

An early Eocene pan-gekkotan from France could represent an extra squamate group that survived the K/Pg extinction
ANDREJ ČERŇANSKÝ, JUAN D. DAZA, RODOLPHE TABUCE, ELIZABETH SAXTON, and DOMINIQUE VIDALENC
Čerňanský, A., Daza, J.D., Tabuce, R., Saxton, E., and Vidalenc, D. 2023. An early Eocene pan-gekkotan from France could represent an extra squamate group that survived the K/Pg extinction. Acta Palaeontologica Polonica 68 (4): 695–708.
In this paper we describe a new lizard from the early Eocene of the Cos locality in the Quercy region (near the Caylus village, Southwest France). The age of the Cos deposit has been proposed as the MP 10–11 interval, close to the transition of the late Ypresian to early Lutetian. The fossil material includes a nearly complete right maxilla and a large section of the right dentary, both elements attributed to Pan-Gekkota. These specimens are morphologically different from crown gekkotans, therefore, we describe them as a new species. Some aspects of the maxilla are very atypical regarding geckos (e.g., the shape of the facial process). The posterior margin of the facial process slopes down gradually dorsoventrally towards the jugal facet, reaching the posterior end of the maxilla, in contrast to gekkotans, where the facial process ends anterior to the posterior end of the maxilla. A similar maxilla is present in the Late Jurassic–Early Cretaceous pan-gekkotan genus Eichstaettisaurus. This suggests that the new fossil taxon represents either a lineage that persisted from the Mesozoic to the early Eocene in Europe, or perhaps a morphology otherwise unrepresented in crown gekkotans. We allocate this taxon provisionally to Pan-Gekkota, and contribute to increase the diversity of this clade in Western Europe during the Paleogene, which now includes the stratigraphically similarly aged Laonogekko lefevrei from France (MP 10), and older Dollogekko from Belgium (MP 7).
Key words: Squamata, Pan-Gekkota, Mesozoic, K/Pg extinction, Paleogene, Phosphorites du Quercy, France.
Andrej Čerňanský [cernansky.paleontology@gmail.com; ORCID: https://orcid.org/0000-0001-8920-2503 ], Department of Ecology, Laboratory of Evolutionary Biology, Faculty of Natural Sciences, Comenius University in Bratislava, Mlynská dolina, 84215, Bratislava, Slovakia.
Juan D. Daza [juand.daza@gmail.com; ORCID: https://orcid.org/0000-0002-5651-0240 ] and Elizabeth Saxton [eas109@shsu.edu; ORCID: https://orcid.org/0009-0004-5137-1685 ], Department of Biological Sciences, Sam Houston State University 2000 Avenue I Life Sciences Building, Room 105 Huntsville, TX 77341, USA.
Rodolphe Tabuce [Rodolphe.Tabuce@umontpellier.fr; ORCID: https://orcid.org/0000-0002-4713-3981 ], ISEM, Université de Montpellier, CNRS, IRD, EPHE, Cc 064, Place Eugène Bataillon, 34095 Montpellier Cedex 5, France.
Dominique Vidalenc [vidalenc.dominique@wanadoo.fr], 103 Avenue François Mitterand, 31800 Saint-Gaudens, France.
Received 31 May 2023, accepted 18 September 2023, available online 13 November 2023.
Copyright © 2023 A. Čerňanský et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
In this paper we describe a new pan-gekkotan lizard from the Cos locality (44°13’11.20”N, 1°44’58.21”E) in France. The Cos fissure was first discovered in Quercy in 1865 by Jean André Poumarède (Pulou 1980), but recently, one of us (DV) discovered vertebrate fossils in this locality. Regarding lizards, only anguimorphs were known from this site (Čerňanský et al. 2023; see this paper for the geological setting and a map). The Cos fissure is the seventh pre-early Eocene locality of Phosphorites du Quercy known and is one of the oldest; estimates using fossil primates (Godinot et al. 2021) and fossil rodents (Vianey-Liaud et al. 2022), place the vertebrate fauna of the Cos site in the period MP 10–11, which spans the late Ypresian and early Lutetian. Thus, it corresponds to the Early Eocene Climatic Optimum (EECO; see Inglis et al. 2020) which is estimated to be 53.3–49.1 Ma. The EECO was a time of major climatic and environmental change caused by perturbations to the global carbon cycle (Hyland et al. 2017). These environmental conditions make the Cos locality important because the early Eocene terrestrial fossil record in Europe is poorly known. The early Eocene is interesting because the climate warmed rapidly and intensely. In fact, the warmest global climates of the past 66 My occurred during the early Eocene epoch (about 56–48 Ma) and arguably it represents the warmest time interval since the Permian–Triassic mass extinction and the Early Triassic (Cramwinckel et al. 2018). During the early Eocene, megathermal floral elements, including palms, were present even in Antarctica (Pross et al. 2012; Greenwood and Wing 1995) and snakes, turtles and alligators are preserved in Canada, in regions that were well above the Arctic Circle (Estes and Hutchinson 1980; Eberle et al. 2010, 2014; Smith and Georgalis 2022). However, much crucial information about the transition between the older Paleocene and later Eocene periods remains unknown.
The Pan-Gekkota clade includes a few fossil groups and the crown group Gekkota. The latter is a clade that includes 2265 extant species (Meiri 2020; Uetz et al. 2020, 2023) represented by limbed and limbless geckos (a.k.a. pygopods). The fossil record of Pan-Gekkota goes back at least to the late Early Cretaceous. Although several Jurassic taxa have been suggested in the past as being related to or even belonging to Gekkota; Eichstaettisaurus Kuhn, 1958, might be the only known Jurassic stem-gekkotan (e.g., Gauthier et al. 2012; Pyron 2017; Tałanda 2018; Simões and Pyron 2021). It should be noted, furthermore, that an, at least, Middle Jurassic date for the split of the total-group Pan-Gekkota lineage, from other crown squamates, was proposed by recent studies (Pyron 2017; Simões et al. 2020; Simões and Pyron 2021). Simões et al. (2018) estimated the divergence date to be as early as Late Triassic. This early estimate relies on the interpretation of Middle Jurassic parviraptorids as snakes, and the fossils Sophineta Evans and Borsuk-Białynicka, 2009 (Early Triassic), Megachirella Renesto and Posenato, 2003 (Middle Triassic), and Marmoretta Evans, 1991 (Middle Jurassic) as part of Squamata (Simões et al. 2018). Recent reanalysis of as Marmoretta confirms its original placement as a stem lepidosauromorph, and recovers Sophineta and Megachirella on the Squamata stem with at best low nodal support (clade credibility of 0.08 and 0.36, respectively, Griffiths et al. 2021). The phylogenetic positions of these groups are highly unstable and have been interpreted in different positions (Evans 1994, 2022; Renesto and Bernardi 2014; Caldwell et al. 2015; Simões et al. 2018; Griffiths et al. 2021), their use as calibration points should be treated cautiously in time-tree analyses.
Some of the oldest pan-gekkotans, the Early Cretaceous Norellius Conrad and Daza, 2015 (Conrad and Norell 2006), and Hoburogekko Alifanov, 1989 (Daza et al. 2012); the Late Cretaceous Gobekko Borsuk-Białynicka, 1990 (Daza et al. 2013), include articulated and very complete skulls. Astonishing specimens with superb preservation of skeletal and soft tissue are also reported in amber from the Cretaceous of Myanmar (Arnold and Poinar 2008; Daza et al. 2016, Fontanarrosa et al. 2018), however the Cenozoic fossil record of these successful and cosmopolitan lizards is generally scarce (Estes 1983; Müller and Mödden 2001; Daza et al. 2014; Čerňanský et al. 2018). Paleogene fossils are mainly known from Europe (Augé 2003, 2005; Estes 1983; Daza et al. 2014; Bolet 2017; Georgalis et al. 2021; Čerňanský et al. 2022), but most of the material is represented by isolated bones (mainly dentaries, frontals, and vertebrae). One exception is a specimen in Baltic amber from the Eocene of north-western Russia described by Bauer et al. (2005) as Yantarogekko balticus Bauer, Böhme, and Weitschat, 2005. The integument is preserved in pristine condition, but the specimen has no skeletal elements, as revealed by micro-CT data of the specimen (Johannes Müller, personal communication 2017). The age of Baltic amber is not certain, and its accumulation was not the result of a single, long-term process during the Lutetian–Priabonian, but was repeated independently in two different stages, Bartonian and Priabonian, respectively (Bukejs et al. 2019). Besides this single Baltic amber specimen, gekkotans are also present in amber from the Miocene of the Dominican Republic, where they are represented by multiple specimens that have been referred to two species of the extant sphaerodactylid genus Sphaerodactylus Wagler, 1830 (Böhme 1984; Daza and Bauer 2012; Daza et al. 2013) although the diversity of species in Dominican amber is higher (JDD personal observation).
Our understanding of the paleobiodiversity of geckos during the early Eocene is very limited. Rare exceptions, where these lizards have been described, are from the locality of Dormaal (MP 7) in Belgium (Dollogekko dormaalensis Čerňanský, Daza, Smith, Bauer, Smith, and Folie, 2022 and Gekkota indet., Augé 2005; Čerňanský et al. 2022) and Silveirinha (~MP 7) in Portugal (Gekkota indet., Rage and Augé 2003). The early Eocene taxa also include Laonogekko lefevrei Augé, 2003, described from the late early Eocene of Prémontré (MP 10, Paris Basin, France). Other Paleogene exceptions from the post-early Eocene include a partly preserved, but undescribed, specimen from Messel (MP 11; Smith et al. 2018) and a sphaerodactylid, Geiseleptes delfinoi Villa, Wings, and Rabi, 2022, from Geiseltal (MP 12 and MP 13, respectively; Villa et al. 2022). Other middle Eocene gekkotans are represented by Rhodanogekko vireti described by Hoffstetter (1946) from Lissieu (MP 14, eastern France). This taxon is based on a single frontal. The late Eocene–early Oligocene gekkotans include Cadurcogekko Hoffstetter, 1946, from localities within the Phosphorites du Quercy, France (MP 16–19; Hoffstetter 1946; Augé 2005; Georgalis et al. 2021). Two species of this genus are currently regarded as valid: the type species, Cadurcogekko piveteaui Hoffstetter, 1946, and Cadurcogekko verus Bolet, Daza, Augé, and Bauer, 2015 (late Eocene MP 17; Bolet et al. 2015). Note that Cadurcogekko rugosus Augé, 2005, has recently been reidentified as a scincid and accordingly placed in its own genus, Gekkomimus Bolet, Daza, Augé, and Bauer, 2015 (see Bolet et al. 2015). Recently, new well-preserved material of C. cf. piveteaui has been described from the Phosphorites du Quercy (Georgalis et al. 2021).
