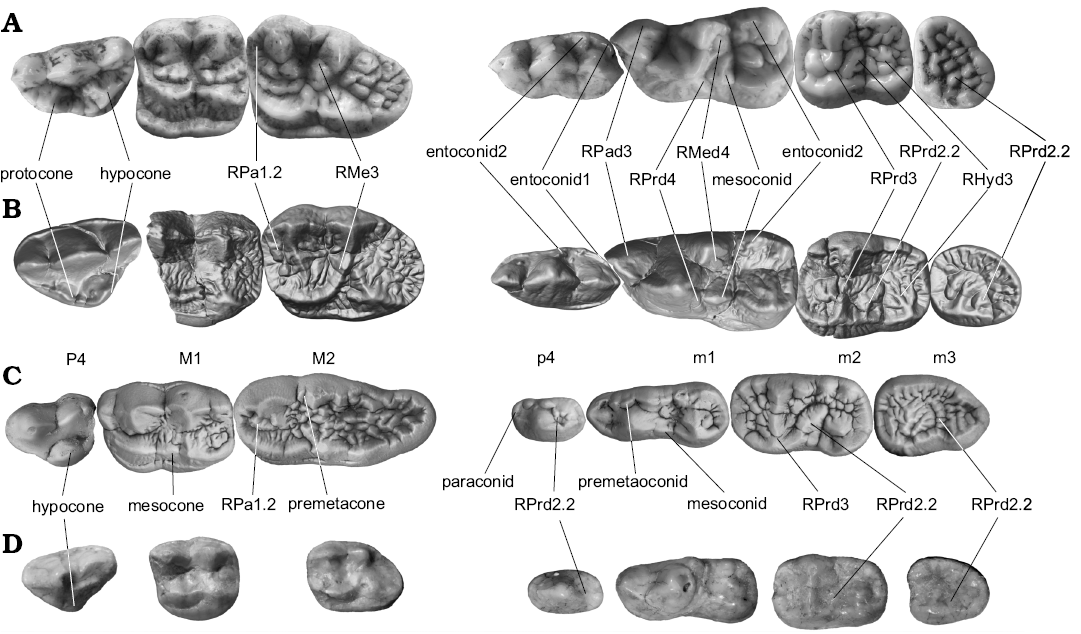
Fig. 1. Occlusal views of cheek teeth of studied bears. A. Ailuropoda sp. from Pleistocene of China. B. Ailurarctos lufengensis Qiu and Qi, 1989 (m1, IVPP V25032 and others IVPP V6892, reconstructed from CT scanning) from Lufeng, Yunnan Province, Latest Miocene. C. Ursus deningeri von Reichenau, 1904 (NMM1946/643, 1953/119, 1953/119, 1955/818, 1956/907, 1953/54, 1956/909; photo Jan Wagner) from Mosbach 2 locality, Germany, early Middle Pleistocene. D. Ursavus tedfordi Qiu, Deng, and Wang, 2014 (HMV1453) from Huaigou, Gansu Province, Late Miocene. For terminology and more details for dental character see Fig. 2. Not to scale. Abbreviations: RHyd3, medial ridge of protoconid; RMe3, medial ridge of metacone; RMed4, postero-medial ridge of metaconid; RPa1.2, medial branch of anterior ridge of paracone; RPad3, medial ridge of paraconid; RPrd2.2, medial branch of the posterior ridge of protoconid; RPrd3, postero-medial ridge of protoconid; RPrd4, postero-lateral ridge of protoconid.
Institutional abbreviations.—AMNH, American Museum of Natural History, New York, USA; HMV, Hezheng fossil Museum, Gansu, China; IOZ, Institute of Zoology, Chinese Academy of Sciences, Beijing, China; IVPP, Institute of Vertebrate Paleontology and Paleoanthropology, Chinese Academy of Sciences, Beijing, China (V, fossil specimens; OV, living specimens; RV, recataloged old specimens); MNHN, Muséum national d’Histoire naturelle, Paris, France; NMM, Naturhistorisches Museum Mainz, Mainz, Germany; NMP, National Museum, Prague, Czech Republic.
Other abbreviations.—We follow standard convention in abbreviating tooth families as I, C, P, and M, with upper and lower case letters referring to upper and lower teeth, respectively. Dental ridge homologous structures: REnd3, medial ridge of entoconid; RHyd1, anterior ridge of protoconid; RHyd2, posterior ridge of protoconid; RHyd3, medial ridge of protoconid; RMe1/RMed1, anterior ridge of metacone/metaconid; RMe2/RMed2, posterior ridge of metacone/metaconid; RMe3/RMed3, medial ridge of metacone/metaconid; RMed4, postero-medial ridge of metaconid; RPa1/RPad1, anterior ridge of paracone/paraconid; RPa1.2, medial branch of anterior ridge of paracone; RPa2/RPad2, posterior ridge of paracone/paraconid; RPa3/RPad3, medial ridge of paracone/paraconid; RPrd1, anterior ridge of protoconid; RPrd2, posterior ridge of protoconid; RPrd2.2, medial branch of the posterior ridge of protoconid; RPrd3, postero-medial ridge of protoconid; RPrd4, postero-lateral ridge of protoconid.
Material and methods
Material of both living and extinct species of giant panda (Ailuropoda melanoleuca, Ailuropoda wulingshanensis, Ailuropoda microta), primitive panda Ailurarctos lufengensis and cast of Ailurarctos yuanmouensis housed in IVPP and IOZ, cave bear Ursus deningeri housed in NMP and NMM, Miocene bear Ursavus tedfordi and Indarctos spp. housed in HMV and AMNH were analysed in this study. Material of living species of Ursus, i.e., Ursus arctos, Ursus maritimus, Ursus americanus, Ursus thibetanus, Ursus malayanus, and Ursus ursinus, living species of musteloids Meles leucurus and Procyon lotor housed in IVPP and AMNH, and fossil specimens of Ursus etruscus orientalis and Indarctos spp. housed in IVPP and Dalian Natural History Museum (Dalian, Liaoning Province, China) were also used for comparison. Other species compared in this study were based on literature and/or photos.
The dental morphology of Ailuropoda and cave bears is described and a brief summary is listed at the end of the description of each character with the following formulation: (morphology of Ailuropoda: morphology of Ursus deningeri, origin of the similarity or dissimilarity, direction of evolution [classified as same direction, uncorrelated direction or opposite direction]). Concerning the cusp terminology, we partly follow Rabeder (1999), with some modification when terms contradict with the principle of homology. Additional cusps in Ailuropoda not stressed by previous authors are named based on their origin and relative position to the main cusps. For example, the cusps developed from the medial slope of the protocone of the M1 are termed as “entprotocone”. The ridges are termed as R + abbreviation of the main cusp name (two letters for upper teeth and two letters + d for lower teeth). From the main cusp, the anterior ridge is termed as R1 and the posterior ridge is termed as R2, while other ridges are termed as R3 and R4. The branch ridges divergent from the main ridges are termed as main ridge name + .2. For example, inner branch ridge divergent from the middle part of the anterior ridge of the paracone of the M2 is termed as RPa1.2, and the inner ridge of the metaconid of the m1 is termed as RMed3. For a summary of terminology, see Fig. 2. For each species, we calculated the score of characters as the total number of derived characters and reversals (i.e., used to be derived but reversed later).
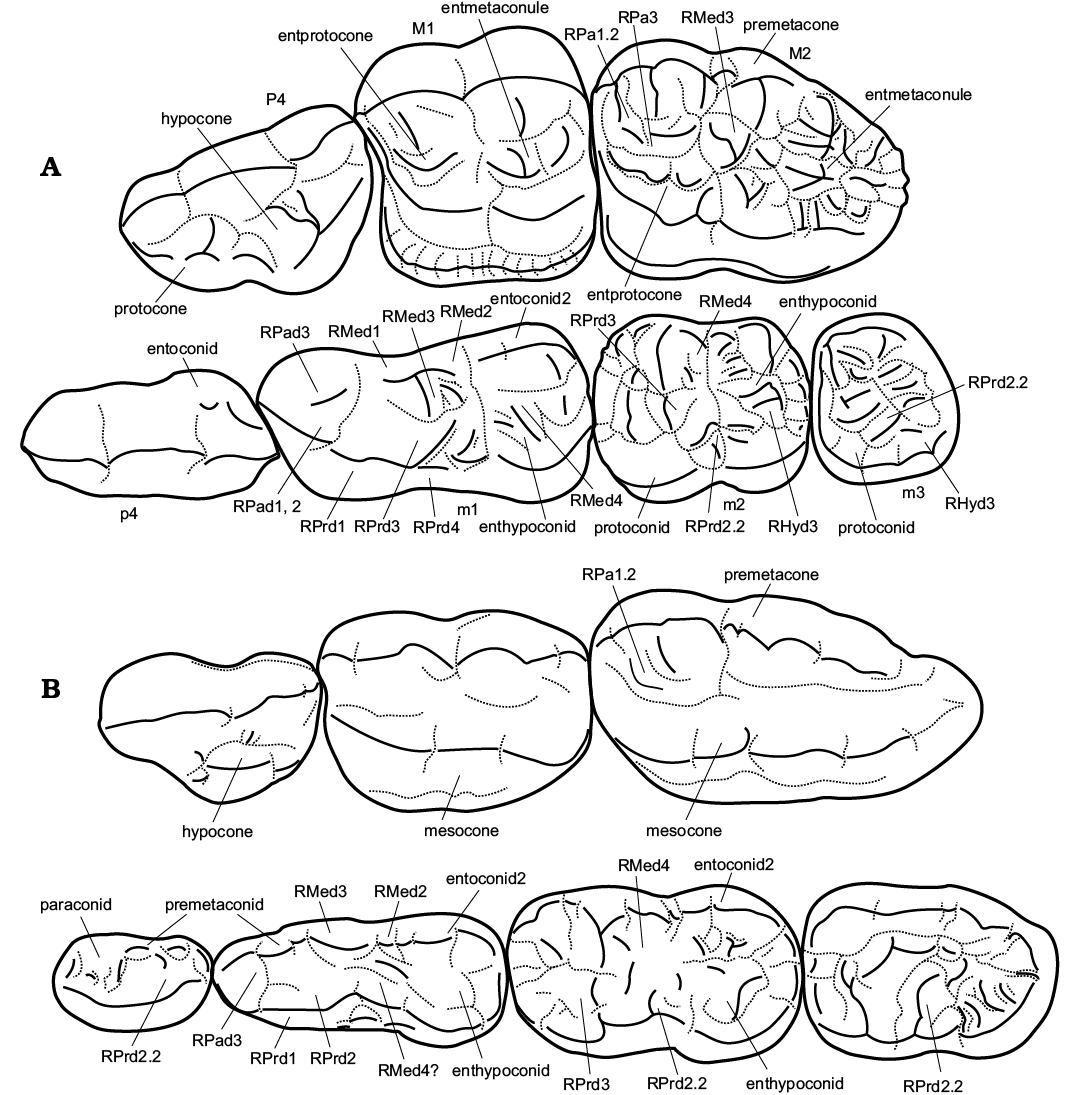
Fig. 2. Illustrations of proposed terminology of teeth of giant panda Ailuropoda melanoleuca (A) and cave bear Ursus deningeri (B). Abbreviations: RHyd3, medial ridge of protoconid; RMed1, anterior ridge of metaconid; RMed2, posterior ridge of metaconid; RMed3, medial ridge of metaconid; RMed4, postero-medial ridge of metaconid; RPad1, anterior ridge of paraconid; RPa1.2, medial branch of anterior ridge of paracone; RPad1, anterior ridge of paraconid; RPad2, posterior ridge of paraconid; RPa3/RPad3, medial ridge of paracone/paraconid; RPrd1, anterior ridge of protoconid; RPrd2, posterior ridge of protoconid; RPrd2.2, medial branch of the posterior ridge of protoconid; RPrd3, postero-medial ridge of protoconid; RPrd4, postero-lateral ridge of protoconid.