Nomenclatural acts.—This published work and the nomenclatural acts it contains have been registered in ZooBank: http://zoobank.org/urn:lsid:zoobank.org:pub:A660D450-F907-40F5-A602-610099AE70B3.
Institutional abbreviations.—BSPG, Bayerische Staatssammlung für Paläontologie und Historische Geologie, München, Germany; CAS, California Academy of Sciences, USA; GMH, “Geiseltalmuseum Halle” = Geiseltal Collection, Martin Luther University Halle-Wittenberg, Germany; MCZ, Museum of Comparative Zology, Harvard University, Cambridge, Massachusetts, USA; MNHN, Muséum national d’Histoire naturelle, Paris, France; NHMW, Naturhistorisches Museum Wien, Austria; UPRRP-RT, Richard Thomas collection, Zoology Museum at University of Puerto Rico, Río Piedras Campus, Puerto Rico; UM-COS, Cos collection, Université de Montpellier, France.
Material and methods
The studied material is housed at the University of Montpellier, cataloged under individual UM-COS-numbers. The fossil material was imaged using the micro-CT facility at the Slovak Academy of Sciences in Banská Bystrica, using a Phoenix micro-CT v|tome|x L240. The settings were as follows: Voxel size = 0.00700002; Current = 230; Voltage = 80; Inttime = 20000; Average = 2; Steps = 1300. The images were recorded over 360°. The CT data were analyzed using VG Studio Max 3.1. and Avizo 8.1. Photographs of the Cos material were taken with a Leica M205 C binocular microscope with an axially mounted DFC 290 HD camera; software: LAS (Leica Application Suite) 4.1.0 (build 1264). GE Phoenix nanotom VR 180 X-ray tomography nano-CTVR system at the Slovak Academy of Sciences in Bratislava was used (Fairfield, CT) for μCT scanning of the left maxilla (NHMW 2019/0052/0001) of Cadurcogekko cf. piveteaui (previously published and figured by Georgalis et al 2021: figs. 3, 4, mirror reversed here due to consistency with other maxillae). The CT data was analyzed using Avizo 8.1 and Avizo lite (Thermo Fisher Scientific). The outline figure of the maxilla of the holotype (BSPG 1937 I) of Eichstaettisaurus schroederi was drawn from photographs published by Simões et al. (2017: fig. 3). The holotype (MNHN PMT 5) of Laonogekko lefevrei was drawn from figure published by Augé (2005: fig. 66b). The right maxilla of the holotype (GMH Ce IV-4057-1933) of Geiseleptes delfinoi was drawn based on CT-scan image published by Villa et al. (2022: fig. 3b). The right maxilla (MNHN QU 17734) of Cadurcogekko piveteaui was drawn based on figure published by Augé (2005: fig. 59a).
Comparative CT data was accessed from morphosource: Sphaerodactylus grandisquamis spanius Stejneger, 1904 (RT 14708; Sphaerodactylidae), Holodactylus africanus Boettger, 1893 (CAS 198932; Eublepharidae), Paradelma orientalis (Günther, 1876) (CAS 77652; Pygopodide), Underwoodisaurus milii Bory de Saint-Vincent, 1823 (CAS 74744; Carphodactylidae), and Pseudothecadactylus australis (Günther, 1877) (MCZ Herp R- 35162; Diplodactylide).
Scanning Electron Microscopy of modern geckos Gonatodes albogularis (Duméril and Bibron, 1836) and Sphaerodactylus klauberi Grant, 1931, were acquired in a Hitachi SU3500 scanning electron microscope using secondary electron detector. Samples were adhered to the SEM stub using carbon tape and sputter coated with gold for 60 sec (~200A) using a Cressington 108. The sputter-coated samples were imaged under a high vacuum at 10 kV.
Systematic palaeontology
Squamata Oppel, 1811
Pan-Gekkota Bauer, 2020
Genus Bauersaurus nov.
Zoobank LSID: urn:lsid:zoobank.org:act:94D1D3D6-2884-4B7E-BD 55-868934FF9ACC
Type species: Bauersaurus cosensis sp. nov.; by monotypy; see below.
Etymology: A combination of the last name of the North American herpetologist Aaron M. Bauer, to recognize his valuable contributions to squamate morphology, taxonomy, and systematics, especially on geckos, and the Greek saura (σαύρα), lizard. The name is masculine.
Diagnosis.—As for Bauersaurus cosensis gen. et sp. nov., by monotypy.
Bauersaurus cosensis sp. nov.
Figs. 1–4.
Zoobank LSID: urn:lsid:zoobank.org:act:94D1D3D6-2884-4B7E-BD 55-868934FF9ACC
Etymology: In reference to the locality of Cos in France.
Holotype: UM-COS-1012, right maxilla (Figs. 1, 3A, 4A, 5A, and Fig. 6 partly—the maxilla at the bottom).
Type locality: Cos, fissure-filling in the Quercy region (Southwest France). GPS coordinates: 44°13’11.20” N; 1°44’58.21” E.
Type horizon: Uppermost lower Eocene (MP 10–11).
Material.—The holotype and UM-COS-1013, right dentary, both from the type locality and horizon.
Diagnosis.—The new taxon differs from all fossil and extant taxa of Pan-Gekkota in the following combination of features from the maxilla: (i) posterior margin of facial process slopes down gradually dorsoventrally to the jugal facet, contra to all known gekkotans where the facial process ends more anteriorly, defining a well-developed posterior process with more or less parallel dorsal and ventral margins. In extant gekkotans the posterior margin of the facial process is highly variable, being mainly gradually sloped (diplodactylids), sometimes sinusoid (some carphodactylids and pygopodids), tall with a stepped posterior margin (eublepharids), mainly narrow and tall (sphaerodactylids), concave posteriorly (phyllodactylids), and combinations of the former (gekkonids); (ii) posterior margin of the facial process ends in a short, posteriorly oriented, and well defined process (posterior free terminus) vs. Eichstaettisaurus schroederi (Broili, 1938), Laonogekko lefevrei, Cadurcogekko piveteaui, Geiseleptes delfinoi, extant gekkotans. Compared with described fossils, the maxilla of L. lefevrei has a low sloping posterior margin, that is still more inclined relative to the Cos taxon; vs. a steep margin in Cadurcogekko piveteaui; (iii) the facial process length is at least twice its height (although its tip is clearly broken) similar to Eichstaettisaurus schroederi, and ?Cadurcogekko piveteaui, differing from L. lefevrei, Geiseleptes delfinoi, most extant gekkotans except long and depressed snouted forms like species of the gekkonid Uroplatus Duméril, 1806; (iv) the jugal facet is mostly dorsolateral in orientation, suggesting that the jugal was large and well braced to the maxilla, differing from extant gekkotans which have a very reduced jugal that contacts usually the dorsal surface of the maxilla; (v) the ventral surface of the maxillary medial shelf has a trough on the lateral side (as Aeluroscalabotes felinus (Günther, 1864); vs. most other geckos). On the medial side, this fossa is bordered by a sharp low ridge, which is reduced in its anterior part. In the posterior region, however, the ridge of the maxillary medial shelf projects ventrally to form a vertical bony septum (almost vertical wall) which covers the tooth bases of the posterior teeth medially; (vi) the external surface of the facial process is smooth; as Geiseleptes delfinoi; vs. Cadurcogekko piveteaui and Cadurcogekko verus, Laonogekko lefevrei (faintly sculptured), and similar to most gekkotans where the maxilla is smooth, except the rugose maxilla in Matoatoa brevipes (Mocquard, 1900), grooved maxilla of Hemidactylus turcicus (Linnaeus, 1758), and the pitted maxilla of Blaesodactylus antongilensis (Böhme and Meier, 1980) and Chondrodactylus briboni (Smith, 1846) (Glynne et al. 2020); (vii) maxilla with six supralabial foramina that increase in diameter gradually and are located in the anterior two thirds of the bone, the posterior section lacks a foramen vs. Laonogekko lefevrei and Geiseleptes delfinoi, these taxa have six as well, but the posteriormost one is located on the posteroventral process. Higher number of more posteriorly distributed foramina are present in Cadurcogekko piveteaui and Cadurcogekko verus, species of Gekko Laurenti, 1768, Euleptes Fitzinger, 1843, Eublepharis Gray, 1827, Rhacodactylus Fitzinger, 1843, and Saltuarius Couper, Covacevich, and Moritz, 1993; (viii) anterior margin of the maxilla with a free rounded and triangular tip (as in Laonogekko lefevrei, Cadurcogekko piveteaui, extant and extinct Euleptes, e.g., Euleptes europaea (Gené, 1839) and Euleptes klembarai Čerňanský, Daza, and Bauer, 2018); (ix) transversally bicuspid teeth with labial and lingual cutting edge running mesiodistally and bent lingually (V-shaped in ventral view), separated by a concave sulcus (as in Eublepharis macularius [Blyth, 1854], in Gonatodes albogularis, Coleonyx elegans Gray, 1845, Oedura tryoni de Vis, 1884, Aeluroscalabotes felinus, Geiseleptes delfinoi; Gekkota indet 2. from Dormaal [Čerňanský et al. 2022]; vs. Laonogekko lefevrei, Euleptes europaea, Gekko gecko [Linnaeus, 1758], Rhacodactylus leachianus [Cuvier, 1829]).
Description.—The holotype UM-COS-1012 represents a nearly complete right maxilla (Figs. 1, 3A). It is a long element, straight and lightly built. The anteroposterior maximum length is 10 mm. It consists of two major portions: the alveolar portion bearing the dentition and the dorsally extending facial process. An almost complete tooth row is preserved, excepting the anterior most region. The dental crest (labial wall) is deep, and the maxilla bears 36 tooth loci (the total number was probably slightly higher), preserving 12 complete teeth. The premaxillary process is damaged and the anterior opening of the superior alveolar canal is exposed. The lateral surface of the maxilla is pierced by six supralabial foramina, which slightly increase in size posteriorly. They are located along the ventral margin in the anterior two thirds of the bone, where the posteriormost one (also the largest) is located at the level of the 16th tooth position (counted from posterior). Posterior to this last foramen, there is a long section of the facial process without any foramen. Dorsal to this series (on the facial process), the surface is pierced by additional eleven, irregularly arranged small foramina. Some of them (those located in the posterodorsal region) are accompanied by short grooves running posterodorsally from the foramen. The facial process forms almost a vertical wall, being roughly trapezoidal in shape (notice that the posterodorsal margin of the facial process is missing). The dorsal margin is angled slightly medially, this section bears a narrow longitudinal facet for the nasal (and maybe frontal) visible in medial view. The process is markedly long anteroposteriorly. Its posterior margin gradually slopes ventrally at a sharp angle (28°) whereas the anterior margin is more ventrally sloped relative to the posterior one. The posterior margin of the facial process terminates in short, free, posterior process. This process is well separated from the alveolar margin by a small rounded notch. The anterior margin possesses a free terminus. It forms a blunt triangular tip (Fig. 1A1; this feature can be seen in extant and extinct species of the Euleptes but also in other taxa such as Hemidactylus Oken, 1817, Tarentola Gray, 1825, Dactylocnemis Steindachner, 1867 and Gekko; see e.g., Čerňanský et al. 2018; Villa et al. 2018). In medial view, there is a fine ridge in the anterior region of the facial process that runs posterodorsally. This ridge originates from the maxillary medial (supradental) shelf at the level of the fourth preserved tooth position (counted from anterior). The internal side of the facial process posterior to this ridge is excavated, forming a cavity. The most prominent structure on the medial side is the maxillary medial shelf. It is relatively thin, but with a marked medial expansion. The maximal expansion is at the level of the large opening for the superior alveolar canal (and the level of the 16th tooth position, counted from posterior); at this expansion, the maxilla would have join the palatine. The posterior section of the maxillary medial shelf protrudes ventrally to form a deep vertical bony septum (almost vertical wall) which covers the tooth bases of the posterior teeth medially. Counting from posterior to anterior, the septum starts to deepen ventrally at the level of the 13-tooth locus and reaches its maximal projection at the level of the ninth to fourth tooth positions. Further, the septum diminishes posteriorly. Thus, its ventral margin is roughly concave. It should be noted, however, that anterior to this vertical septum, there is a trough (sensu Grismer 1988) clearly visible in ventral view. This trough is bordered laterally by the dental crest supporting the teeth and medially by a distinct low ridge. Thus, the wall in the posterior region is only a continuation of this ridge running almost along the entire length of the maxillary medial shelf (although it is less distinct in the anterior region). The above-mentioned opening of the superior alveolar canal is located on the dorsal surface of the shelf approximately in its mediolateral mid-line. Laterally, this superior alveolar foramen is bordered by a sharp horizontal lip of bone. Posteriorly, a groove runs from the foramen. The groove turns and continues on the lateral surface, at the level of the posterior free terminus of the facial process. Here, the bottom of the groove is pierced by a small foramen. At this position, a large facet for jugal is visible, which suggests that the jugal had a strong connection with the maxilla including some extension onto the lateral surface of the maxilla.
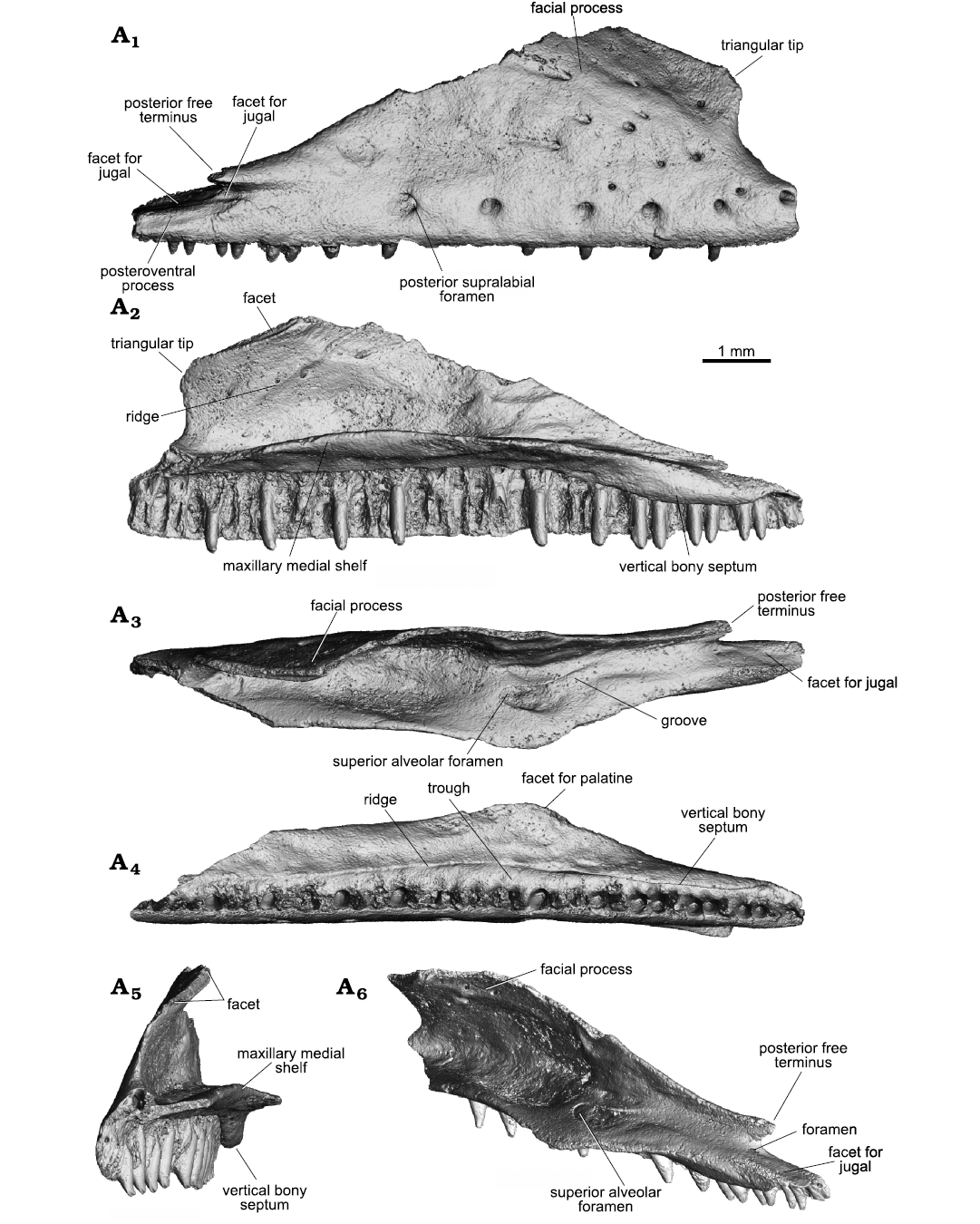
Fig. 1. Pan-gekkotan squamate Bauersaurus cosensis gen. et sp. nov. from Cos, France, early Eocene (MP 10–11). Holotype (UM-COS-1012), right maxilla, in lateral (A1), medial (A2), dorsal (A3), ventral (A4), anteromedial (A5), and dorsomedioposterior (A6) views (all micro-CT visualizations).
UM-COS-1013 represents the posterior portion of the right dentary (Figs. 2, 3B). It has a robust appearance and preserves 19 tooth loci (with two teeth still attached). Based on this preserved portion it can be estimated that the dentary, when complete, was long and tubular, with a completely enclosed Meckelian canal, as in crown gekkotans, contrary to the Cretaceous gekkonomorph Hoburogekko suchanovi, in which the borders of the Meckelian canal are partially fused (Alifanov 1989; Daza et al. 2012). The posterior region has a wedge-shaped opening of the Meckelian canal. At first glance, it seems as though some remains of the splenial are ankylosed to the dentary, but what is visible is the anterior part of the facet of the splenial. This is consistent with observation in extant gekkotans where: (i) the splenial is recessed onto the dentary, creating a flush profile; (ii) the facet indicates clearly the splenial abutment with the anteromedial process of the coronoid, and the coronoid is certainly missing, and (iii) there is no clear indication of the anterior dental or the anterior mylohyoid foramina, which are usually in the anteromedial portion of the splenial. The preserved splenial facet indicates that the anterior portion has the characteristic triangular shape of crown gekkotans. The dental crest is tall, but its height does not exceed the height of the ventral portion of the dentary (the alveolar part of the dentary is less than half the total height of the dentary). The subdental shelf is well-developed, moderately expanded medially, forming a clearly visible surface in dorsal view. In the posterior region, it bears a partly preserved facet for the coronoid. The otherwise smooth external surface of the preserved portion is pierced by one elliptical labial foramen. The foramen is in the midline of the bone. The dentary is broken posteriorly, in the region where the facet for the anterolateral process of the coronoid would be visible, a coronoid clasping the dentary on both the lateral and medial sides of the dentary can be used to differentiate gekkotans from skinks (Daza et al. 2015), although these facets, especially the one for the anteromedial process of the splenial can be found in the dentary of almost any lizard.
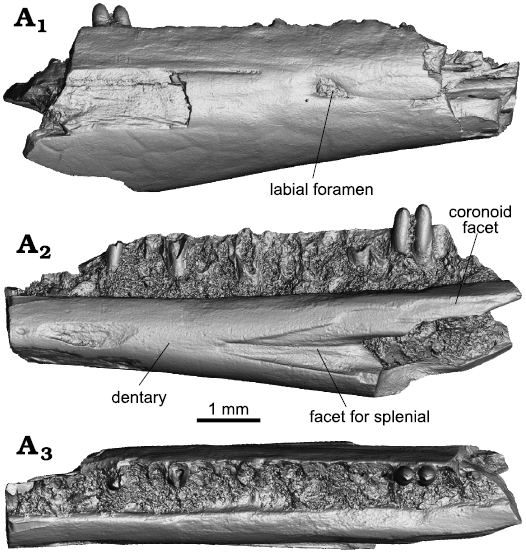
Fig. 2. Pan-gekkotan squamate Bauersaurus cosensis gen. et sp. nov. from Cos, France, early Eocene (MP 10–11). UM-COS-1013, right dentary, in lateral (A1), medial (A2), dorsal (A3) views (all micro-CT visualizations).

Fig. 3. Pan-gekkotan squamate Bauersaurus cosensis gen. et sp. nov. from Cos, France, early Eocene (MP 10–11). Photographs of the holotype (UM-COS-1012) maxilla (A) and UM-COS-1013 dentary (B) in lateral (A1, B1) and medial (A2, B2) views.
Dentition: The following description is of the maxillary and dentary teeth (both exhibit an identical morphology, although note that only two teeth are preserved in the dentary, Fig. 4). Tooth attachment is pleurodont and tooth replacement occurs lingually. The dental crest (labial wall) is high, and teeth exceed this structure only by one quarter of their length or less. The teeth are tall and conical, with rounded crowns. They are slender, being slightly more robust only in the posterior part of the tooth row. Their apices possess two cusps, labial and lingual, which are separated by a concave sulcus. Because both cusps are inclined medially, the sulcus is V-shaped (Fig. 4A2, B1).