The teeth of Ailurarctos lufengensis are relatively small. For better observation of fine enamel characters of Ailurarctos lufengensis, a high-resolution CT scanning was performed using the 225 kV micro-computerised tomography (developed by the Institute of High Energy Physics, Chinese Academy of Sciences) at the Key Laboratory of Vertebrate Evolution and Human Origins, CAS. The specimens were scanned with the beam energy of 130 kV and a flux of 120 µA at a resolution of 40.78 micrometers per pixel using a 360° rotation with a step size of 0.5° and an unfiltered aluminum reflection target. A total of 720 transmission images were reconstructed in a 2048×2048 matrix of 1536 slices using a two-dimensional reconstruction software developed by the Institute of High Energy Physics, CAS.
To investigate whether a character is an autapomorphy for the lineage, a symplesiomorphy for all studied species, or resulting from convergence in different lineages, comparisons with the ancestral species are performed. Three pairs of nearly contemporary bears of the two lineages (Ailuropoda melanoleuca and Ursus deningeri; Ailuropoda microta and Ursus etruscus; Ailurarctos lufengensis and Ursavus tedfordi) and common ancestor Ballusia are chosen for the current analysis. For example, if one character is absent in Ballusia and Ursavus tedfordi, but present in the other five species, then it is interpreted as first appearing in the giant panda lineage and later convergently acquired in the cave bear lineage.
For the classification system, we generally adopted that of Qiu et al. (2014) in regarding the crown group Ursidae as divided into two subfamilies, the subfamily Ailuropodinae, including two tribes Ailuropodini and Indarctini, and the subfamily Ursinae, including Ursini and Arctotheriini (=Tremarctini).
Morphological homology and evolution of bear dentition
The tooth morphologies of the different known species of Ailuropoda are very similar and the characters described below are exactly the same for all species of the genus, so when we only use the genus name in our description, we mean that the character generally fits all species of the genus.
General evaluation.—The teeth of Ailuropoda are wide but low-crowned. The width of M1 is larger than the length. In Ursus M1 width is generally much smaller than its length (in Ursus malayanus the teeth are relatively wide, yet the width of M1 is always smaller than the length (Jiangzuo et al. 2014). Especially, Ursus deningeri has relatively slenderer teeth compared to its closest relative Ursus arctos (Wagner and Čermák 2012) and ancestor Ursus etruscus. In Ursavus and Ailurarctos, the width/length ratio of the tooth is intermediate between that of Ailuropoda and Ursus deningeri. (Wide teeth: slender teeth, widened tooth of Ailuropoda and elongated tooth of Ursus deningeri, opposite direction).
Anterior premolars.—P1–3/p1–3 are generally present in Ailuropoda, though P1/p1 is small, button-like and occasionally lost. P2/p2 and P3/p3 are enlarged with strong anterior and posterior accessory cusps (Davis 1964). In Ailurarctos lufengensis only P2 and P3/p3 are known. They are already enlarged (especially P3/p3) with distinct anterior and posterior accessory cusps, though of a much smaller size than those of Ailuropoda (Qiu and Qi 1989). In Kretzoiarctos the p1–3 are still very similar to those of Ursavus since they are double-rooted and of medium size but lacking the anterior and posterior accessory cusps (Abella et al. 2012). In Ursus deningeri the P1–3/p1–3 are generally lost (P1/p1 are more frequently present) (Rabeder 1999; Pacher 2017). In Ursus etruscus P1–3/p1–3 are generally present but rather small in size (Mazza and Rustioni 1992). (Well-developed anterior premolars: absence of anterior premolars, enlarged premolars of Ailuropoda and loss of anterior premolars of Ursus deningeri, opposite direction).
P4.—P4 of Ailuropoda is quite different from other bears (Fig. 3). Parastyle: Parastyle in Ailuropoda, there is a large parastyle in line with the paracone and the metacone. A similar situation can be found in Ailurarctos lufengensis. Most bears including Ursus and Ursavus do not have a parastyle. In Plithocyon the parastyle is small, formed by the cingulum and located buccal to the paracone (Ginsburg and Morales 1998). In Indarctos, on the contrary, the parastyle is often located lingual to the paracone and there is a distinct inner ridge in the lingual slope of the parastyle (Petter and Thomas 1986). The parastyle of Agriotherium is similar to that of Ailuropoda, which is large but lacks the lingual ridge and is in line with the paracone in most cases. (Parastyle present: parastyle absent, development of parastyle in Ailuropoda, uncorrelated direction).
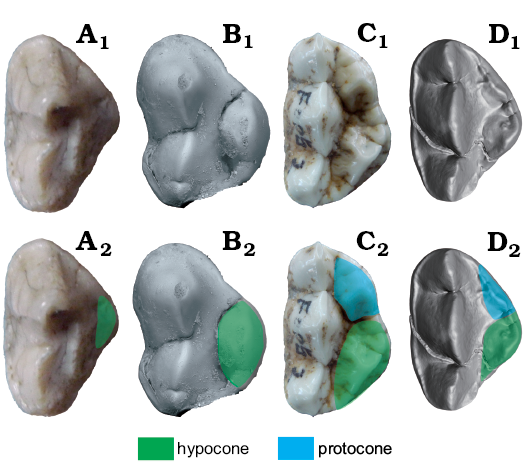
Fig. 3. Occlusal views of P4 homologous structures in cave bear (A, B) and giant panda (C, D) lineages. A. Ursavus tedfordi Qiu, Deng, and Wang, 2014 (HMV1453) from Huaigou, Gansu Province, Late Miocene. B. Ursus deningeri Von Reichenau, 1904 (NMM1946/643, photo Jan Wagner) from Mosbach 2, Germany, early Middle Pleistocene. C. Ailuropoda sp. (uncatalogued) from southern China, Pleistocene. D. Ailurarctos lufengensis Qiu and Qi, 1989 (IVPP V6892) from Lufeng, Yunnan Province, Latest Miocene. A1–D1, photographs; A2–D2, photographs with homologous structures indicated. Not to scale.
Paracone: Paracone in both Ailuropoda and Ursus, the lingual slope of the paracone is smooth. In Ailurarctos lufengensis it is also smooth while in Ailurarctos yuanmouensis there is a weak ridge (RPa3) connecting to the anteriorly placed protocone. This ridge is also present in Indarctos, Agriotherium, Hemicyoninae and more primitive bears (e.g., Cephalogalini). So it is highly probable that the loss of this ridge occurred independently in the two lineages. (RPa3 absent: RPa3 absent, convergence, same direction).
Protocone and hypocone: There are two cusps on the lingual side of the tooth. The anterior one (protocone) is just lingual to the notch between the parastyle and paracone, and the posterior one (hypocone) is lingual to the notch between the paracone and metacone. In most cases, one or several small cusps are present between these two main cusps. Buccal ridges are often present in the two large cusps but they are easily worn out and can only be observed in the unworn teeth. In Ailurarctos lufengensis, the morphology of P4 is very similar to that of Ailuropoda but the tooth is shorter and the two lingual cusps are closer to each other. The apex of the anterior cusp is located slightly posterior to the notch between the parastyle and the paracone and the apex of the posterior cusp is located slightly anterior to the notch between the paracone and the metacone. Ailurarctos yuanmouensis is primitive in lacking the small cusp between the protocone and the hypocone. In Ursus, there is only one cusp at the postero-lingual corner. The homologue of this cusp in Ailuropoda and other Carnivora is worth discussing. In most members of Carnivora, the protocone is located at the antero-lingual corner of P4 and is connected with the RPa3. In many clades, the cusp of the inner lobe is posteriorly shifted and the RPa3 is connected to the anterior corner of the inner cusp. This arrangement is not just the result of the posterior shift of the protocone. A much more likely process is that the original protocone is reduced and that the hypocone develops to become the main lingual cusp during the rearrangement. Robust evidence of this can be seen in Musteloidea (Fig. 4). In some representatives, like Meles, both the protocone and hypocone are present. RPa3 connects with a rather small protocone, while the hypocone is larger. In many other musteloid taxa such as Enhydriodon and Procyon, the protocone is still larger, and both the protocone and hypocone are well-developed. A similar situation has been recently noticed by Geraads and Spassov (2016) who argued that in Mephitidae, the main cusp of the inner lobe is the hypocone rather than protocone. Early representatives of bears (Cephalogale, Hemicyoninae, and Ballusia) generally have a uniform P4 structure, with only one cusp at the inner lobe and the RPa3 being connected to the anterior corner of the inner cusp. The main axis of this cusp (long axis of the cusp) is also similar to the hypocone of Meles and Enhydriodon (antero-lingual to posterior-buccal in direction). These characters provide firm evidence that the main cusps of the inner lobe of these bears are homologous to the hypocone of Meles and Enhydriodon (as well as other Carnivora). A transition form can be seen in Phoberogale depereti from Saint Gérand-le-Puy (MNHN.F.SG4300), where a small protocone is still present but the hypocone is already become the dominant cusp of the inner lobe (see De Bonis 2013: fig. 8). Therefore, the main lingual cusp in most bears is homologous to the hypocone of other carnivores. This shift of the main cusp in the inner lobe of the P4 has been documented by Beaumont (1982), though he did not compare this cusp with non-ursid Carnivora. An independent way of judging the property of the inner lobe cusps is through the occlusal relationship as proposed by Geraads and Spassov 2016. They believed that the protocone should be located at the anterior side of the m1 trigonid during occlusion, whereas the hypocone should be located within the trigonid, i.e., between the paraconid and metaconid. In all bears, the major cusp in the inner lobe of the P4 indeed interlocks within the trigonid of the m1, suggesting the hypocone property of the cusp.
Even though the RPa3 is lost in Ailuropoda, Ailurarctos yuanmouensis still retains this trait. The connection of the anterior cusp with the RPa3 in Ailurarctos yuanmouensis indicates that the protocone reappeared in the curse of Ailuropodini evolution, the anterior cusp being thus the protocone and the posterior cusp being the hypocone. Tooth occlusion pattern also supports this arrangement. In Ursus deningeri, only the hypocone is present, with occasionally some small cusps anterior or posterior to the main cusp. In Indarctos, there are often two closely located cusps in the inner lobe, but the RPa3 connects to the anterior corner of the inner lobe instead of to the anterior cusp as in Ailurarctos yuanmouensis and the whole inner lobe mainly interlocks within the m1 trigonid. Therefore, the two cusps of derived Indarctos both origin from the subdivision of the hypocone. The bicuspid inner lobe evolved independently in Indarctos and Ailuropoda, and thus cannot be viewed as a synapomorphy for these two genera. A detailed review of the P4 protocone/hypocone of Carnivora is in preparation and will be discussed elsewhere. (Reappearance of protocone: absence of protocone, reappearance of protocone in Ailuropoda, uncorrelated direction.)
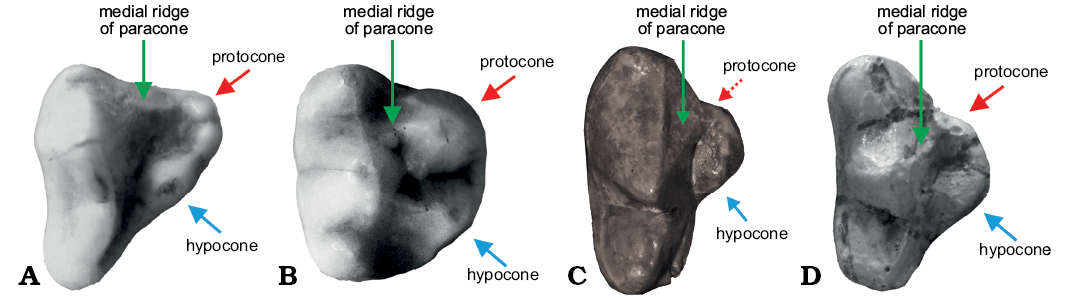
Fig. 4. Occlusal views of P4 inner lobe structure of Musteloidea and Ursidae, showing the homology of inner lobe cusps. A. Meles leucurus Hodgson, 1847 (IOZ08129), Recent, China. B. Procyon lotor (Linnaeus, 1758) (IVPP OV1551), Recent, zoo specimen. C. Plithocyon teilhardi (Colbert, 1939) (AMNH FM26594) from the Tunggur Formation, Middle Miocene; the dashed line indicates that the protocone is absent in this species. D. Ailurarctos yuanmouensis Zong, 1996 (IVPP RV97001, cast) from Yuanmou, Yunnan Province. Not to scale.