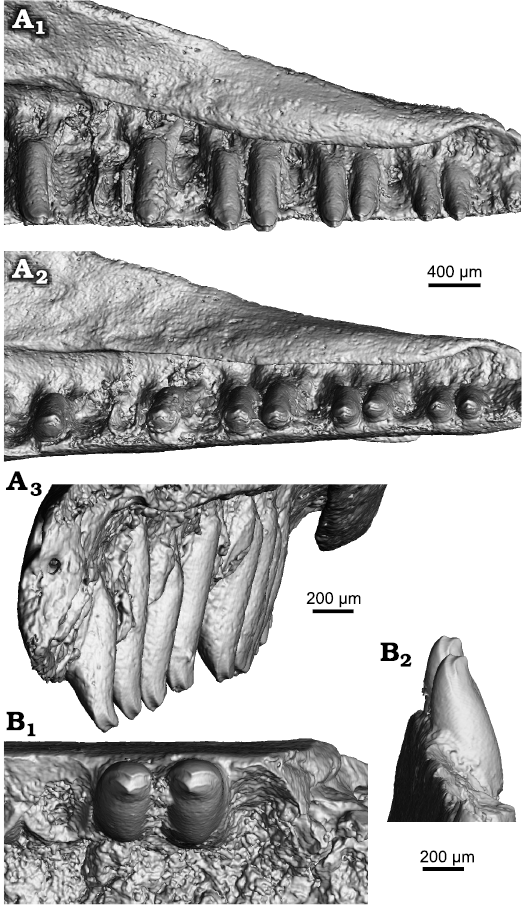
Fig. 4. Pan-gekkotan squamate Bauersaurus cosensis gen. et sp. nov. from Cos, France, early Eocene (MP 10–11). Tooth details of the holotype (UM-COS-1012) maxilla (A) and UM-COS-1013 dentary (B) in ventromedial (A1), ventral (A2), anteromedial (A3), dorsal (B1), anterior (B2) views (all micro-CT visualizations).
Remarks.—Although it is very approximate to estimate the size of an animal using a few disarticulated bones, using the proportions of a comparable living species (Pseudothecadactylus australis), Bauersaurus cosensis gen. et sp. nov. would have been a medium-sized gecko with an estimated skull length 23 mm, and snout-vent length (SVL) of 70 mm. This species is too incomplete to be included on a large scale morphological data set, but based on the overall morphology of the two bones preserved and the cylindrical/conical teeth with transversally bicuspid tooth crowns, it is provisionally assigned to total clade Pan-Gekkota.
Stratigraphic and geographic range.—Type locality and horizon only.
Discussion
The distribution, richness, and diversity of squamates is highly dependent on climatic conditions, and climate change has a marked impact on their life history (Sinervo et al. 2010; Rage 2013). The Cos locality is an important deposit because it documents the paleobiodiversity of lizards during the early Eocene “greenhouse world” in Europe (Čerňanský et al. 2023). Considering the scarcity of early Eocene herpetofaunas from Eurasia, the new pan-gekkotan described here is a rare finding, possibly documenting the persistence of a lineage beyond the K/T boundary, and adding a new taxon to the early Eocene lizard paleodiversity in Europe. Bauersaurus gen. nov. exhibits an atypical morphology, not previously known in any early Paleogene forms. Today, members of Gekkota are distributed worldwide from warm temperate to tropical areas (Bauer 2013; Meiri 2020), although some species reach temperate regions in Asia, Patagonia, and New Zealand (Szczerbak and Golubev 1996; Bauer 2013; Daza et al. 2017). In Europe, gekkotans such as Euleptes, Tarentola, Hemidactylus, and Mediodactylus Szczerbak and Golubev, 1977, are today distributed in the areas along the Mediterranean region, including southern France, Italy, and Greece (Uetz et al. 2023). Bauersaurus gen. nov. differs morphologically from all the living and known fossil members of Gekkota. However, Bauersaurus gen. nov. extends the history of Pan-Gekkota in southern France, at least, to the early Eocene, and maybe even more recently as indicated by undescribed material from Messel pits (Krister Smith personal communication 2022; Smith et al. 2018).
Taxonomic allocation of the Cos fossils and comparison.—These two bones are assigned to the same species based on their provenance, similar proportions and, especially by their identical tooth morphology. The maxilla and dentary described here can be referred to Gekkota based on the following characters (Estes 1983; Daza et al. 2014): (i) absence of osteoderms fused to the facial process; (ii) tubular dentary with fused margins of the Meckelian canal (i.e., no indication of suture); and (iii) the high number of conical pleurodont teeth.
The Bauersaurus cosensis gen. et sp. nov. maxilla shows several unique features (see Diagnosis and Description sections), distinguishing it from all other known Eocene taxa (Fig. 5) as well as extant members of the clade Gekkota (Fig. 6). Bauersaurus cosensis gen. et sp. nov. is similar in age to Laonogekko lefevrei from France (MP 10; Augé 2003), but clearly differs from it by several features, such as (i) the overall shape of maxilla: Bauersaurus cosensis gen. et sp. nov. has a robust appearance, whereas in L. lefevrei, it appears to be anteroposteriorly elongated rather than robust; (ii) length of the facial process; (iii) rather smooth surface of the facial process (vs. faint sculpture in L. lefevrei, for surface sculpturing in geckos, see Glynne et al. 2020); (iv) presence of the posterior free terminus on the posterior margin of the facial process; (v) absence of a well-defined long posteroventral process; (vi) presence of a trough on the ventral surface of the maxillary medial shelf; (vii) presence of a bony septum in the posterior region that partly covers the teeth in medial view; (viii) much anteriorly distributed supralabial foramina; and (ix) the maxilla of L. lefevrei has a low sloping posterior margin of the facial process, that is still more inclined relative to the Cos taxon. The anterior margin of the facial in L. lefevrei (the triangular tip here is present in both taxa) is more inclined ventrally relative to that in B. cosensis gen. et sp. nov.
The maxilla of Bauersaurus cosensis gen. et sp. nov. differs from that of Geiseleptes delfinoi from Geiseltal (MP 12 and MP 13, respectively) by (i) low sloping posterior margin of the facial process rather than steeply inclined; (ii) absence of the groove posterior to the last supralabial foramen; and by the most features observed above in comparison to Laonogekko (ii, iv–viii).
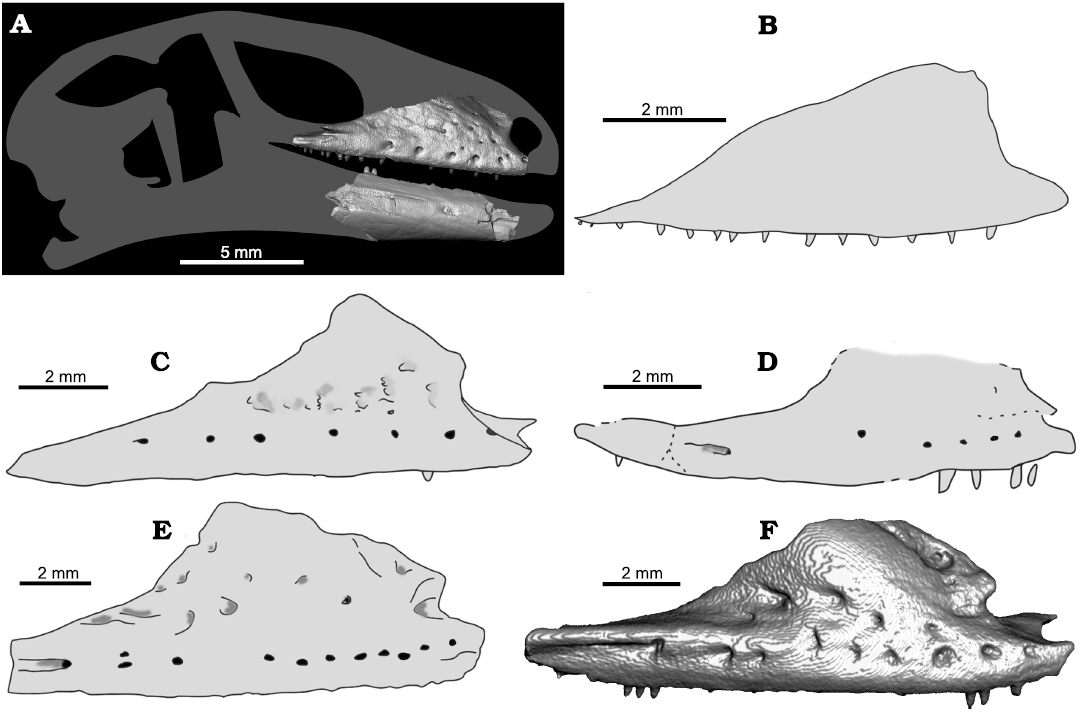
Fig. 5. A. Reconstruction of the complete skull of an early Eocene pan-gekkotan squamate Bauersaurus cosensis gen. et sp. nov. from Cos, France, using a 3D model of a living gekkotan with similar morphology and proportions of bones, Pseudothecadactylus australis (MCZ Herp R- 35162), grey silhouette; Bauersaurus, micro-CT visualizations of jaws in lateral view. B–F. Comparison of the overall shape of the facial process with other gekkotans. B. The Late Jurassic Eichstaettisaurus schroederi (holotype BSPG 1937 I; modified from Simões et al. 2017). C. The early Eocene (MP 10) Laonogekko lefevrei (holotype MNHN PMT 5; modified from Augé 2005). D. The middle Eocene (MP 12 and MP 13) Geiseleptes delfinoi (holotype GMH Ce IV-4057-1933; modified from Villa et al. 2022). E. The middle–late Eocene (MP 16–19) Cadurcogekko piveteaui (the right maxilla MNHN QU 17734; modified from Augé 2005). F. Cadurcogekko cf. piveteaui (mirrored left maxilla NHMW 2019/0052/0001 originally described by Georgalis et al. 2021).
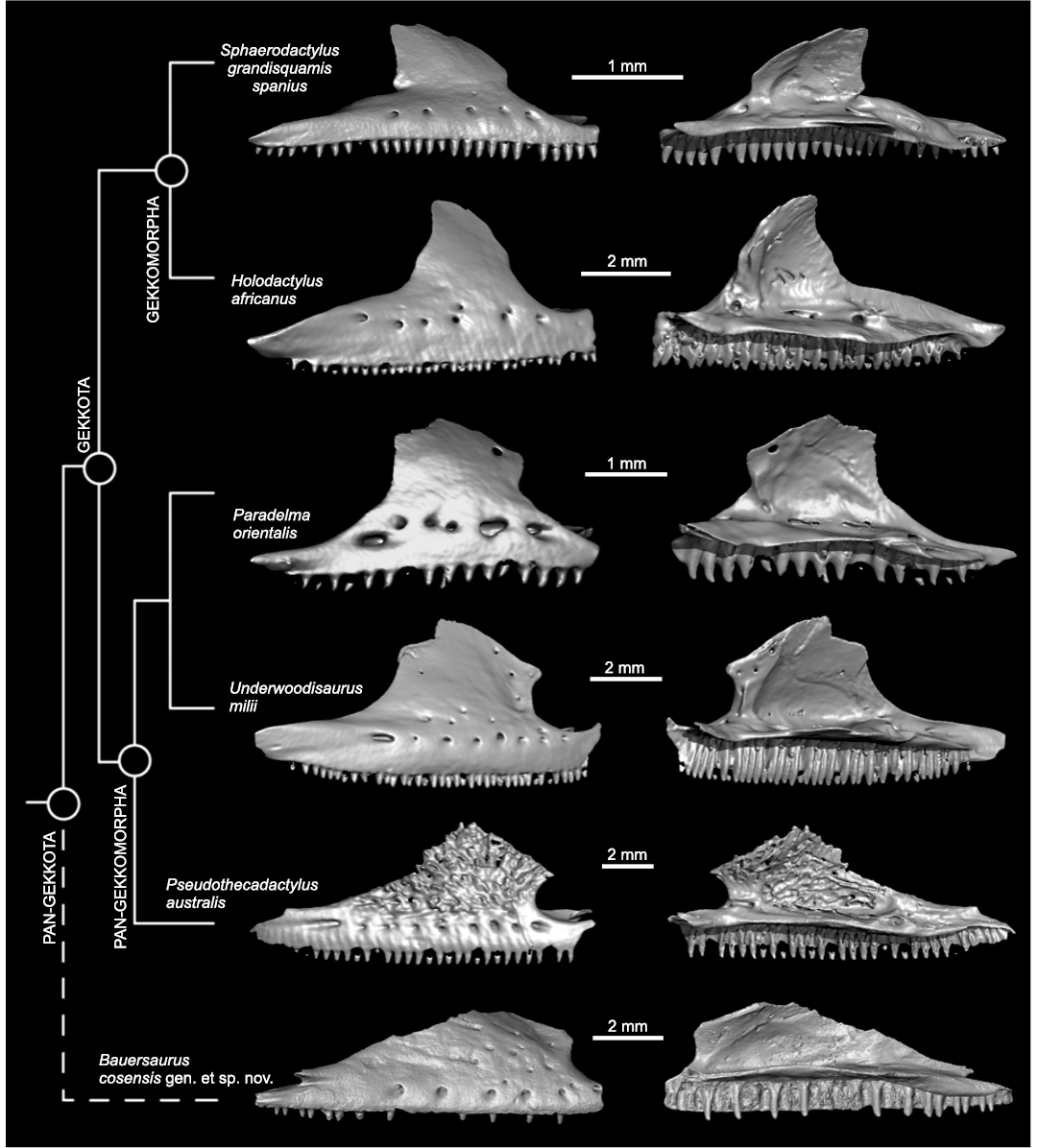
Fig. 6. Maxillae of several gekkotans compared with Bauersaurus gen. nov. in lateral (left column) and medial (right column) views; 3D models of gekkotans were mirrored. Top to bottom, Sphaerodactylus grandisquamis spanius Stejneger, 1904, RT 14708 (Sphaerodactylidae), Holodactylus africanus Boettger, 1893, CAS 198932 (Eublepharidae), Paradelma orientalis (Günther, 1876) CAS 77652 (Pygopodide), Underwoodisaurus milii Bory de Saint-Vincent, 1823, CAS 74744 (Carphodactylidae), Pseudothecadactylus australis (Günther, 1877) MCZ Herp R- 35162 (Diplodactylide), Bauersaurus cosensis the holotype UM-COS-1012 (Pan-Gekkota). Bauersaurus cosensis is inferred as sister to Gekkota.
Bauersaurus cosensis gen. et sp. nov. differs from younger Cadurcogekko piveteaui from the Phosphorites du Quercy (MP 16–19) by (i) low sloping posterior margin of the facial process rather than steeply inclined; (ii) smaller number of supralabial foramina (six vs. ~ten of much posteriorly distributed foramina); (iii) absence of the groove posterior to the last supralabial foramen and by the most features observed above in comparison to Laonogekko (iii–viii).
A direct comparison with older Dollogekko from Dormaal (MP 7, ~6–7 million years older) and younger Rhodanogekko from Lissieu (MP 14; ~7–8 million years younger) is not possible due to their different currently available elements—these two taxa are based on isolated single frontals (although dentaries from Dormaal are described as Gekkota indet. 1 and 2, Čerňanský et al. 2022).
The presence of a trough on the ventral side of the maxilla medial to the teeth is a very interesting feature. It is not present in any other Paleogene geckos. In modern geckos, it is present in the eublepharid Aeluroscalabotes Boulenger, 1885 (Grismer 1988; Digimorph.org, 2002–2012). In all others, except for the diplodatylid Naultinus elegans and pygopod Lialis jicaris, this portion of the maxilla is smooth (Grismer 1988). Another shared feature with members of Eublepharidae is the transversally bicuspid teeth with labial and lingual cutting edges running mesiodistally and lingually oriented (V-shaped in ventral view), separated by a concave sulcus. This condition is present mainly in eublepharids (see Sumida and Murphy 1987), but a similar condition is also present in other modern lineages (Fig. 7). Moreover, this condition has been reported for the middle Eocene sphaerodactylid Geiseleptes delfinoi (Villa et al. 2022) and for Gekkota indet 2. from Dormaal (Čerňanský et al. 2022). There are at least two recorded taxa from the Dormaal locality, which are older on geological time scale. The Cos dentary slightly resembles the mandible from Dormaal described by Čerňanský et al. (2022) as Gekkota indet. 2 in the following combination of features: (i) the robustness; (ii) the dorsoventral height of the dental crest being less than the dorsoventral height of the ventral portion of the bone (i.e., the part of the bone ventral to the alveolar margin); and (iii) the above mentioned transversally bicuspid teeth. Based on such limited material, however, it remains unknown if these two taxa represent one lineage.
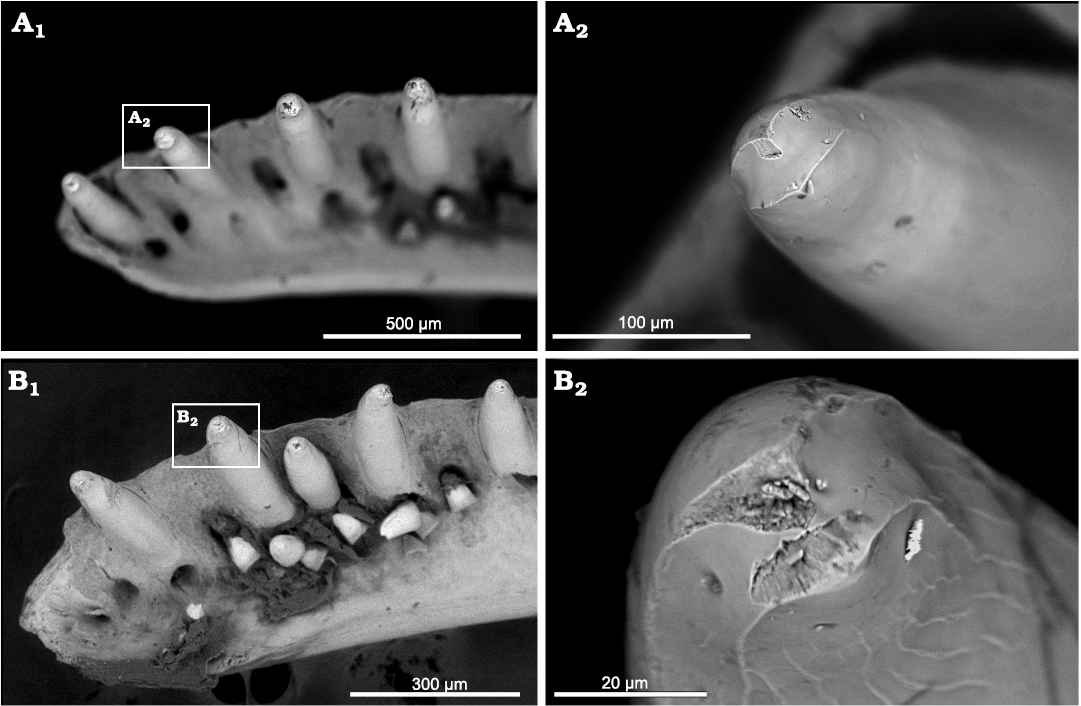
Fig. 7. SEM images of the jaw symphyseal region and details of tooth crowns in two extant gekkotans, Gonatodes albogularis (Duméril and Bibron, 1836) (A) and Sphaerodactylus klauberi Grant, 1923 (B), showing a similar sulcus on the crown as present in Bauersaurus gen. nov.
Transversally bicuspid teeth can be present, however, in other lizard clades, e.g., in Scincoidea (sensu Brownstein et al. 2022: Scincidae, Cordyliformes, Xantusiidae, and the fossil Globauridae, Carusiidae, but not Paramacellodidae, which was included in the larger Pan-Scincoidea). On the other hand, members of this clade usually have very clearly defined lingual cups (the presence of the lingual, well separated cusp is more common among the cordylids than the scincids; Folie et al. 2005). In scincids, the lingual cusp is usually framed by broadly medially and distally running cristae lingualis anterior and posterior, whereas in the Mesozoic Paramacellodidae, there are more-or-less vertical striae dominans anterior and posterior (e.g., Richter 1994; Caputo 2004; Kosma 2004; Čerňanský et al. 2020; Čerňanský and Syromyatnikova 2021). These seems to be different to Bauersaurus gen. nov., although note that the tendency toward closure of the Meckelian canal could be a characteristic of Scincidae as well, as many scincid lizards (not all) show a closed Meckelian canal, in contrast to cordylids (Greer 1970, 1974; Rieppel 1981; Estes 1983; Evans 2008; Augé and Smith 2009; Hutchinson and Scanlon 2009; Gauthier et al. 2012; Čerňanský et al. 2020, Čerňanský and Syromyatnikova 2021). The Meckelian canal in paramacellodids is open (e.g., Estes 1983). Transversely bicuspid teeth can be also present in teiids (in members of this group, however, the teeth have extensive cementum depositions on tooth bases, see Estes 1983) and polyglyphanodontians, but the lingual cusp is much more well developed in these taxa (see Nydam 1999). In lacertids, the lingual cusp, if present, is only weakly developed and, moreover, the Meckelian canal in lacertids is widely open (Kosma 2004; Čerňanský and Syromyatnikova 2019).