M1.—M1 of Ailuropoda is rather square in shape (Fig. 5). Parastyle: Parastyle is present in both Ailuropoda and Ursus deningeri, while the metastyle is only present in Ursus deningeri. A distinct parastyle or metastyle is absent in Ailurarctos lufengensis and Ursavus tedfordi, thus the presence of the parastyle in the two lineages is probably due to convergence, though these two characters are in fact very variable between and within species of Ursini and Arctotheriini. (Parastyle present: parastyle present, convergence, same direction; metastyle present: metastyle absent, development of metastyle in Ursus deningeri, uncorrelated direction).
Paracone: Paracone is relatively simple in Ailuropoda. A medial branch (RPa1.2) is developed at the anterior ridge and sometimes curves and contacts with the inner ridge (RPa3). RPa3 is well developed and often develops into a separate cusp (which is termed entparacone here). This cusp is often slightly anteriorly curved. In Ailurarctos lufengensis, the anterior part of the tooth with RPa1 is broken (RPa1.2 is absent in Ailurarctos yuanmouensis) and the RPa3 is very weak. In all other bears including Ursus deningeri, Ursus etruscus, and Ursavus tedfordi, the RPa1.2 is absent and the RPa3 is barely developed (RPa1.2 present: RPa1.2 absent, development of RPa1.2 in Ailuropoda, uncorrelated direction; RPa3 well developed: RPa3 weak, development of RPa3 in Ailuropoda, uncorrelated direction). (iii) The protocone of Ailuropoda is not subdivided into two cusps as in Ursus. In Ursus, the posterior protocone is located opposite to the notch between the paracone and the metacone, and is often termed as mesocone (Rabeder 1999). This mesocone originated either from the subdivision of original protocone or the separation of original postprotocrista. However, in Ballusia, the preprotocrista-protocone-postprotocrista forms a continuous ridge and there is no way to define which part can be called postprotocrista. Here we treat the mesocone as the posterior cusp of the subdivided protocone. In Ailurarctos lufengensis and Ursavus tedfordi, the protocone is still not subdivided while in Ursavus sylvestris (Qiu and Qi 1990) it is weakly subdivided. In Indarctos and some specimens of Agriotherium, the protocone is also weakly subdivided. Between the paracone and protocone, there is a distinct but low tubercle in Ailuropoda. In Ailurarctos lufengensis this tubercle is still absent, so it does not give much information about whether this tubercle was developed from a part of the paracone or the protocone. In general, the new structures are more likely to develop in more open space. Considering that the medial slope of the protocone is always wider than the medial slope of the paracone in Ursavus and Ailurarctos lufengensis, and considering the presence of several small ridges in the medial slope of the protocone in Ailurarctos lufengensis, it is more probable that this tubercle is developed from strong bulging of the medial slope ridges of the protocone. This tubercle is here termed as the entprotocone. The entprotocone is not known in any other bears. (Mesocone absent: mesocone present, development of posterior protocone in Ursus, uncorrelated direction; entprotocone present: entprotocone absent, development of entprotocone in Ailuropoda, uncorrelated direction).
Metaconule: The postero-lingual cusp of bears used to be called as hypocone. However, the hypocone is generally referred to the cusp of the lingual cingulum, while the postero-lingual cusp in bears is developed from a metaconule on the protocone-metacone ridge as stressed by Beaumont (1965, 1982), Qiu and Schmidt-Kittler (1983) and Baryshnikov (2007), an interpretation that is adopted here. As in the protocone, there is a distinct tubercle between the metaconule and the metacone in Ailuropoda. This tubercle (termed as entmetaconule) is also absent in other bears. (Entmetaconule present: entmetaconule absent, development of entmetaconule in Ailuropoda, uncorrelated direction.)
Lingual cingulum: Lingual cingulum of Ailuropoda is very wide and strongly serrated. In Ailurarctos lufengensis the lingual cingulum is without serration, similar to that of Ursavus tedfordi. In Ursus deningeri, the cingulum is variably serrated but much thinner than that of Ailuropoda. (Lingual cingulum wide: lingual cingulum thin, widen of lingual cingulum in Ailuropoda, uncorrelated direction).
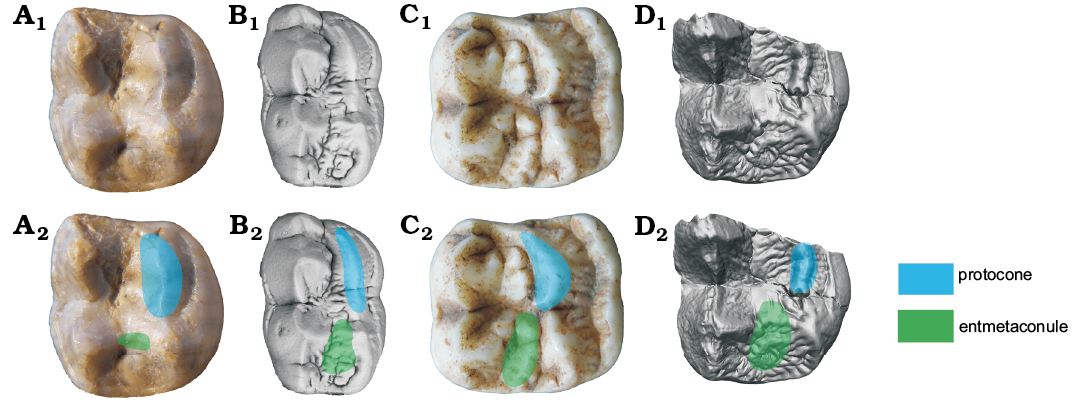
Fig. 5. Occlusal views of M1 homologous structures in cave bear (A, B) and giant panda (C, D) lineages. A. Ursavus tedfordi Qiu, Deng, and Wang, 2014 (HMV1453) from Huaigou, Gansu Province, Late Miocene. B. Ursus deningeri Von Reichenau, 1904 (NMM1953/119, photo Jan Wagner) from Mosbach 2, Germany, early Middle Pleistocene. C. Ailuropoda melanoleuca (David, 1869) (IVPP V87025.109) from cave deposit of Guangxi Province, Late Pleistocene. D. Ailurarctos lufengensis Qiu and Qi, 1989 (IVPP V6892) from Lufeng, Yunnan Province, Latest Miocene. A1–D1, photographs; A2–D2, photographs with homologous structures indicated. Not to scale.
M2.—The M2 of Ailuropoda is similar to that of other bears with a distinct talon. The talon is not significantly elongated but the whole tooth is widened as in the M1 (Fig. 6).
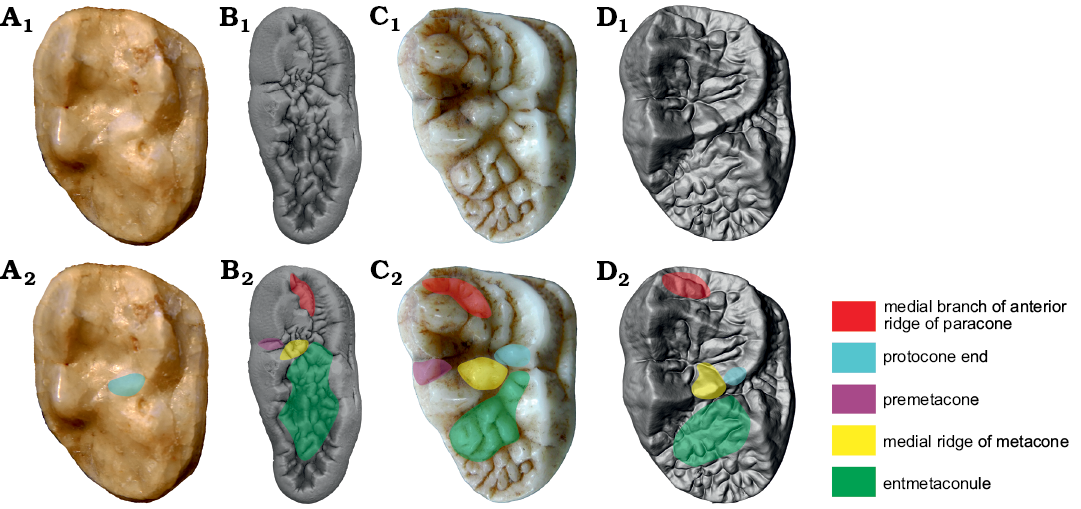
Fig. 6. Occlusal views of M2 homologous structures in cave bear (A, B) and giant panda (C, D) lineages. A. Ursavus tedfordi Qiu, Deng, and Wang, 2014 (HMV1453) from Huaigou, Gansu Province, Late Miocene. B. Ursus deningeri Von Reichenau, 1904 (NMM1953/119, photo Jan Wagner) from Mosbach 2, Germany, early Middle Pleistocene. C. Ailuropoda melanoleuca (David, 1869) (IVPP V87025.153) from cave deposit of Guangxi Province, Late Pleistocene. D. Ailurarctos lufengensis Qiu and Qi, 1989 (IVPP V6892) from Lufeng, Yunnan Province, Latest Miocene. A1–D1, photographs; A2–D2, photographs with homologous structures indicated. Not to scale.
Paracone: The paracone of M2 is very similar to that of M1, but the corresponding characters are even more developed. The RPa1.2 is strongly medially curved and always contacts with the RPa3. The RPa3 is more separated than that of M1. It extends along the medial slope of the paracone, forming a well-developed ridge parallel to the sagittal axis. The RPa2 also turns slightly medially. These three ridges form a semicircle surrounding the paracone. In Ailurarctos lufengensis, the RPa1.2 is present. The RPa3 is well-developed, but its medial part is not separated and forms only weak ridge parallel to the sagittal axis. In Ursus deningeri, the RPa1.2 is variable present and always weaker than in Ailuropoda and the RPa3 only occasionally develops into a ridge similar to that in Ailuropoda. The RPa1.2 is widely present in Ursus, but it is always weak and only present at low frequency in the sample. The RPa1.2 is absent in Ursavus tedfordi. The RPa3 is often weak or absent in Ursus deningeri, but some specimens do have a large and weakly separated RPa3 (Fig. 7). (RPa1.2 present: RPa1.2 present, convergence, same direction; RPa3 separated: RPa3 separated, convergence, same direction).
Fig. 7. Occlusal views of weakly separated RPa3 of Ursus deningeri Von Reichenau, 1904. A. NMP Rv21000, Koněpruské jeskyně. B. NMP Ra153, cave C718. C. NMP Ra2402, Chlum 1. D. NMP Rv20999, Koněpruské jeskyně. All from Czech Republic, early Middle Pleistocene. Photo Jan Wagner.
Metacone: There is always a small cusp developed in the anterior ridge of the metacone in Ailuropoda. This cusp is termed as premetacone. It is absent in Ailurarctos lufengensis as well as in most Ursus, while it is sometimes present in Ursus deningeri (and later cave bears). The medial ridge of the metacone (RMe3) is strong and connected with the protocone. The RMe3 is subdivided into two parts. In Ursus, the RMe3 is generally is very weak or absent, though in some specimens of Ursus deningeri this ridge is still present but not subdivided. This ridge is distinct in Ailurarctos lufengensis, while it is already very weak in Ursavus tedfordi, Indarctos, and Agriotherium. (Premetacone present: premetacone present, convergence, same direction; RMe3 subdivided: RMe3 not subdivided, subdivision of RMe3 in Ailuropoda, uncorrelated direction).
Protocone: The protocone of Ailuropoda is not subdivided. In the medial slope, there is a ridge parallel to the protocone and this ridge may be subdivided into two or three cusps. This ridge is homologous to the entprotocone of the M1. There is no trace of the entprotocone in Ailurarctos lufengensis, and it is absent in other bears. The distal end of the protocone ridge is medially shifted in most primitive bears. This end is connected with RMe3 in Ailuropodini. In Ursus, this distal end is directly connected with the metaconule and not medially shifted (or only very slightly), leaving the middle valley open. In Ursavus tedfordi, this distal end is still similar to the situation seen in primitive bears with a strong medial shift. In Indarctos and Agriotherium, the distal end is similar to Ursus without medial shift. (Entprotocone present: entprotocone absent, development of entprotocone in Ailuropoda, uncorrelated direction; protocone distal end medially shifted: protocone distal end not medially shifted, direct connection of protocone distal end to metaconule in Ursus deningeri, uncorrelated direction).