The Cos taxon resembles Mesozoic forms rather than modern geckos.—The Cretaceous/Palaeogene (K/Pg) extinction clearly had an impact on squamate faunas too (see Longrich et al. 2012). In some cases, however, a high survivorship across the K/Pg boundary (e.g., Pan-Xenosauridae) is documented (Longrich et al. 2015; Smith et al. 2022). The maxilla of Bauersaurus gen. nov. is atypical with respect to the shape of the facial process when compared to modern geckos (Fig. 6). It shares features of the maxillary shape with the Late Jurassic Eichstaettisaurus schroederi from Solnhofen in Germany (see Simões et al. 2017: fig. 3 and Fig. 5B here; note that the exact shape of the facial process in Eichstaettisaurus gouldi Evans, Raia, and Barbera, 2004, from the Lower Cretaceous of Italy is unknown, see Evans et al. 2004), the steep angle of the anterior margin of the facial process, in combination with a posterior margin of the facial process that slopes gradually dorsoventrally to the jugal facet, that is, the posterior margin reaches almost to the end of the maxilla in contrast to other geckos where there is usually a distinct posterior maxillary process. Eichstaettisaurus schroederi possesses a long jugal, which is not present in modern gekkotans. Its presence is plesiomorphic for Lepidosauria (Simões et al. 2017). In Bauersaurus gen. nov., the jugal facet suggests that the jugal was well braced to the maxilla, as indicated by a lateral facet of the facial process. However, given the paucity of Mesozoic and Paleogene gekkotan fossils, and the apparently even greater paucity of well-preserved maxillae, caution is needed. Although it is worth noting the similarities in shape between the Cos maxilla and that of the Late Jurassic Eichstaettisaurus schroederi, it does not necessarily indicate their closer relationship. More complete material of this and other early Eocene forms might shed more light to this issue. Bauersaurus gen. nov. might thus either represent a “survivor”, a lineage that persisted from the Mesozoic to the early Eocene in Europe or perhaps a morphology otherwise unrepresented in “Modern” geckos. For all these reasons, we favor the allocation of Bauersaurus gen. nov. to Pan-Gekkota.
Conclusions
The presence of Bauersaurus gen. nov. in the early Eocene of the Cos locality provides new insight into the morphology and evolution of pan-gekkotans during the Paleogene. The unique morphology of this lizard documents a group of pan-gekkotans with a facial process of the maxilla, more similar to Mesozoic genus Eichstaettisaurus than to the extant gekkotans. We propose that Bauersaurus gen. nov. might either represent a lineage that persisted from the Mesozoic or that Bauersaurus might be part of an unrepresented lineage within crown group Gekkota. In both cases, its placement in a new lineage is largely justified. Together with the nearly contemporary species of Laonogekko from France (MP 10), and Dollogekko from Belgium (MP 7), Bauersaurus cosensis gen. et sp. nov. adds to the known diversity of species of Pan-Gekkota living in Western Europe during the early Eocene.
Acknowledgements
For access to this fossil material, we thank Mehdi Mouana (Institut des Sciences de l’Evolution de Montpellier, France). We are grateful to Aaron M. Bauer (Villanova University, Villanova, USA) for access to comparative material from extant geckos, and for his opinion on the identification of Bauersaurus. We also thank Justin Williams (Sam Houston State University, Huntsville, USA) for assistance acquiring SEM images and to Jessie Maisano and Matt Colbert (both University of Texas, High-Resolution X-Ray CT Facility, Austin, USA) for access to comparative CT scan material. For critically reading the manuscript, we thank Susan E. Evans (University College London, UK) and Georgios Georgalis (Institute of Systematics and Evolution of Animals, Polish Academy of Sciences, Kraków, Poland). This work was supported by the Scientific Grant Agency of the Ministry of Education of Slovak Republic and Slovak Academy of Sciences, Grant Nr. 1/0191/21.
References
Alifanov, V.R. 1989. The oldest gecko (Lacertilia, Gekkonidae) from the Lower Cretaceous of Mongolia [in Russian]. Paleontologičeskij žurnal 1989 (1): 124–126.
Arnold, E.N. and Poinar, G.A. 2008. 100 million year old gecko with sophisticated adhesive toe pads, preserved in amber from Myanmar. Zootaxa 1847: 62–68. Crossref
Augé, M.L. 2003. La faune de Lacertilia (Reptilia, Squamata) de l’Eocène inférieur de Prémontré (Bassin de Paris, France). Geodiversitas 25: 539–574.
Augé, M.L. 2005. Évolution des lézards du Paléogène en Europe. Muséum national d’Histoire naturelle, Paris. Mémoires du Museum national d’histoire naturelle 192: 1–369.
Augé, M.L. and Smith, R. 2009. An assemblage of early Oligocene lizards (Squamata) from the locality of Boutersem (Belgium), with comments on the Eocene–Oligocene transition. Zoological Journal of the Linnean Society 155: 14–170. Crossref
Bauer, A.M. 2013. Geckos: The Animal Answer Guide. 159 pp. Johns Hopkins University Press, Baltimore. Crossref
Bauer, A.M. 2020. Pan-Gekkota A.M. Bauer, new clade name. In: K. de Queiroz, P.D. Cantino, and J.A. Gauthier (eds.), Phylonyms: A Companion to the PhyloCode, 1109–1111. CRC Press, Boca Raton. Crossref
Bauer, A.M, Böhme, W., and Weitschat, W. 2005. An Early Eocene gecko from Baltic amber and its implications for the evolution of gecko adhesion. Journal of Zoology 265: 327–332. Crossref
Blyth, E. 1854. Proceedings of the Society. Report of the Curator, Zoological Department. The Journal of the Asiatic Society of Bengal 23: 737–740.
Boettger, O. 1893. Übersicht der von Prof. C. Keller anlässlich der Ruspoli’schen Expedition nach den Somaliländern gesammelten Reptilien und Batrachier. Zoologischer Anzeiger 16 (416): 113–119.
Böhme, W. 1984. Erstfund eines fossilien Kugelfingergeckos (Sauria: Gekkonidae: Sphaerodactylinae) aus Dominikanischem Bernstein (Oligozän von Hispaniola, Antillen). Salamandra 20: 212–220.
Böhme, W. and Meier, H. 1980. Revision der madagassischen Homopholis (Blaesodactylus)-Arten (Sauria: Gekkonidae). Senckenbergiana Biologica 60 (5–6) [for 1979]: 303–315.
Bolet, A. 2017. First early Eocene lizards from Spain and a study of the compositional changes between late Mesozoic and early Cenozoic Iberian lizard assemblages. Palaeontologica Electronica 20.2.20A: 1–22. Crossref
Bolet, A., Daza, J.D., Augé, M.L., and Bauer, A.M. 2015. New genus and species names for the Eocene lizard Cadurcogekko rugosus Augé, 2005. Zootaxa 3985: 265–274. Crossref
Borsuk-Białynicka, M. 1990. Gobekko cretacicus gen. et. sp. n., a new gekkonid lizard from the Cretaceous of the Gobi Desert. Acta Palaeontologica Polonica 35: 67–76.
Bory de Saint-Vincent, J.B.G.M. 1823. Dictionnaire Classique d’Histoire Naturelle. Volume 7. 640 pp. Rey et Gravier, Paris.
Boulenger, G.A. 1885. Catalogue of the Lizards in the British Museum (Natural History). Vol. 2, Second edition. xiii + 497 pp. British Museum (Natural History), London.
Broili F. 1938. Ein neuer Fund von? Ardeosaurus H. v. Meyer. Sitzungsberichte Bayerische Akademie der Wissenschaften zu München Mathematisch-Naturwissenschaftliche Abteilung 1938: 97–114.
Brownstein, C.D., Meyer, D.L., Fabbri, M., Bhullar B.-A.S., and Gauthier, J.A. 2022. Evolutionary origins of the prolonged extant squamate radiation. Nature Communications 13: 7087. Crossref
Bukejs, A., Alekseev, V.I., and Pollock, D.A. 2019. Waidelotinae, a new subfamily of Pyrochroidae (Coleoptera: Tenebrionoidea) from Baltic amber of the Sambian Peninsula and the interpretation of Sambian amber stratigraphy, age, and location. Zootaxa 4664: 261–273. Crossref
Caldwell, M.W., Nydam, R.L., Palci, A., and Apesteguía, S. 2015. The oldest known snakes from the Middle Jurassic–Lower Cretaceous provide insights on snake evolution. Nature Communications 6: 5996. Crossref
Caputo, V. 2004. The cranial osteology and dentition in the scincid lizards of the genus Chalcides (Reptilia, Scincidae). Italian Journal of Zoology 2: 35–45. Crossref
Čerňanský, A. and Syromyatnikova, E.V. 2019. The first Miocene fossils of Lacerta cf. trilineata (Squamata, Lacertidae) with a comparative study of the main cranial osteological differences in green lizards and their relatives. PLoS ONE 14: e0216191. Crossref
Čerňanský, A. and Syromyatnikova, E.V. 2021. The first Pre-Quaternary fossil record of the clade Mabuyidae with a comment on the enclosure of the Meckelian canal in skinks. Papers in Palaeontology 7: 195–215. Crossref
Čerňanský, A., Daza, J.D., and Bauer, A.M. 2018. Geckos from the middle Miocene of Devínska Nová Ves (Slovakia): new material and a review of the previous record. Swiss Journal of Geosciences 111: 183–190. Crossref
Čerňanský, A., Daza, J.D., Smith, R., Bauer, A.M., Smith, T., and Folie, A. 2022. A new gecko from the earliest Eocene of Dormaal, Belgium— a thermophilic element of the “greenhouse world”. Royal Society Open Science 9: 220429. Crossref
Čerňanský, A., Syromyatnikova, E.V., Kovalenko, E.S., Podurets, K.M., and Kaloyan, A.A. 2020. The key to understanding the European Miocene Chalcides (Squamata, Scincidae) comes from Asia: the lizards of the East Siberian Tagay locality (Baikal Lake) in Russia. The Anatomical Record—Advances in Integrative Anatomy and Evolutionary Biology 303: 1901–1934. Crossref
Čerňanský, A., Tabuce, R., and Vidalenc, D. 2023. Anguimorph lizards from the early Eocene (MP 10–11) of the Cos locality, Phosphorites du Quercy, France and the early evolution of Glyptosaurinae in Europe. Journal of Vertebrate Paleontology 42: e2211646. Crossref
Conrad, J.L. and Daza, J.D. 2015. Naming and rediagnosing the Cretaceous gekkonomorph (Reptilia, Squamata) from Öösh (Övörkhangai, Mongolia). Journal of Vertebrate Paleontology 35: e980891. Crossref
Conrad, J.L. and Norell, M. 2006. High-resolution X-ray computed tomography of an Early Cretaceous gekkonomorph (Squamata) from Öösh (Övorkhangai; Mongolia). Historical Biology 18: 405–431. Crossref
Couper, P.J., Covacevich, J.A., and Moritz, C. 1993. A review of the leaf-tailed geckos endemic to eastern Australia: a new genus, four new species, and other new data. Memoirs of the Queensland Museum 34: 95–124.
Cramwinckel, M.J., Huber, M., Kocken, I.J., Agnini, C., Bijl, P.K., Bohaty, S.M., Frieling, J., Goldner, A., Hilgen, F.J., Kip, E.L., Peterse, F., Van der Ploeg, R., Röhl, U., Schouten, S., and Sluijs, A. 2018. Synchronous tropical and polar temperature evolution in the Eocene. Nature 559 (7714): 382–386. Crossref
Cuvier, G.J.L.N.F.D. 1829. Le Regne Animal Distribué, d’apres son Organisation, pur servir de base à l’Histoire naturelle des Animaux et d’introduction à l’Anatomie Comparé. Nouvelle Edition [second edition]. Vol. 2. xvi + 406 pp. Les Reptiles. Déterville, Paris.