Metaconule: As in other bears, the metaconule of Ailuropoda is elongated to support the talon. The metaconule is sometimes subdivided into several parts. Medial to the metaconule, there are many tubercles and ridges, which seem to be rather variable in strength and arrangement. Considering the overall similarity between M1 and M2 (e.g., RPa3, RMe3, and entprotocone), structures similar to those in M1 should be developed in the same area. The observation of a large number of specimens confirms that two distinct elements are present in this area: entmetaconule and ridge band along the talon border (often forming radiated tiny ridges perpendicular to the talon border). Due to the elongation of the talon, the metaconule and the entmetaconule are also elongated and have enough space to further subdivide into several parts. In some cases, the entmetaconule and the talon border are clearly separated, but in most cases, these two parts are both highly subdivided into small cusps/ridges and their boundary is no longer clear. In Ailurarctos the metaconule is not subdivided and the talon is filled with small tubercles but there is no trend for subdivision of the entmetaconule and the talon border. In Ursus, the talon is further elongated and the metaconule sometimes subdivided into several parts, but the entmetaconule is always weaker. In Ursavus tedfordi, the talon is relatively short. The metaconule is not subdivided, and the talon is relatively smooth, without entmetaconule. (Subdivision of metaconule present: subdivision of metaconule present, convergence, same direction; distinct entmetaconule: weak entmetaconule, Strong development of entmetaconule in Ailuropoda, uncorrelated direction).
p4.—p4 of Ailuropoda is very different from other bears. There are prominent anterior and posterior accessory cusps. The anterior accessory cusp is developed from the anterior cingulum. There are two ways to form a posterior accessory cusp: one is from the posterior ridge of p4 (RPrd2), as in Canidae or Phoberocyon, and another is from the posterior cingulum, as in Ursus. In Ailuropoda, there is a small posterior cingulum cusp in p2. Considering the serial homology of p2–p4 of Ailuropoda, the posterior accessory cusp of p4 is homologous to the posterior cingulum of p2. The same is true for the anterior accessory cusp. There are two structures often present on the inner slope of the posterior accessory cusp: (i) a small cusp that is often present on the antero-lingual margin of the posterior accessory cusp and (ii) a cingulum that is often present at the postero-lingual corner of the posterior accessory cusp. The latter sometimes develops into a cusp. There is no ridge between the main cusp (protoconid) and the cusp that is present on the antero-lingual border of the posterior accessory cusp, so this cusp cannot be homologous to the metaconid of m1. Considering their position at the inner part of the posterior accessory cusp (which is homologous to the hypoconid of m1). These two structures are probably homologous to the entoconid-complex of m1, so the anterior one is designated as entoconid 2 and the posterior one is designated as entoconid 1. The situation in Ailurarctos lufengensis is nearly the same as in Ailuropoda, though the entoconid 2 is smaller and placed at the postero-lingual corner of the protoconid and the entoconid1 is weaker. In Ailuropoda, the posterior ridge of the main cusp (protoconid) is never subdivided into two ridges, but such a subdivision is known for ursine bears. The lingual ridge (termed here RPrd2.2) is widely present in Ursus (though in some specimens of some species, e.g., Ursus malayanus, this ridge is absent) and occasionally present in Arctotheriini bears. It is absent in all other bears. The RPrd2.2 is present in Ursavus tedfordi but it is very weak and short. In Ursus deningeri, the RPrd2.2 is often very weak or lost, but the cusps developed from this ridge are often present, which in Ursus are often called the metaconid (Wagner and Čermák 2012), though whether this cusp is homologous to the metaconid of the lower molars is still open to discussion since the metaconid in molars is directly connected with the protoconid rather than with the RPrd2.2. The paraconid and the hypoconid are often present in Ursus deningeri, and the cingulid cusp (entoconid) is often developed lingual to the hypoconid. These two cusps (paraconid and hypoconid) are located at the anterior and posterior tips of the crown and are probably developed from the cingulid; they are homologous to the anterior and posterior accessory cusps in Ailuropoda. The anterior and posterior accessory cusps in Ailuropoda are in line with the protoconid, while in Ursus deningeri the paraconid is always located at the antero-lingual corner of the tooth (which is more similar to that of m1). Several small cusps (premetaconid) can be present in the lingual slope of the protoconid in Ursus deningeri (see Fig. 2). These cusps are absent in Ailuropoda. In Ursus etruscus, the paraconid is sometimes present, while other accessory cusps are generally absent. All these accessory cusps are absent in Ursavus tedfordi. (Paraconid present: paraconid present, convergence; hypoconid present: hypoconid present, convergence; metaconid absent: metaconid present, development of metaconid in Ursus deningeri, uncorrelated direction; entoconid present: entoconid present; convergence; premetaconid absent: premetaconid present, development of RPrd2.2 in Ursus deningeri, uncorrelated direction; RPrd2.2 absent: RPrd2.2 absent, reversal of RPrd2.2 in Ursus deningeri, uncorrelated direction)
m1.—m1 of Ailuropoda
remains primitive without distinct modification (Fig. 8). Paraconid: There is a lingual ridge (RPad3) in
the paraconid of Ailuropoda. This ridge is
absent in Ailurarctos lufengensis,
Arctotheriini and more primitive bears. In Ailurarctos
lufengensis, the lingual slope of the paraconid is convex. The
RPad3 is sometimes present in advanced Ursus (including
Ursus deningeri), but it is often connected
with the metaconid through the premetaconid complex (either cusps or
ridges). In Ursavus tedfordi and primitive
Ursus, this ridge is absent. (RPad3 present:
RPad3 present, convergence, same direction).
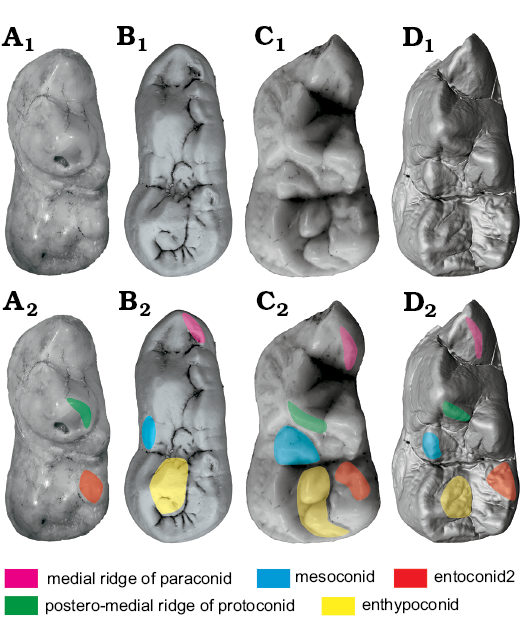
Fig. 8. Occlusal views of m1 homologous structures in cave bear (A, B) and giant panda (C, D) lineages. A. Ursavus tedfordi Qiu, Deng, and Wang, 2014 (HMV1453) from Huaigou, Gansu Province, Late Miocene. B. Ursus deningeri Von Reichenau, 1904 (NMM1956/907, photo Jan Wagner) from Mosbach 2, Germany, early Middle Pleistocene. C. Ailuropoda wulingshanensis Wang and Lin, 1982 (IVPP V13459.10) from Longgu Cave, Jianshi, Hubei Province. D. Ailurarctos lufengensis Qiu and Qi, 1989 (IVPP V25032) from Lufeng, Yunnan Province, Latest Miocene. A1–D1, photographs; A2–D2, photographs with homologous structures indicated. Not to scale.
Protoconid: Two posterior ridges are present in Ailuropoda, one (RPrd4) towards the lateral side and another (RPrd3) towards the metaconid. The presence of the RPrd3 is primitive characters for mammals (part of protolophid of Osborn [1907]). In Carnivora, the RPrd3 is present in most taxa, while the RPrd4 is less common (e.g., absent or very weak in Canidae). In Ailuropodinae, the RPrd4 is connected to the anterior end of the mesoconid (Ailurarctos lufengensis, Indarctos) or to its lateral side (Ailuropoda, Agriotherium). In Tremarctos, there are three ridges in the posterior side, the postero-buccal ridge is often located lateral to mesoconid and represents the RPrd4 as in Ailuropodinae. The postero-medial ridge is RPrd3. There is still a posterior ridge slightly lingual to the apex of the protoconid, representing RPrd2, a structure not present in Ailuropodinae. In Ursinae, the RPrd2 is absent and RPrd2 is often laterally turned and connects with the medial side of the mesoconid (forming a ridge in Ursinae). In Ursus deningeri, the RPrd2 is often directly connected with the mesoconid, but some specimens remain in the primitive stage with the RPrd2 being connected to the medial side of the mesoconid. The RPrd3 is widely present in primitive bears (such as Hemicyoninae, Cephalogalini and amphicynodontids), but becomes weaker and anteriorly shifted in advanced Ursavus and primitive Ursus, while it is lost in advanced Ursus (Fig. 9). (RPrd4 present: RPrd4 absent, reduced of RPrd4 in Ursinae, uncorrelated direction; RPrd2 absent: RPrd2 present, appearance of RPrd2 in Ursinae, uncorrelated direction; RPrd3 present: RPrd3 absent, loss of RPrd3 in Ursus deningeri, uncorrelated direction)
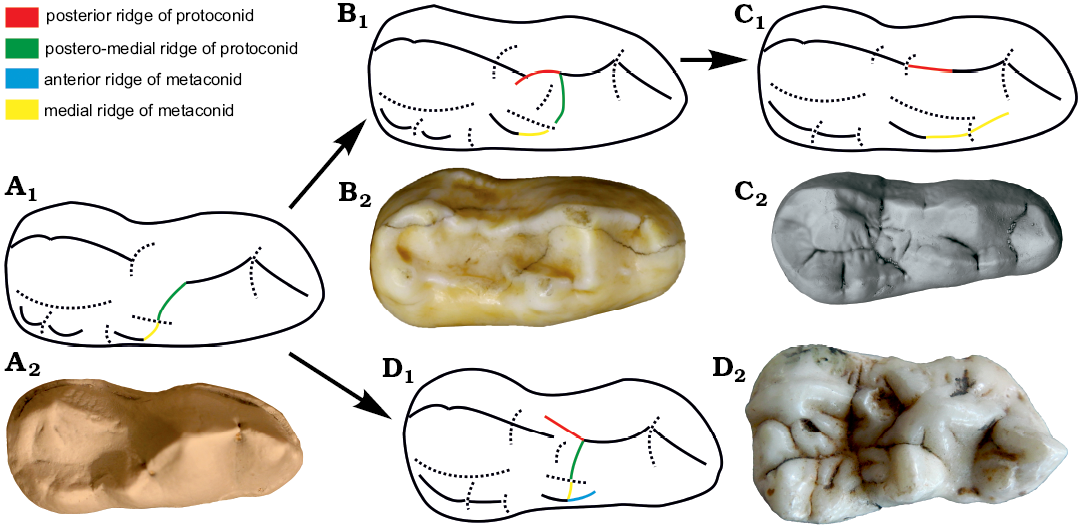
Fig. 9. Illustration showing the evolution of m1 protoconid-metaconid ridges (in occlusal view). A. Stem Ursavus evolutionary grade Ursavus cf. brevirhinus (MNHN Ar2399, photo Jan Wagner) from Artenay, France. B. Early Ursus evolutionary grade Ursus malayanus (Raffles, 1821) (AMNH M19154) from Borneo, living. C. Derived Ursus evolutionary grade Ursus deningeri von Reichenau, 1904 (NMM1956/668; photo Jan Wagner, reversed) from Mosbach 2, Germany, early Middle Pleistocene. D. Ailuropoda evolutionary grade: Ailuropoda wulingshanensis Wang and Lin, 1982 (IVPP V13459.266) from Longgu Cave, Jianshi, China, middle Early Pleistocene. A1–D1, explanatory drawings; A2–D2, photographs. Not to scale.
Metaconid: Four ridges are present in the metaconid of Ailuropoda: one (RMed1) towards the paraconid, one (RMed2) towards the entoconid, another (RMed3) towards and connected with the postero-lingual ridge of the protoconid (RPrd3) and the small RMed4 (see below for details). Among these ridges, the RMed3 and RMed2 are the original antero-medial and posterior ridges of Carnivora, but the anterior shift of the metaconid changes the direction of the RMed3 (more medially, nearly perpendicular to the sagittal axis of the tooth). A small but distinct ridge (RMed4) is present between the RMed2 and the RMed3. This ridge appears in the valley between the RMed2 and the RMed3, and does not reach to the apex of the metaconid. In Ailurarctos lufengensis, the RMed 1 is still absent, while a very weak RMed4 is already present. In Ursus, the metaconid is relatively posteriorly shifted and the RMed3 is anteriorly shifted, directed towards the paraconid. In Ursus deningeri, Ursus etruscus and other advanced Ursus, the RMed3 is no longer connected with the RPrd3 and often develops into one or several cusps (premetaconid complex). There is often a ridge connecting the metaconid and the hypoconid (or enthypoconid) in Ursus. Whether this ridge is homologous to the RMed4 of Ailuropoda and Ailurarctos lufengensis is still unclear. Since the anterior tip of this ridge is not connected with the medial slope of the metaconid as in Ailuropoda but often slightly lingual to the posterior tip of the metaconid, it is not viewed as RMed4 here. In Ursavus tedfordi, the RMed3 is still connected with the RPrd3, and the RMed4 is absent. (RMed1 present: RMed1 absent, development of RMed1 in Ailuropoda, uncorrelated direction; RMed3 anteriorly extended: RMed3 Buccally extended, anterior shift of metaconid in Ursus deningeri, uncorrelated direction; RMed4 present: RMed4 absent, development of RMed4 in Ailuropoda, uncorrelated direction).
Mesoconid: An accessory cusp (mesoconid) between the protoconid and the hypoconid is present in Ailuropoda, with three distinct ridges, one towards the protoconid, another towards the hypoconid and the last one (homologous to RPrd2.2 in p4 and m2) towards the medial side. This cusp is also present in Arctotheriini (Tedford and Martin 2001), Ailurarctos lufengensis and Indarctos. It is absent in Ursavus (and Hemicyoninae or primitive bears) and ridge-like in most members of Ursus, but often becomes slightly cusp-like in Ursus deningeri. The medial ridge of the mesoconid is present in one specimen of Ailurarctos lufengensis (V6892.9), but absent in another. This ridge is generally absent in other bears. In Ursus deningeri, this ridge is occasionally present, but always weak. (Mesoconid present: mesoconid present, convergence, same direction; medial ridge of mesoconid distinct: medial ridge of mesoconid generally absent, development of medial ridge of mesoconid in Ailuropoda, uncorrelated direction)
Hypoconid: The hypoconid is simple in Ailuropoda, but there are several tubercles present on the medial slope of the hypoconid. A similar structure in Ursus deningeri was termed as enthypoconid by Rabeder (1999). In general, there are two large tubercles in Ailuropoda, the anterior one being sometimes subdivided by a groove. In Ailurarctos lufengensis, the enthypoconid is absent, but several fine ridges are present, starting from the tip of the hypoconid. In Ursus deningeri, the enthypoconid is always present but variable in the degree of further subdivision. In Ursus, a distinct groove in the antero-lingual slope of the hypoconid is always present, and another groove in the postero-medial slope is also present in most taxa. These two grooves result from the expansion of the medial slope of the hypoconid and define the boundary of the enthypoconid. (Enthypoconid present: enthypoconid present, convergence, same direction).
Entoconid-complex: Generally, there is only one main cusp in the entoconid region in Ailuropoda. A small cusp anterior to the main apex is occasionally present. This small cusp is evidently developed from the anterior ridge of the main apex of the entoconid. In Ailurarctos lufengensis, there are two distinct cusps, with the anterior one (entoconid 2) slightly larger than the posterior one (entoconid 1). The same is true for Indarctos. There are several possible scenarios for the transition from Ailurarctos lufengensis to Ailuropoda arrangement. Considering the large size of entoconid 2 in Ailurarctos lufengensis, it is possible that the single cusp in Ailuropoda is derived from the anterior cusp in Ailurarctos lufengensis. Alternatively, the single cusp in Ailuropoda may be derived from the posterior cusp of Ailurarctos lufengensis since they are in a similar position. Another possibility is that the two cusps in Ailurarctos lufengensis merge into the single cusp seen in Ailuropoda. As has been mentioned above, the anterior ridge sometimes develops into a cusp. In some specimens of Ailuropoda (present in all species of the genus, from Ailuropoda microta to Ailuropoda melanoleuca), this cusp enlarges to nearly the same size as the posterior cusp and resembles the situation in Ailurarctos lufengensis (Fig. 10). The variability of this cusp in Ailuropoda, thus supports the third hypothesis stating that the single cusp in Ailuropoda is mainly derived from the posterior cusp of Ailurarctos and that the anterior cusp is merged with the posterior cusp. As is known from the developmental process, two cusps may merge when their second enamel knots are too closed to each other (Ungar 2010). Therefore, the merging of the entoconid 2 to the entoconid 1 may be due an increase of the entoconid size relative to the talonid length, forcing the two cusp primers closer to each other until finally merging. In Ursavus, the entoconid consists of two subequal-sized cusps, while only one cusp is present in most early Ursus. Among the early representatives of Ursus with double entoconid, Ursus “minimus” from Moiseevka, Kazakhstan shows two ridge-like entoconids, with the anterior one being slightly larger, while in Ursus “minimus” from Kosyakino of Caucasus, the opposite is true, with the anterior entoconid being smaller (Baryshnikov and Lavrov 2013). Therefore, it is hard to decide which cusp in Ursavus is equivalent to the posterior cusp in living Ursus. The situation in Ursus is probably different from the cusp merging pattern in Ailuropoda, since in Ursus minimus the only entoconid is restricted to the postero-lingual corner of the talonid, leaving the talonid basin open lingually. In Ursus etruscus the anterior ridge of the entoconid is anteriorly extended and the anterior cusp sometimes reappears as further development of the anterior ridge, whereas in Ursus deningeri the anterior cusp is nearly always present and further enlarged or even splits into several cusps. Such a condition is more or less similar to that seen in Ailuropoda. Considering the position of the entoconid in most primitive Ursus, the homology of this cusp (the apex of this cusp is never anterior to the apex of the hypoconid) to the original posterior cusp in Ursavus is more likely. Therefore, the main entoconid cusps in Ailuropoda and Ursus deningeri are probably homologous. The anterior cusp in Ailuropoda represents the original entoconid 2 (plesiomorphic characters), while in Ursus deningeri the anterior cusp is a newly appeared cusp. Anyway, the developmental basis of the entoconid-complex in both taxa may be similar. In the medial slope of the entoconid 1 and 2, one or more ridges (REnd3) are often present in Ailuropoda. These ridges are also present in Ailurarctos lufengensis, but very rare in other bears. (Entoconid 2 tends to be absent: entoconid 2 present, reversal appearance of entoconid 2 in Ursus deningeri, uncorrelated direction; REnd3 present: REnd3 absent, development of REnd3 in Ailuropoda, uncorrelated direction).
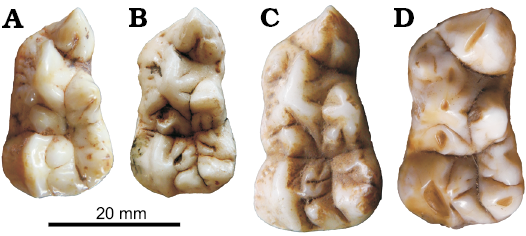
Fig. 10. Occlusal views of large entoconid 2 of Ailuropoda spp. A, B. Ailuropoda wulingshanensis Wang and Lin, 1982 from Longgudong Cave, Jiangshi, China, middle Early Pleistocene. A. IVPP V13459.11. B. IVPP V13459.266. C, D. Ailuropoda melanoleuca (David, 1869). C. IVPP V87025.246 from cave deposit, Guangxi Province, China, Late Pleistocene. D. IOZ 32752, Recent, Sichuan Province.
m2.—The m2 of Ailuropoda is not elongated (Fig. 11). There is distinct middle constriction as in cave bears. The middle constriction is generally absent in other Ursus (though sometimes weakly present in U. arctos) and Ailurarctos lufengensis. The constriction in Ailuropoda is mainly developed on the buccal side to accommodate the enlarged paracone complex of the M2. In Ursus deningeri, the whole talonid is often widened while the paracone of M2 is not enlarged. It is possible that the constriction in Ursus deningeri only results from the enlargement of the talonid. (Middle constriction present: middle constriction present, convergence, same direction).
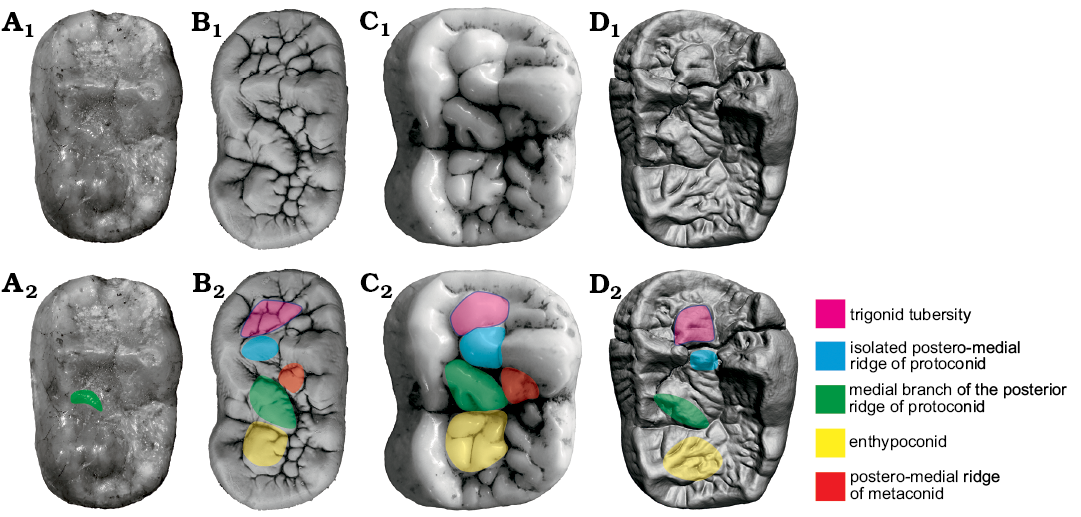
Fig. 11. Occlusal views of m2 homologous structures in cave bear (A, B) and giant panda (C, D) lineages. A. Ursavus tedfordi Qiu, Deng, and Wang, 2014 (HMV1453) from Huaigou, Gansu Province, Late Miocene. B. Ursus deningeri von Reichenau, 1904 (NMM1956/909, photo Jan Wagner) from Mosbach 2, Germany, early Middle Pleistocene. C. Ailuropoda wulingshanensis Wang and Lin, 1982 (IVPP V13459.91) from Longgu Cave, Jianshi, Hubei Province. D. Ailurarctos lufengensis Qiu and Qi, 1989 (IVPP V6892) from Lufeng, Yunnan Province, Latest Miocene. A1–D1, photographs; A2–D2, photographs with homologous structures indicated. Not to scale.