Daza, J.D. and Bauer, A.M. 2012. A new amber-embedded sphaerodactyl gecko from Hispaniola, with comments on the morphological synapomorphies of the Sphaerodactylidae. Breviora 529: 1–28. Crossref
Daza, J.D., Alifanov, V.R., and Bauer, A.M. 2012. A redescription and phylogenetic reinterpretation of the fossil lizard Hoburogekko suchanovi Alifanov, 1989 (Squamata, Gekkota), from the Early Cretaceous of Mongolia. Journal of Vertebrate Paleontology 32: 1303–1312. Crossref
Daza, J.D., Bauer, A.M., and Snively, E. 2013. Gobekko cretacicus (Reptilia: Squamata) and its bearing on the interpretation of gekkotan affinities. Zoological Journal of the Linnean Society 167: 430–448. Crossref
Daza, J.D., Bauer, A.M., and Snively, E. 2014. On the gekkotan fossil record. The Anatomical Record—Advances in Integrative Anatomy and Evolutionary Biology 297: 433–462. Crossref
Daza, J.D., Bauer, A.M., Sand, C., Lilley, I., Wake, T.A., and Valentin, F. 2015. Reptile remains from Tina (Tokanod), Loyalty Islands, New Caledonia. Pacific Science 69: 531–557. Crossref
Daza, J.D., Bauer, A.M., Wagner, P., and Böhme, W. 2013. A reconsideration of Sphaerodactylus dommeli Böhme, 1984 (Squamata: Gekkota: Sphaerodactylidae), a Miocene lizard in amber. Journal of Zoological Systematics and Evolutionary Research 51: 55–63. Crossref
Daza, J.D., Gamble, T., Abdala, V., and Bauer, A.M. 2017. Cool geckos: Does plesiomorphy explain morphological similarities between geckos from the Southern cone. Journal of Herpetology 51: 330–342. Crossref
Daza, J.D., Stanley, E.L., Wagner, P., Bauer, A.M., and Grimaldi, D.A. 2016. Mid-Cretaceous amber fossils illuminate the past diversity of tropical lizards. Science Advances 4: e1501080. Crossref
de Vis, C.W. 1884. On new Australian lizards. The Proceedings of the Royal Society of Queensland 1: 53–56. Crossref
Digimorph.org. 2002–2012. Digital morphology: a national science foundation digital library at the University of Texas at Austin [internet]. Austin, TX: The High Resolution X-ray Computed Tomography Facility at the University of Texas at Austin. Available at: http://www.digimorph.org/. Accessed May 2023.
Duméril, A.M.C. 1806. Zoologie analytique, ou méthode naturelle de classification des animaux. xxxii + 344 pp. Allais, Paris.
Duméril, A.M.C. and Bibron, G. 1836. Erpetologie Générale ou Histoire Naturelle Complete des Reptiles. Vol. 3. 528 pp. Libr. Encyclopédique Roret, Paris.
Eberle, J.J., Fricke, H.C., Humphrey, J.D., Hackett, L., Newbrey, M.G., and Hutchison, J.H. 2010. Seasonal variability in Arctic temperatures during early Eocene time. Earth and Planetary Science Letters 296: 481–486. Crossref
Eberle, J.J., Gottfried, M.D., Hutchison, J.H., and Brochu, C.A. 2014. First Record of Eocene Bony Fishes and Crocodyliforms from Canada’s Western Arctic. PloS ONE 9: e96079 Crossref
Estes, R. 1983. Encyclopedia of Paleoherpetology, Part 10A. Sauria terrestria, Amphisbaenia. 249 pp. Gustav Fisher Verlag, Stuttgart.
Estes, R. and Hutchison, J.H. 1980. Eocene lower vertebrates from Ellesmere Island, Canadian Arctic Archipelago. Palaeogeography, Palaeoclimatology, Palaeoecology 30: 325–347. Crossref
Evans, S.E. 1991. A new lizard-like reptile (Diapsida: Lepidosauromorpha) from the Middle Jurassic of England. Zoological Journal of the Linnean Society 103: 391–412. Crossref
Evans, S.E. 1994. A new anguimorph lizard from the Jurassic and lower Cretaceous of England. Palaeontology 37: 33–49.
Evans, S.E. 2008. The skull of lizards and tuatara. In: C. Gans, A.S. Gaunt, and K. Adler (eds.), Biology of the Reptilia, Vol. 20, Morphology H, The skull of Lepidosauria, 1348. Society for the Study of Reptiles and Amphibians, Ithaca.
Evans, S.E. 2022. The origin and early diversification of squamates. In: D.J. Gower and H. Zaher (eds.), The Origin and Early Evolutionary History of Snakes, 7–25. Cambridge University Press, Cambridge. Crossref
Evans, S.E. and Borsuk-Białynicka, M. 2009. A small lepidosauromorph reptile from the Early Triassic of Poland. Paleontologica Polonica 65: 179–202.
Evans, S.E., Raia, P., and Barbera, C. 2004. New lizards and rhynchocephalians from the Lower Cretaceous of southern Italy. Acta Palaeontologica Polonica 49: 393–408.
Fitzinger, L. 1843. Systema Reptilium, fasciculus primus, Amblyglossae. 106 pp. Braumüller et Seidel, Wien. Crossref
Folie, A., Sigé, B., and Smith, T. 2005. A new scincomorph lizard from the Palaeocene of Belgium and the origin of Scincoidea in Europe. Naturwissenschaften 92: 542–546. Crossref
Fontanarrosa, G., Daza, J.D., and Abadala, V. 2018. Cretaceous fossil gecko hand reveals a strikingly modern scansorial morphology: Qualitative and biometric analysis of an amber-preserved lizard hand. Cretaceous Research 84: 120–133. Crossref
Gauthier, J.A., Kearney, M., Maisano, J.A., Rieppel, O., and Behlke, A.D.B. 2012. Assembling the squamate tree of life: perspectives from the phenotype and the fossil record. Bulletin of the Peabody Museum of Natural History 53: 3–308. Crossref
Gené, J. 1839. Synopsis reptilium Sardiniae indigenoruni. Memorie della Reale Accademia delle Scienze di Torino 1: 257—285.
Georgalis, G.L., Čerňanský, A., and Klembara, J. 2021. Osteological atlas of new lizards from the Phosphorites du Quercy (France), based on historical, forgotten, fossil material. In: J.S. Steyer, M.L. Augé, and G. Métais (eds.), Memorial Jean-Claude Rage: A Life of Paleo-herpetologist. Geodiversitas 43: 219–293. Crossref
Glynne, E., Daza, J.D., Bauer, A.M. 2020. Surface sculpturing in the skull of gecko lizards (Squamata: Gekkota). Biological Journal of the Linnean Society 131: 801–813. Crossref
Grant, C. 1931. The sphaerodactyls of Porto Rico, Culebra and Mona islands. The Journal of the Department of Agriculture of Porto Rico 15: 199–213. Crossref
Gray, J.E. 1825. A synopsis of the genera of reptiles and amphibia, with a description of some new species. Annals of Philosophy, London 10: 193–217.
Gray, J.E. 1827. A Synopsis of the Genera of Saurian Reptiles in which some new Genera are indicated, and the others reviewed by actual Examination. Philosophical Magazine, London 2: 54–58. Crossref
Gray, J.E. 1845. Description of a new genus of night lizards from Belize. The Annals and Magazine of Natural History; Zoology, Botany, and Geology 16: 162–163. Crossref
Griffiths, E.F., Ford, D.P., Benson, R.B.J., and Evans, S.E. 2021. New information on the Jurassic lepidosauriomorph Marmoretta oxoniensis. Papers in Paleontology 7: 2255–2278. Crossref
Greenwood, D.R. and Wing, S.L. 1995. Eocene continental climates and latitudinal temperature gradients. Geology 23: 1044–1048. Crossref
Greer, A.E. 1970. A subfamilial classification of scincid lizards. Bulletin, Museum of Comparative Zoology 139: 151–184.
Greer, A.E. 1974. The generic relationships of the scincid lizard genus Leiolopisma and its relatives. Australian Journal of Zoology 31: 1–67. Crossref
Grismer, L.L. 1988. Phylogeny, taxonomy, classification, and biogeography of eublepharid geckos. In: R. Estes and G. Pregill (eds.), Phylogenetic Relationships of the Lizard Families, 369–469. Stanford University Press, Stanford.
Godinot, M., Blondel, C., Escarguel, G., Lézin, C., Pélissié, T., Tabuce, R., and Vidalenc, D. 2021. Primates and Plesiadapiformes from Cos (Eocene; Quercy, France). Geobios 66–67: 153–176. Crossref
Günther, A. 1864. The Reptiles of British India. xxvii + 452 pp. Taylor & Francis, London. Crossref
Günther, A. 1876. Descriptions of new species of reptiles from Australia collected by Hr. Dämel for the Godeffroy Museum. Journal des Museum Godeffroy 5 (12): 45–47.
Günther, A. 1877. Descriptions of three new species of lizards from Islands of Torres Straits. The Annals and Magazine of Natural History 19: 413–415. Crossref
Hoffstetter, R. 1946. Sur les Gekkonidae fossiles. Bulletin du Muséum national d’histoire naturelle 18: 195–203.
Hutchinson, M.N., and Scanlon, J.D. 2009. New and unusual Plio-Pleistocene lizard (Reptilia: Scincidae) from Wellington Caves, New South Wales, Australia. Journal of Herpetology 43: 139–147. Crossref
Hyland, E.G., Sheldon, N. D., and Cotton, J.M. 2017. Constraining the early Eocene climatic optimum: A terrestrial interhemispheric comparison. GSA Bulletin 129: 244–252. Crossref
Inglis, G.N., Bragg, F., Burls, N.J., Cramwinckel, M.J., Evans, D., Foster, G.L., Huber, M., Lunt, D.J., Siler, N., Steinig, S., Tierney, J.E., Wilkinson, R., Anagnostou, E., de Boer, A.M., Dunkley Jones, T., Edgar, K.M., Hollis, C.J., Hutchinson, D.K., and Pancost, R. D. 2020. Global mean surface temperature and climate sensitivity of the early Eocene Climatic Optimum (EECO), Paleocene–Eocene Thermal Maximum (PETM), and latest Paleocene. Climate of the Past 16: 1953–1968. Crossref
Kosma, R. 2004. The Dentitions of Recent and Fossil Scincomorphan Lizards (Lacertilia, Squamata). Systematics, Functional Morphology, Paleoecology. 229 pp. Ph.D. Dissertation, University of Hannover, Hannover.