Anterior border: The anterior border of the m2 in Ailuropoda and Ursus is composed of several cusps of variable number and size. In Ailuropoda, there are often two or three cusps at the anterior lingual corner and the rest of the border is only serrated without distinct cusps. In Ailurarctos lufengensis the situation is similar to that of Ailuropoda, but there is only one cusp at the antero-lingual corner. In Ursavus tedfordi and Indarctos the anterior border is generally smooth. Whether these small cusps at the anterior border of Ailuropoda and Ursus are homologous to the paraconid is not known. In Ursus deningeri the anterior border often comprises a variable number of cusps while the cusp at the antero-lingual corner is usually the most distinct one. The paraconid is present in some Hemicyoninae, such as Phoberocyon and Dinocyon, as a distinct cusp at the antero-lingual corner (Ginsburg and Morales 1998). (Serrated anterior border present: serrated anterior border present, convergence, same direction)
Protoconid: As in other bears, the protoconid is not subdivided into several cusps in Ailuropoda. In all bears, the protoconid is connected with the metaconid by a distinct ridge, but this ridge is separated by a central valley into two parts (RPrd3 and RMed3; entprotoconid and entmetaconid sensu Rabeder 1999). In Ailuropoda, the ridge is further separated by valleys into three distinct parts. In Ailurarctos lufengensis the RPrd3 shows an initial trace of further subdivision, while the RMed3 is still complete. This suggests the two buccal parts in Ailuropoda are derived from the subdivision of the RPrd3 in Ailurarctos lufengensis. In most Ursus, the RPrd3 is continuous while in Ursus deningeri RPrd3 is often subdivided into 2 or even more parts, but the separation is not as distinct as in Ailuropoda. In the middle of the posterior ridge (RPrd2), another ridge (RPrd2.2; mesolophid sensu Rabeder 1999) diverges from the main ridge. These two ridges end at the deep valley separating the trigonid and the talonid. The lingual ridge (RPrd2.2) is absent in Hemicyoninae and more primitive bears, but present in Ailurarctos lufengensis, Arctotheriini and Ursus. It is absent in Ursavus brevirhinus from Can Llobateres (Spain, MN9) (Crusafont Pairó and Kurtén 1976) but present in Ursavus tedfordi from Huaigou (Northwestern China, 8Ma) and Ursinae indet. from Lufeng (Southern China, dated from the late Baodean according to Dong and Qi 2013). In Indarctos the RPrd2.2 is variably present and always weak when present, while it is absent in Agriotherium. (Subdivided RPrd3 present: subdivided RPrd3 present, convergence, same direction).
Metaconid: The metaconid in Ailuropoda is generally simple. The posterior ridge (RMed2) occasionally develops into a small cusp. The RMed4 is not as distinct as in m1 but present in most specimens of Ailuropoda. In Ursus the RMed2 usually develops into a large cusp. The RMed4 is absent in most species of Ursus, but many specimens of Ursus deningeri do have a small cusp equivalent to the RMed4 of Ailuropoda. In Ailurarctos lufengensis the RMed2 does not develop into a cusp and the RMed4 is absent. (RMed2 cusp generally absent: RMed2 cusp generally present, development of RMed2 cusp in Ursus, uncorrelated direction; RMed4 present: RMed4 present, convergence, same direction).
Trigonid basin: The trigonid basin in Ailuropoda is filled with a large tubercle. There may be a groove near the antero-lingual corner separating the tubercle into two distinct parts. In Ailurarctos lufengensis the trigonid basin is not so bumped as in Ailuropoda, with several fine ridges on it. In Ursus deningeri, the basin is filled with several small tubercles. In Ursus etruscus the tubercles are also present but with a much smaller size and in a lower number. The tubercles in the trigonid basin are indistinct or absent in Ursavus tedfordi and more basal bears. (Tubercles in trigonid basin present: tubercles in trigonid basin present, convergence, same direction).
Hypoconid: The hypoconid in bears is relatively conservative like the protoconid, without further subdivision in most taxa. Besides the anterior and posterior ridges (RHyd1 and RHyd2), a separated antero-medial ridge (RHyd3) is also present. The RHyd3 in Ailuropoda is highly variable in form. It is often subdivided into several parallel ridges with various interruptions and connections. The RHyd3 is also greatly subdivided into several parts in Ursus deningeri (enthypoconid sensu Rabeder 1999). It is generally weakly separated in other Ursus species (though this character is variable, and a highly separated RHyd3 is occasionally seen in some Ursus species, such as Ursus etruscus). The RHyd3 is also subdivided into several branches in Ailurarctos lufengensis, but these ridges are still much thinner than those of Ailuropoda. Starting from the apex of the hypoconid, two fine ridges are present in Ailurarctos lufengensis, with one extending towards the RPrd2.2 and the other going transversely to the posterior entoconid. In Ursavus the RHyd3 is generally not distinct. It does not form a ridge in Ursavus tedfordi. (RHyd3 strongly subdivided: RHyd3 strongly subdivided, convergence, same direction).
Entoconid-complex: The situation of the entoconid-complex of m2 in Ailuropoda is very similar to that of m1. The anterior ridge develops into a separated cusp at a higher frequency than in m1. This is in accordance with the fact that the m2 has a proportionally longer talonid than the m1, and therefore has larger space to accommodate the bicuspid entoconid-complex. In Ailurarctos lufengensis, Ursavus tedfordi, and Ursus, there are two subequal-sized entoconids (the anterior one is the entoconid 2 and the posterior one is the entoconid 1). Though the entoconid 2 is present in all these taxa, only Ailuropoda displays the trend of merging this cusp with the entoconid1, so this character is viewed as derived in Ailuropoda. (Entoconid 2 tends to be absent: entoconid 2 present, reduction of entoconid 2 in Ailuropoda, uncorrelated direction)
m3.—The m3 of Ailuropoda is not elongated as in Ursus (Fig. 12). On the contrary, Ursus deningeri has an extremely elongated m3. The occlusal surface of the Ailuropoda m3 is not so different from other bears, except for the abundant tubercles. The ridge connecting the protoconid and the metaconid is not as high as in Hemicyoninae or more primitive bears, but nearly as high as other tubercles. This is the same as in Ursus, Ursavus, and Ailurarctos. The RPrd2.2 of m3 is also present in Ursus, Ursavus, and Ailurarctos. It is present in some specimens of Indarctos. (m3 not elongated: m3 elongated, elongation of m3 in Ursus deningeri, uncorrelated).
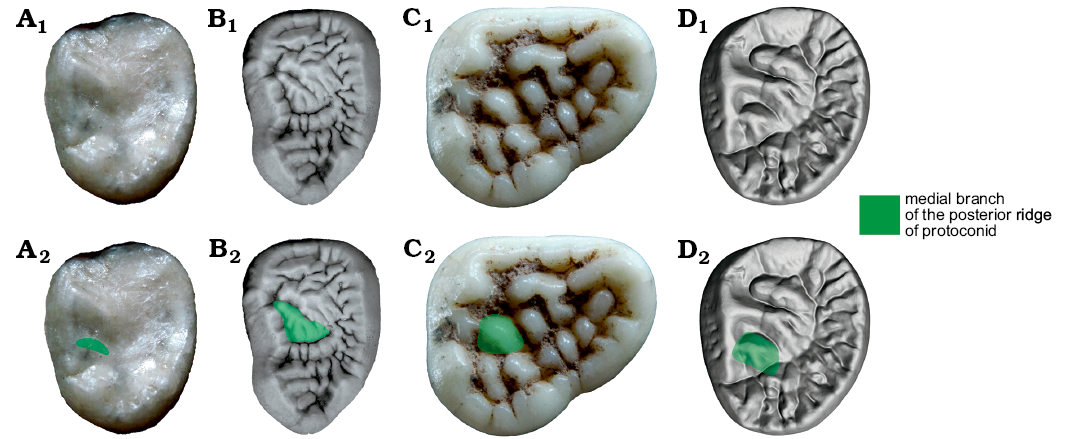
Fig. 12. Occlusal views of m3 homologous structures in cave bear (A, B) and giant panda (C, D) lineages. A. Ursavus tedfordi Qiu, Deng, and Wang, 2014 (HMV1453) from Huaigou, Gansu Province, Late Miocene. B. Ursus deningeri von Reichenau, 1904 (NMM1953/54, photo Jan Wagner) from Mosbach 2, Germany, early Middle Pleistocene. C. Ailuropoda melanoleuca (David, 1869) (IVPP V87025.362) from cave deposit of Guangxi Province, Late Pleistocene. D. Ailurarctos lufengensis Qiu and Qi, 1989 (IVPP V6892) from Lufeng, Yunnan Province, Latest Miocene. A1–D1, photographs; A2–D2, photographs with homologous structures indicated. Not to scale.
Discussion
Origin of the complicated occlusal structure of Ailuropoda.—As analysed above, Ailuropoda has a very complicated dental structure, but many characters and /or trends have already started in Ailurarctos. For a summary of the 45 characters see Tables 1, 2. Among these 45 molar characters, Ailuropoda has a total score of 33 (i.e., 33 characters from 45 are recognised here as derived characters). Among these 33 derived characters, 16 already appeared in Ailurarctos lufengensis, including the presence of a large parastyle, loss of the RPa3 (still present in Ailurarctos yuanmouensis) and the presence of the protocone in P4; the presence of the RPa1.2 of M2; the presence of the paraconid, hypoconid and entoconid in p4; the presence of the mesoconid, RPrd2.2, RPrd4, and REnd3 in m1; and the serrated anterior border, subdivided RPrd3, the presence of RHyd3 and the initial development of the tubercles in the trigonid and the talonid basin in m2. Among seven cheek teeth (P4–M2, p4–m3), the P4 of Ailurarctos lufengensis is the tooth that shows the highest similarity to that of Ailuropoda. The basic structure of the P4 in Ailurarctos lufengensis is already close to that of Ailuropoda. The only difference is that the whole tooth of Ailurarctos lufengensis is not as elongated as in Ailuropoda. The most conservative tooth of Ailurarctos is the M1. None of the eight derived characters of Ailuropoda is present in A. lufengensis. The M2 is also relatively conservative in Ailurarctos, with the only presence of a weak RPa1.2. The lower molars of Ailurarctos are more similar to those of Ailuropoda, and most characters of Ailuropoda already show their origin in A. lufengensis, though much less developed. The general dental structure of Ailurarctos lufengensis is already in the direction to Ailuropoda, especially for the premolars. Similar to Ailuropoda melanoleuca, Ursus deningeri also has a rather high score (31), indicating a complex dental structure. In contrast, the older species of the cave bear lineage, i.e. Ursus etruscus and Ursavus tedfordi, have a much lower score (17 and 3, respectively) than their contemporary species of Ailuropodinae Ailuropoda microta and Ailurarctos lufengensis (33 and 16, respectively). This indicates that the dental specialization of cave bears is relatively recent (from the late Early Pleistocene) and fast, whereas such a trend of complexification for occlusal dental structures appeared early in Ailuropodinae evolution and remained more stable since the beginning of the Quaternary.
Table 1. Summary and evaluation of morphology of Ailuropoda and Ursus deningeri. Abbreviations: +, present; –, absent; REnd3, medial ridge of entoconid; RHyd3, medial ridge of protoconid; RMed1, anterior ridge of metaconid; RMed2, posterior ridge of metaconid; RMe3/RMed3, medial ridge of metacone/metaconid; RMed4, postero-medial ridge of metaconid; RPa1.2, medial branch of anterior ridge of paracone; RPa3/RPad3, medial ridge of paracone/paraconid; RPrd2, posterior ridge of protoconid; RPrd2.2, medial branch of the posterior ridge of protoconid; RPrd3, postero-medial ridge of protoconid; RPrd4, postero-lateral ridge of protoconid.