Kuhn, O. 1958. Ein neuer Lacertilier aus dem fränkischen Lithographieschiefer. Neues Jahrbuch fűr Geologie und Paläontologie, Monatshefte 1958: 380–382.
Laurenti, J.N. 1768. Specimen medicum, exhibens synopsin reptilium emendatam cum experimentis circa venena et antidota reptilium austracorum, quod authoritate et consensu. 217 pp. Joan. Thomae, Vienna. Crossref
Linnaeus, C. 1758. Systema naturæ per regna tria naturæ, secundum classes, ordines, genera, species, cum characteribus, differentiis, synonymis, locis. Tomus I. 10th Edition: 824 pp. Laurentii Salvii, Holmiæ. Crossref
Longrich, N.R., Bhullar, B.A.S., and Gauthier, J.A. 2012. Mass extinction of lizards and snakes at the Cretaceous–Paleogene boundary. Proceedings of the National Academy of Sciences 109: 21396–21401. Crossref
Longrich, N.R., Vinther, J., Pyron, R.A., Pisani, D., and Gauthier, J.A. 2015. Biogeography of worm lizards (Amphisbaenia) driven by end-Cretaceous mass extinction. Proceedings of the Royal Society B: Biological Sciences 282: 20143034. Crossref
Meiri, S. 2020. What geckos are—an ecological-biogeographic perspective. Israel Journal of Ecology and Evolution 66: 1–11. Crossref
Mocquard, F. 1900. Diagnoses d’espèces nouvelles de Reptiles de Madagascar. Bulletin du Muséum national d’Histoire naturelle Paris 6: 345–348. Crossref
Műller, J. and Mödden, C. 2001. A fossil leaf-toed gecko from the Oppenheim/Nierstein Quarry (Lower Miocene, Germany). Journal of Herpetology 35: 532–537. Crossref
Nydam, R.L. 1999. Polyglyphanodontinae (Squamata: Teiidae) from the medial and Late Cretaceous: New records from Utah, U.S.A. and Baja California del Norte, Mexico. In: D.D. Gillettem (ed.), Vertebrate Paleontology in Utah. Utah Geological Survey Miscellaneous Publication 99-1, 303–317. Utah Geological Survey, Salt Lake City.
Oken, L. 1817. Cuvier’s und Oken’s zoologien naben einande gestellt. Isis, Encyclopädische Zeitung 8 (148): 1779–1782.
Oppel, M. 1811. Die Ordnungen, Familien und Gattungen der Reptilien als Prodrom einer Naturgeschichte derselben. 87 pp. Joseph Lindauer, Munich. Crossref
Pross, J, Contreras, L., Bijl, P.K., Greenwood, D.R., Bohaty, S.M., Schouten, S., Bendle, J.A., Röhl, U., Tauxe, L., Raine, J.I., Huck, C.E., Flierdt, T., Jamieson, S.S.R., Stickley, C.E., Schootbrugge, B., Escutia, C., Brinkhuis, H., and Integrated Ocean Drilling Program Expedition 318 Scientists. 2012. Persistent near-tropical warmth on the Antarctic continent during the early Eocene epoch. Nature 488: 73–77. Crossref
Pulou, R. 1980. Jean-André Poumarède et l’industrie des phosphates du Quercy au XIXe siècle. Mémoires de l’Académie des Sciences, Inscriptions et Belles Lettres de Toulouse 16: 83–92.
Pyron, R.A. 2017. Novel approaches for phylogenetic inference from morphological data and total-evidence dating in squamate reptiles (lizards, snakes, and amphisbaenians). Systematic Biology 66: 38–56. Crossref
Rage, J.-C. 2013. Mesozoic and Cenozoic squamates of Europe. In: J.D. Gardner and R.L. Nydam (eds.), Mesozoic and Cenozoic Lissamphibian and Squamate Assemblages of Laurasia. Palaeobiodiversity and Palaeoenvironments 93: 517–534. Crossref
Rage, J.C. and Augé, M.L. 2003. Amphibians and squamate reptiles from the lower Eocene of Silveirinha (Portugal). Ciências da Terra (UNL) 15: 103–116.
Renesto, S. and Bernardi, M. 2014. Redescription and phylogenetic relationships of Megachirella wachtleri Renesto et Posenato, 2003 (Reptilia, Diapsida). Paläontologische Zeitschrift 88: 197–210. Crossref
Renesto, S. and Posenato, R. 2003. A new lepidosauromorph reptile from the Middle Triassic of the Dolomites (Northern Italy). Italian Journal of Paleontology and Stratigraphy 109: 463–474.
Richter, A. 1994. Lacertilia aus der Unteren Kreide von Una und Galve (Spanien) und Anoual (Marokko). Berliner geowissenschaftliche Abhandlungen (E: Paläobiologie) 14: 1–147.
Rieppel, O. 1981. The skull and the jaw adductor musculature in some burrowing scincomorph lizards of the genera Acontias, Typhlosaurus and Feylinia. Journal of Zoology, London 195: 493–528. Crossref
Simões, T.R. and Pyron, R.A. 2021. The squamate tree of life. Bulletin of the Museum of Comparative Zoology 163: 47–95. Crossref
Simões, T.R., Caldwell, M.W., Nydam, R.L., and Jiménez-Huidobro, P. 2017. Osteology, phylogeny, and functional morphology of two Jurassic lizard species and the early evolution of scansoriality in geckoes. Zoological Journal of the Linnean Society 180: 216–241. Crossref
Simões, T.R., Caldwell, M.W., Tałanda, M., Bernardi, M., Palci, A., Vernygora, O., Bernardini, F., Mancini, L., and Nydam, R.L. 2018. The origin of squamates revealed by a Middle Triassic lizard from the Italian Alps. Nature 557: 706–709. Crossref
Simões, T.R., Vernygora, O.V., Caldwell, M.W. and Pierce, S.E. 2020. Megaevolutionary dynamics and the timing of evolutionary innovation in reptiles. Nature Communications 11: 3322. Crossref
Sinervo, B., Mendez-de-la-Cruz, F., Miles, D.B., Heulin, B., Bastiaans, E., Villagran-Santa Cruz, M., Lara-Resendiz, R., Martinez-Mendez, N., Calderon-Espinosa, M.L., Meza-Lazaro, R.N., Gadsden, H., Avila, L.J., Morando, M., De la Riva, I.J., Victoriano Sepulveda, P., Rocha, C.F., Ibarguengoytia, N., Aguilar, Puntriano, C., Massot, M., Lepetz, V., Oksanen, T.A., Chapple, D.G., Bauer, A.M., Branch, W.R., Clobert, J., and Sites, J.W., Jr. 2010. Erosion of lizard diversity by climate change and altered thermal niches. Science 328: 894–899. Crossref
Smith, A. 1846. Illustrations of the Zoology of South Africa, Reptilia. Smith, Elder, and Co., London.
Smith, K.T. and Georgalis, G.L. 2022. The diversity and distribution of Palaeogene snakes: a review with comments on vertebral sufficiency. In: D. Gower and H. Zaher (eds.), The Origin and Early Evolution of Snakes, 55–84. Cambridge University Press, Cambridge. Crossref
Smith, K.T., Bhullar, B.-A.S., and Bloch, J.I. 2022. New diminutive Eocene lizard reveals high K-Pg survivorship and taxonomic diversity of stem xenosaurs in North America. American Museum Novitates 3986:1–36. Crossref
Smith, K.T., Čerňanský, A., Scanferla, A., and Schaal, S.F.K. 2018. Lizards and snakes: warmth-loving sunbathers. In: K.T. Smith, S.F.K. Schaal, and J. Habersetzer (eds.), Messel, An Ancient Greenhouse Ecosystem, 122–147. Senckenberg Gesellschaft für Naturforschung, Frankfurt am Main.
Steindachner, F. 1867. Reptilien. In: B. von Wüllerstorf-Urbair (ed.), Reise der Österreichischen Fregatte Novara um die erde in den Jahren 1857, 1858, 1859. Zoologischer Theil. Erster Band (Wirbelthiere), 1–98. Kaiserlich-Königlichen Hof- und Staatsdrucke, Wien.
Stejneger, L. 1904. The herpetology of Porto Rico. Report of the United States National Museum 1902: 549–724. Crossref
Sumida, S.S. and Murphy, R.W. 1987. Form and function of the tooth crown structure in gekkonid lizards (Reptilia, Squamata, Gekkonidae). Canadian Journal of Zoology 65: 2886–2892. Crossref
Szczerbak, N.N. and Golubev, M.L. 1977. Systematics of the Palearctic geckos (genera Gymnodactylus, Bunopus, Alsophylax) [in Russian]. Proceedings of the Zoological Institute, Academy of Sciences of the USSR, Herpetological Collected Papers 74: 120–133.
Szczerbak, N.N. and Golubev, M.L. 1996. Gecko Fauna of the USSR and Contiguous Regions. 233 pp. Society for the Study of Amphians and Reptiles, Ithaca.
Tałanda, M. 2018. An exceptionally preserved Jurassic skink suggests lizard diversification preceded fragmentation of Pangaea. Palaeontology 61: 659–677. Crossref
Uetz, P., Slavenko, A., Meiri, S., and Heinicke, M. 2020. Gecko diversity: a history of global discovery. Israel Journal of Ecology and Evolution 66: 117–125. Crossref
Uetz, P., Freed, P., and Hošek, J. 2023. The Reptile Database. http://www.reptile-database.org [accessed March 2023].
Vianey-Liaud, M., Vidalenc, D., Orliac M.J., Maugoust, J., Lézin, C., and Pélissié, T. 2022. Rongeurs de la localité éocène de Cos (Tarnet-Garonne, Quercy, France). Comparaison avec les rongeurs de localités de la transition Éocène inférieur/Éocène moyen. Geodiversitas 44: 753–800. Crossref
Villa, V., Daza, J.D., Bauer, A.M., and Delfino, M. 2018. Comparative cranial osteology of European gekkotans (Reptilia, Squamata). Zoological Journal of the Linnean Society 184: 857–895. Crossref
Villa, A., Wings, O., and Rabi, M. 2022. A new gecko (Squamata, Gekkota) from the Eocene of Geiseltal (Germany) implies long-term persistence of European Sphaerodactylidae. Papers in Palaeontology 8 (3): e1434. Crossref
Wagler, J.G. 1830. Natürliches System der Amphibien, mit vorangehender Classification der Säugetiere und Vögel. Ein Beitrag zur vergleichenden Zoologie. 1.0. 354 pp. Cotta, München. Crossref
Zheng, Y., and Wiens, J.J. 2016. Combining phylogenomic and supermatrix approaches, and a time-calibrated phylogeny for squamate reptiles (lizards and snakes) based on 52 genes and 4162 species. Molecular Phylogenetics and Evolution 94: 537–547. Crossref
Acta Palaeontol. Pol. 68 (4): 695–708, 2023
https://doi.org/10.4202/app.01083.2023