|
Cheek teeth |
Characters |
Ailuropoda |
Ursus deningeri |
Origin of the characters |
Direction |
|
P4 |
parastyle |
+ |
– |
autapomorphy; plesiomorphy |
uncorrelated |
|
RPa3 |
– |
– |
convergence |
same direction |
|
|
protocone |
+ |
– |
reversal; plesiomorphy |
uncorrelated |
|
|
M1 |
parastyle |
+ |
+ |
convergence |
same direction |
|
metastyle |
– |
+ |
plesiomorphy; autapomorphy |
uncorrelated |
|
|
RPa1.2 |
+ |
– |
autapomorphy; plesiomorphy |
uncorrelated |
|
|
RPa3 |
+ |
– |
autapomorphy; plesiomorphy |
uncorrelated |
|
|
mesocone |
– |
+ |
plesiomorphy; autapomorphy |
uncorrelated |
|
|
entprotocone |
+ |
– |
autapomorphy; plesiomorphy |
uncorrelated |
|
|
entmetaconule |
+ |
– |
autapomorphy; plesiomorphy |
uncorrelated |
|
|
lingual cingulum |
wide |
thin |
autapomorphy; plesiomorphy |
uncorrelated |
|
|
M2 |
RPa1.2 |
+ |
+ |
convergence |
same direction |
|
separated RPa3 |
+ |
+ |
convergence |
same direction |
|
|
premetacone |
+ |
+ |
convergence |
same direction |
|
|
subdivided RMe3 |
+ |
– |
plesiomorphy; autapomorphy |
uncorrelated |
|
|
labial shifted protocone tip |
+ |
– |
plesiomorphy; autapomorphy |
uncorrelated |
|
|
metaconule |
subdivided |
subdivided |
convergence |
same direction |
|
|
entmetaconule |
+ |
– |
autapomorphy; plesiomorphy |
uncorrelated |
|
|
p4 |
paraconid |
+ |
+ |
convergence |
same direction |
|
hypoconid |
+ |
+ |
convergence |
same direction |
|
|
metaconid |
– |
+ |
autapomorphy; plesiomorphy |
uncorrelated |
|
|
entoconid |
+ |
+ |
convergence |
same direction |
|
|
premetaconid |
– |
+ |
autapomorphy; plesiomorphy |
uncorrelated |
|
|
RPrd2.2 |
– |
+ |
plesiomorphy; autapomorphy |
uncorrelated |
|
|
m1 |
RPad3 |
+ |
+ |
convergence |
same direction |
|
RPrd4 |
+ |
– |
autapomorphy; plesiomorphy |
uncorrelated |
|
|
RPrd2 |
– |
+ |
plesiomorphy; autapomorphy |
uncorrelated |
|
|
RPrd3 |
+ |
– |
plesiomorphy; autapomorphy |
uncorrelated |
|
|
RMed1 |
+ |
– |
autapomorphy; plesiomorphy |
uncorrelated |
|
|
RMed3 |
connected |
isolated |
plesiomorphy; autapomorphy |
uncorrelated |
|
|
RMed4 |
+ |
– |
autapomorphy; plesiomorphy |
uncorrelated |
|
|
mesoconid |
+ |
+ |
convergence |
same direction |
|
|
RPrd2.2 |
+ |
– |
autapomorphy; plesiomorphy |
uncorrelated |
|
|
enthypconid |
+ |
+ |
convergence |
same direction |
|
|
entoconid2 reduction trend |
+ |
+ |
convergence |
same direction |
|
|
entoconid2 reappearance |
– |
+ |
plesiomorphy; autapomorphy |
uncorrelated |
|
|
REnd3 |
+ |
– |
autapomorphy; plesiomorphy |
uncorrelated |
|
|
m2 |
middle contraction |
+ |
+ |
convergence |
same direction |
|
serrated anterior rim |
+ |
+ |
convergence |
same direction |
|
|
subdivided RPrd3 |
+ |
+ |
convergence |
same direction |
|
|
RMed2 cusp |
– |
+ |
plesiomorphy; autapomorphy |
uncorrelated |
|
|
RMed4 |
+ |
+ |
convergence |
same direction |
|
|
strong tubercles in trigonid basin |
+ |
+ |
convergence |
same direction |
|
|
entoconid2 reduction trends |
– |
+ |
autapomorphy; plesiomorphy |
uncorrelated |
|
|
strongly subdivided RHyd3 |
+ |
+ |
convergence |
same direction |
Table 2. Character matrix of giant panda, cave bear, and Ballusia lineages. Abbreviations: REnd3, medial ridge of entoconid; RHyd3, medial ridge of protoconid; RMed1, anterior ridge of metaconid; RMed2, posterior ridge of metaconid; RMe3/RMed3, medial ridge of metacone/metaconid; RMed4, postero-medial ridge of metaconid; RPa1.2, medial branch of anterior ridge of paracone; RPa3/RPad3, medial ridge of paracone/paraconid; RPrd2, posterior ridge of protoconid; RPrd2.2, medial branch of the posterior ridge of protoconid; RPrd3, postero-medial ridge of protoconid; RPrd4, postero-lateral ridge of protoconid.
|
Cheek teeth |
Characters |
Ailuropoda melanoleuca |
Ursus |
Ailuropoda |
Ursus |
Ailurarctos |
Ursavus |
Ballusia |
|
Total score |
|
33 |
31 |
33 |
17 |
16 |
3 |
0 |
|
P4 |
parastyle |
1 |
0 |
1 |
0 |
1 |
0 |
0 |
|
RPa3 |
1 |
1 |
1 |
1 |
1 |
1 |
0 |
|
|
protocone |
1 |
0 |
1 |
0 |
1 |
0 |
0 |
|
|
M1 |
parastyle |
1 |
1 |
1 |
1 |
0 |
0 |
0 |
|
metastyle |
0 |
1 |
0 |
1 |
0 |
0 |
0 |
|
|
RPa1.2 |
1 |
0 |
1 |
0 |
0 |
0 |
0 |
|
|
RPa3 |
1 |
0 |
1 |
0 |
0 |
0 |
0 |
|
|
mesocone |
0 |
1 |
0 |
1 |
0 |
0 |
0 |
|
|
entprotocone |
1 |
0 |
1 |
0 |
0 |
0 |
0 |
|
|
entmetaconule |
1 |
0 |
1 |
0 |
0 |
0 |
0 |
|
|
lingual cingulum |
1 |
0 |
1 |
0 |
0 |
0 |
0 |
|
|
M2 |
RPa1.2 |
1 |
1 |
1 |
1 |
1 |
0 |
0 |
|
separated RPa3 |
1 |
1 |
1 |
0 |
0 |
0 |
0 |
|
|
premetacone |
1 |
1 |
1 |
0 |
0 |
0 |
0 |
|
|
subdivided RMe3 |
0 |
1 |
0 |
1 |
0 |
0 |
0 |
|
|
labially shifted protocone tip |
0 |
1 |
0 |
1 |
0 |
0 |
0 |
|
|
metaconule |
1 |
1 |
1 |
1 |
0 |
0 |
0 |
|
|
entmetaconule |
1 |
0 |
1 |
0 |
0 |
0 |
0 |
|
|
p4 |
paraconid |
1 |
1 |
1 |
1 |
1 |
0 |
0 |
|
hypoconid |
1 |
1 |
1 |
0 |
1 |
0 |
0 |
|
|
metaconid |
0 |
1 |
0 |
0 |
0 |
0 |
0 |
|
|
entoconid |
1 |
1 |
1 |
0 |
1 |
0 |
0 |
|
|
premetaconid |
0 |
1 |
0 |
0 |
0 |
0 |
0 |
|
|
RPrd2.2 |
0 |
1 |
0 |
1 |
0 |
1 |
0 |
|
|
m1 |
RPad3 |
1 |
1 |
1 |
1 |
0 |
0 |
0 |
|
RPrd4 |
1 |
0 |
1 |
0 |
1 |
0 |
0 |
|
|
RPrd2 |
0 |
1 |
0 |
1 |
0 |
1 |
0 |
|
|
RPrd3 |
0 |
1 |
0 |
1 |
0 |
0 |
0 |
|
|
RMed1 |
1 |
0 |
1 |
0 |
0 |
0 |
0 |
|
|
RMed3 |
0 |
1 |
0 |
1 |
0 |
0 |
0 |
|
|
RMed4 |
1 |
0 |
1 |
0 |
1 |
0 |
0 |
|
|
mesoconid |
1 |
1 |
1 |
0 |
1 |
0 |
0 |
|
|
RPrd2.2 |
1 |
0 |
1 |
0 |
1 |
0 |
0 |
|
|
enthypoconid |
1 |
1 |
1 |
0 |
0 |
0 |
0 |
|
|
entoconid2 reduction trend |
1 |
1 |
1 |
1 |
0 |
0 |
0 |
|
|
entoconid2 reappearance |
0 |
1 |
0 |
0 |
0 |
0 |
0 |
|
|
REnd3 |
1 |
0 |
1 |
0 |
1 |
0 |
0 |
|
|
m2 |
middle contraction |
1 |
1 |
1 |
0 |
0 |
0 |
0 |
|
serrated anterior rim |
1 |
1 |
1 |
1 |
1 |
0 |
0 |
|
|
subdivided RPrd3 |
1 |
1 |
1 |
0 |
1 |
0 |
0 |
|
|
RMed2 cusp |
0 |
1 |
0 |
1 |
0 |
0 |
0 |
|
|
RMed4 |
1 |
1 |
1 |
0 |
0 |
0 |
0 |
|
|
strong tubercles in trigonid basin |
1 |
1 |
1 |
0 |
1 |
0 |
0 |
|
|
entoconid2 trends |
1 |
0 |
1 |
0 |
0 |
0 |
0 |
|
|
strongly subdivided RHyd3 |
1 |
1 |
1 |
0 |
1 |
0 |
0 |
Serial homology between the M1 and M2 and among the m1 to m3.—Serial homology is widely present in mammalian teeth (Van Valen 1993). In Insectivora, structures between M1 and M2 and among m1 to m3 are very similar. In primitive members of Carnivoraformes, however, the similarity tends to decrease due to the enlargement and modification of m1 and the reduction of posterior molars (M2/m2, M3/m3). Members of the crown group Ursidae have an enlarged m2 (and m3 in Ursus) and M2 talon, which provides enough space for the development of serial homology characters. Though appearing very different in shape, the basic elements in M1 and M2 are nearly the same. The only major difference is the elongation of the metaconule in M2. Other minor differences include the development of the parastyle and the metastyle in M1, and the connection of the protocone tip and the RMe3 in the M2 of some taxa. The lower molars display fewer similarities due to the reduction of the paraconid in both the m2 and m3, and the great reduction of the m3 talonid. In Ailuropoda, the molars are highly modified with tubercles, ridge and groove system (Davis 1964; Huang 1993), and both the M1 and M2 and the m1 to m3 show a very similar modification (Figs. 13, 14). The M1 and M2 share the presence of the RPa1.2, RPa3, RMe3, the entprotocone, and the entmetaconule. The m1 to m3 share the presence of the RPrd2, RPrd2.2, RMed1, RMed4, and RHyd3 (enthypoconid). In both upper and lower molars, the M2/m2 display the highest degree of complexity. The M1 is more simple than the M2 in the lesser degree of development of the RPa1.2, RPa3 and RMe3, the general lesser degree of subdivision of the entprotocone and the entmetaconule, and the absence of the premetacone. The m1 is more simple than the m2 in the opening of the trigonid basin, the lack of a connection between the paraconid and metaconid, and the weaker development of the RPrd2.2 and the RHyd3. The structures of the m3 are highly variable. Only RPrd2.2 is stable, all other structures being only occasionally present (e.g., RMed4, RHyd3) or always absent (e.g., entoconid).
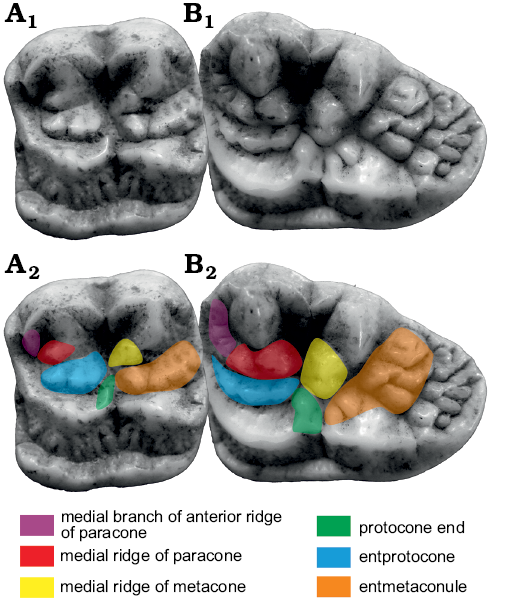
Fig. 13. Homologous structures of upper molars (occlusal views) of Ailuropoda melanoleuca (David, 1869) (A. IVPP V87025.109, B. IVPP V87025.153) from cave deposits of Guangxi Province, Late Pleistocene. Not to scale.
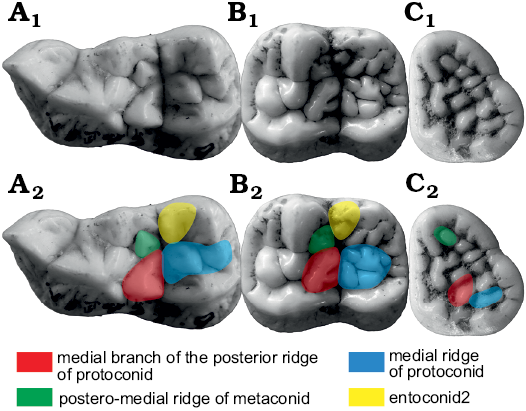
Fig. 14. Serial homology of lower molars (occlusal views) of Ailuropoda sp. (A. IVPP V13459.10, B. IVPP V13459.91, C. IVPP V87025.362) from various fossil sites in southern China, Pleistocene. A1–C1, photographs; A2–C2, photographs with homologous structures indicated. Not to scale.
Key characters with potential importance in understanding the bear evolution.—Through the comparison of the lineages of the giant panda and cave bear and homology clarification, some characters can be used to evaluate the evolutionary status of certain fossil taxa.
Ursavus tedfordi: The RPrd2.2 of the p4 is a synapomorphy for Ursini + Arctotheriini, supporting the presence of this character as ancestral for Ursini, as has been proposed by Qiu et al. (2014). The RPrd2.2s of the m2 and m3 are present in most specimens of Indarctos, Ailuropodini and Ursinae. This suggests these characters appeared even before the divergence of Ailuropodinae and Ursinae. Ursinae indet. from Lufeng (Qiu and Qi 1990) has distinct RPrd2.2s in m2 and m3, which suggests this bear is probably a small representative of the crown group Ursidae.
“Ursus minimus” from Moiseevka and Ursus abstrusus: The mandible from Moiseevka, Kazakhstan (MN14), was viewed as a special specimen of Ursus minimus (Baryshnikov and Lavrov 2013). According to the present analysis, the m2 of this specimen is very short, and the m1 retains a rudimentary RPrd3 and the typical bicuspid and ridge-like entoconid. These characters suggest a close relationship with Ursavus. The same is true for Ursus abstrusus from North America (Bjork 1970; Wang et al. 2017). These taxa should be viewed as relict representatives of the Ursavus evolutionary grade in the Pliocene.
Acknowledgments
We are greatly thankful to Zhanxiang Qiu (IVPP) for providing the unpublished lower m1 of Ailurarctos lufengensis, which is crucial for understanding the bear evolution. We are also greatly thankful to Jan Wagner (NMP) for in depth communication and discussion about the homology of bear dental characters and for providing a large number of photos of European material, including Ursus deningeri, Ursus etruscus, Ursus minimus, and Ursavus cf. brevirhinus for comparisons. We are thankful to Marisa Surovy, Eleanor Hoeger, and Sara Ketelsen (AMNH) for their help in accessing the AMNH modern mammal collections. We are thankful to Yemao Hou (IVPP) for helping with CT scanning of teeth of Ailurarctos lufengensis. We are also thankful Jan Wagner and an anonymous reviewer, whose suggestions greatly improved the quality of the manuscript. The present study is supported by the Strategic Priority Research Program of Chinese Academy of Sciences (grant XDB26000000), Chinese Natural Science Foundation Program (grants 41772018, 41872005, 41430102, 41872001), Key Frontier Science Research Program of the Chinese Academy of Sciences (grant QYZDY-SSW-DQC-22), Sino-Czech Joint Program (grant CAS-17-06), Administrative Committee of Dalian Puwan Economic Zone, Liaoning Province (The Comprehensive Research Project on Quaternary vertebrate fossils in Luotuo Hill from Dalian Puwan Economic Zone) and China Scholarship Council.
References
Abella, J., Alba, D.M., Robles, J.M., Valenciano, A., Rotgers, C., Carmona, R., Montoya, P., and Morales, J., 2012. Kretzoiarctos gen. nov., the oldest member of the giant panda clade. PLoS ONE 7: 1–7. Crossref
Abella, J., Montoya, P., and Morales, J. 2011. A New species of Agriarctos (Ailuropodinae, Ursidae, Carnivora) in the locality of Nombrevilla 2 (Zaragoza, Spain). Estudios Geológicos 67: 187–191. Crossref
Baryshnikov, G. 1998. Cave bears from the Paleolithic of the Greater Caucasus. Illinois State Museum Scientific Papers 27: 69–118.
Baryshnikov, G. 2007. Fauna of Russia and Neighbouring Countries. Mammals. Ursidae. 541 pp. Nauka, Saint Petersburg.
Baryshnikov, G.F. and Lavrov, A.V. 2013. Pliocene bear Ursus minimus Devèze de Chabriol et Bouillet, 1827 (Carnivora, Ursidae) in Russia and Kazakhstan. Russian Journal of Theriology 12: 107–118. Crossref
Beaumont, G.D. 1965. Contribution à l’etude genre Cephalogale Jourdan (Carnivora). Schweizerische Paläontologische Abhandlungen 82: 1–34.
Beaumont, G.D., 1982. Brèves remarques sur la dentition de certains Ursidés (Mammifères). Archives des Sciences 35: 153–156.
Bjork, P.R., 1970. The Carnivora of the Hagerman local fauna (late Pliocene) of southwestern Idaho. Transactions of the American Philosophical Society 60: 3–54. Crossref
Bocherens, H. 2015. Isotopic tracking of large carnivore palaeoecology in the mammoth steppe. Quaternary Science Reviews 117: 42–71. Crossref
Bocherens, H., Stiller, M., Hobson, K.A., Pacher, M., Rabeder, G., Burns, J.A., Tütken, T., and Hofreiter, M. 2011. Niche partitioning between two sympatric genetically distinct cave bears (Ursus spelaeus and Ursus ingressus) and brown bear (Ursus arctos) from Austria: isotopic evidence from fossil bones. Quaternary International 245: 238–248.
Crusafont Pairó, M. and Kurtén, B. 1976. Bears and bear-dogs from the Vallesian of the Vallés-Penedés basin, Spain. Acta Zoologica Fennica 144: 1–30.
Davis, D.D. 1964. The giant panda: a morphological study of evolutionary mechanisms. Fieldiana: Zoology Memoirs 3: 1–339. Crossref
De Bonis, L. 2013. Ursidae (Mammalia, Carnivora) from the Late Oligocene of the “Phosphorites du Quercy”(France) and a reappraisal of the genus Cephalogale Geoffroy, 1862. Geodiversitas 35: 787–814. Crossref
Dong, W. and Qi, G.-Q. 2013. Hominoid-Producing Localities and Biostratigraphy in Yunnan. In: X.M. Wang, L.J. Flynn, and M. Fortelius (eds.), Fossil Mammals of Asia, 293–313. Columbia University Press, New York. Crossref
Endo, H. 2001. Carpal bone movements in gripping action of the giant panda (Ailuropoda melanoleuca). Journal of Anatomy 198: 243. Crossref
Endo, H., Yamagiwa, D., Hayashi, Y., Koie, H., Yamaya, Y., and Kimura, J. 1999. Role of the giant panda’s “pseudo-thumb”. Nature 397: 309–310. Crossref
Erdbrink, D. 1953. A Review of Fossil and Recent Bears of the World with Remarks on their Phylogeny Based on their Dentition. 597 pp. Drukkerij Jan de Lange, Deventer.
Geraads, D. and Spassov, N. 2016. Musteloid carnivores from the upper Miocene of South-Western Bulgaria, and the phylogeny of the Mephitidae. Geodiversitas 38: 543–558. Crossref
Ginsburg, L. and Morales, J. 1998. Les Hemicyoninae (Ursidae, Carnivora, Mammalia) et les formes apparentées du Miocène inférieur et moyen d’Europe occidentale. Annales de Paléontologie 84: 71–123. Crossref
Gregory, W.K. 1936. On the phylogenetic relationships of the giant panda (Ailuropoda) to other arctoid Carnivora. American Museum Novitates 878: 1–29.
Huang, W.B. 1993. The skull, mandible and dentition of giant pandas (Ailuropoda): morphological characters and their evolutionary implications. Vertebrata PalAsiatica 31: 191–207.
Jiangzuo, Q.G., Cong, H.L., Ma, R., Feng, H., and Liu, J.Y. 2014. Middle and Late Pleistocene Sun Bears (Ursidae, Carnivora) from Southern China. In: W. Dong (ed.), Proceedings of the Fourteenth Annual Meeting of the Chinese Society of Vertebrate Paleontology, 119–134. China Ocean Press, Beijing.
Mazza, P. and Rustioni, M. 1992. Morphometric revision of the Eurasian species Ursus etruscus Cuvier. Palaeontographia Italica 79: 101–146.
Osborn, H.F. 1907. Evolution of Mammalian Molar Teeth. 250 pp. Macmillan, New York.
Pacher, M. 2017. Anterior premolar variability in Pleistocene cave and brown bears and its signifi cance in species determination. Fossil Imprint 73: 482–494. Crossref
Petter, G. and Thomas, H. 1986. Les Agriotheriinae (Mammalia, Carnivora) néogènesde l’Ancien Monde présence du genre Indarctos dans la faune de Menacer (Ex-Marceau), Algérie. Geobios 19: 573–586. Crossref
Qiu, Z.X. and Qi, G.Q. 1989. Ailuropod found from the Late Miocene deposits in Lufeng, Yunnan. Vertebrata PalAsiatica 27: 153–169.
Qiu, Z.X. and Qi, G.Q. 1990. Restudy of mammalian fossils referred to Ursinae indet. from Lufengpithecus locality. Vertebrata Palasiatica 4: 270–283.
Qiu, Z.X. and Schmidt-Kittler, N. 1983. Agriotherium intermedium (Stach, 1957) from a Pliocene fissure filling of Xiaoxian County (Anhuei Province, China) and the phylogenetic position of the genus. Palaeovertebrata 13: 65–81.
Qiu, Z.X., Deng, T., and Wang, B.Y. 2014. A Late Miocene Ursavus skull from Guanghe, Gansu, China. Vertebrata PalAsiatica 52: 265–302.
Rabeder, G. 1999. Die Evolution des Höhlenbärengebisses. Mitteilung der Kommission für Quartärforschung der Österreichischen Akademie der Wissenschaften 11: 1–102.
Sicher, H. 1944. Masticatory apparatus in the giant panda and the bears. Fieldiana: Zoological Series 29: 61–73. Crossref
Tedford, R.H. and Martin, J. 2001. Plionarctos, a tremarctine bear (Ursidae: Carnivora) from western North America. Journal of Vertebrate Paleontology 21: 311–321. Crossref
Thenius, E. 1979. Zur systematischem und phylogenetischen Stellung des Bambusbären: Ailuropoda melanoleuca David (Carnivora, Mammalia). Zeitschrift Für Säugetierkunde 44: 286–305.
Torress Perezhidalgo, T.D. 1992. The European descendants of Ursus etruscus C. Cuvier (Mammalia, Carnivora, Ursidae). Boletíno Geológico y Minero 103: 632–642.
Ungar, P.S. 2010. Mammal Teeth: Origin, Evolution, and Diversity. 320 pp.The Johns Hopkins University Press, Baltimore.
Van Heteren, A.H., Arlegi, M., Santos, E., Arsuaga, J.-L., and Gómez-Olivencia, A. 2018. Cranial and mandibular morphology of Middle Pleistocene cave bears (Ursus deningeri): implications for diet and evolution. Historical Biology 31: 1–15. Crossref
Van Valen, L.M. 1993. Serial homology: The crests and cusps of mammalian teeth. Acta Palaeontologica Polonica 38: 145–158.
Wagner, J. and Čermák, S. 2012. Revision of the early Middle Pleistocene bears (Ursidae, Mammalia) of Central Europe, with special respect to possible co-occurrence of spelaeoid and arctoid lineages. Bulletin of Geosciences 87: 461–496. Crossref
Wang, X.M., Rybczynski, N., Harington, C.R., White, S.C., and Tedford, R.H. 2017. A basal ursine bear (Protarctos abstrusus) from the Pliocene High Arctic reveals Eurasian affinities and a diet rich in fermentable sugars. Scientific Reports 7: 1–14. Crossref
Acta Palaeontol. Pol. 64 (4): 693–710, 2019
https://doi.org/10.4202/app.00629.2019